Abstract
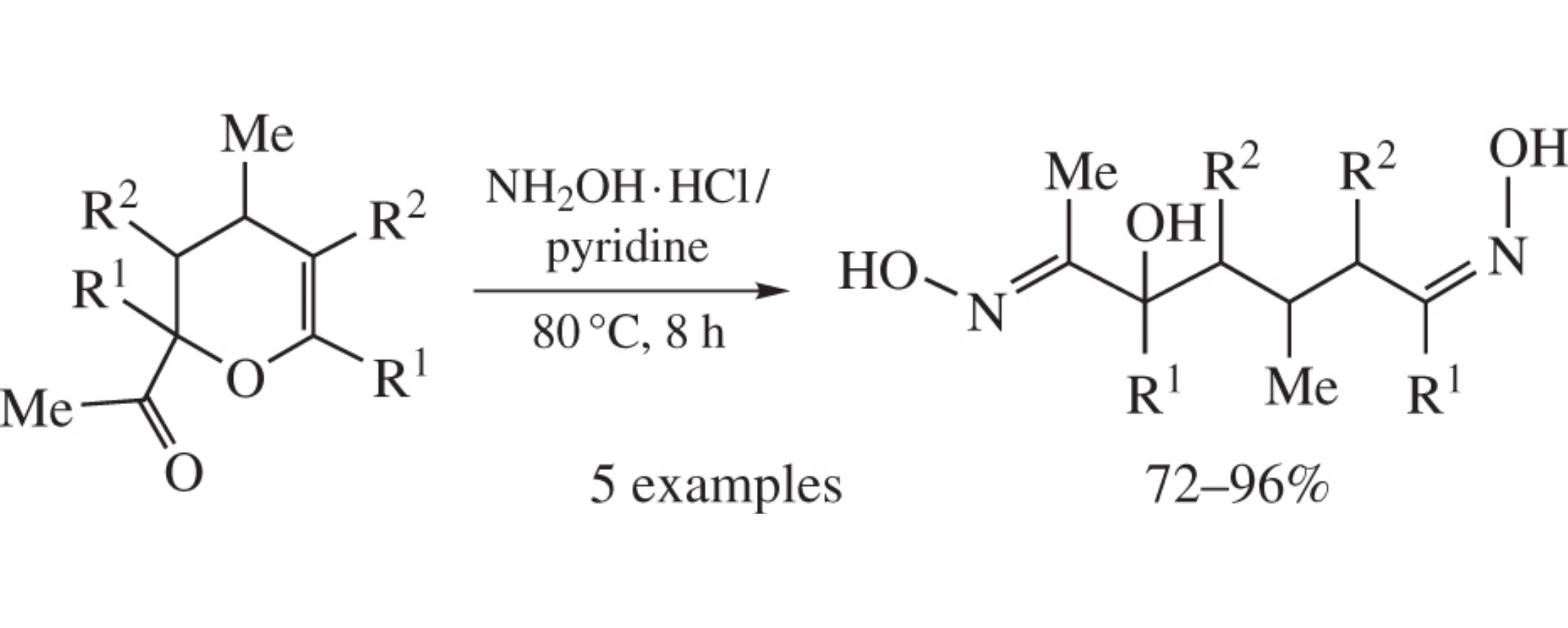
2-Acetyl-3,4-dihydropyrans, synthesized from acetylene and ketones in two steps, react with hydroxylamine to afford 5-hydroxy-1,6-heptanedione dioximes (E,E-isomers) in 72–96% yields.
References
1.
Domino Reactions: Concepts for Efficient Organic Synthesis, ed. L. F. Tietze, Wiley-VCH, Weinheim, 2014.
2.

Trofimov B.A., Schmidt E.Y.
Russian Chemical Reviews,
2014
3.
Rodygin K.S., Kostin A.A., Ananikov V.P.
Mendeleev Communications,
2015
4.

Pirnot M.T., Wang Y., Buchwald S.L.
Angewandte Chemie,
2015
5.

Galkin K.I., Ananikov V.P.
Russian Chemical Reviews,
2016
6.

Rodygin K.S., Werner G., Kucherov F.A., Ananikov V.P.
Chemistry - An Asian Journal,
2016
7.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
8.
Trofimov B.A., Belyaeva K.V., Andriyankova L.V., Nikitina L.P., Mal’kina A.G.
Mendeleev Communications,
2017
9.
Schmidt E.Y., Tatarinova I.V., Ivanova E.V., Trofimov B.A.
Mendeleev Communications,
2017
10.
Petrova O.V., Sobenina L.N., Ushakov I.A., Budaev A.B., Ivanov A.V., Samsonov V.A., Tikhonov A.Y., Trofimov B.A.
Mendeleev Communications,
2017
11.
![One-Pot Assembly of 7-Methylene-6,8-dioxabicyclo[3.2.1]octanes, Congeners of Frontalin, from Ketones and Acetylene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Trofimov B.A., Schmidt E.Y., Ushakov I.A., Mikhaleva A.I., Zorina N.V., Protsuk N.I., Senotrusova E.Y., Skital'tseva E.V., Kazheva O.N., Alexandrov G.G., Dyachenko O.A.
European Journal of Organic Chemistry,
2009
12.
10.1016/j.mencom.2018.03.010_bib0060
Mori
in Topics in Current Chemistry. The Chemistry of Pheromones and Other Semiochemicals I,
2004
13.

Greenwood D.R., Comeskey D., Hunt M.B., Rasmussen L.E.
Nature,
2005
14.

Schmidt E.Y., Trofimov B.A., Zorina N.V., Mikhaleva A.I., Ushakov I.A., Skital'tseva E.V., Kazheva O.N., Alexandrov G.G., Dyachenko O.A.
European Journal of Organic Chemistry,
2010
15.
10.1016/j.mencom.2018.03.010_bib0075
Afonin
Russ. J. Org. Chem,
2000
16.

Mikhaleva A.I., Zaitsev A.B., Trofimov B.A.
Russian Chemical Reviews,
2006
17.

Kölmel D.K., Kool E.T.
Chemical Reviews,
2017