Keywords
imidazothiazolotriazines
isatin
oxindolylidene derivatives
skeletal amidine rearrangement
thiazolidinones
Abstract
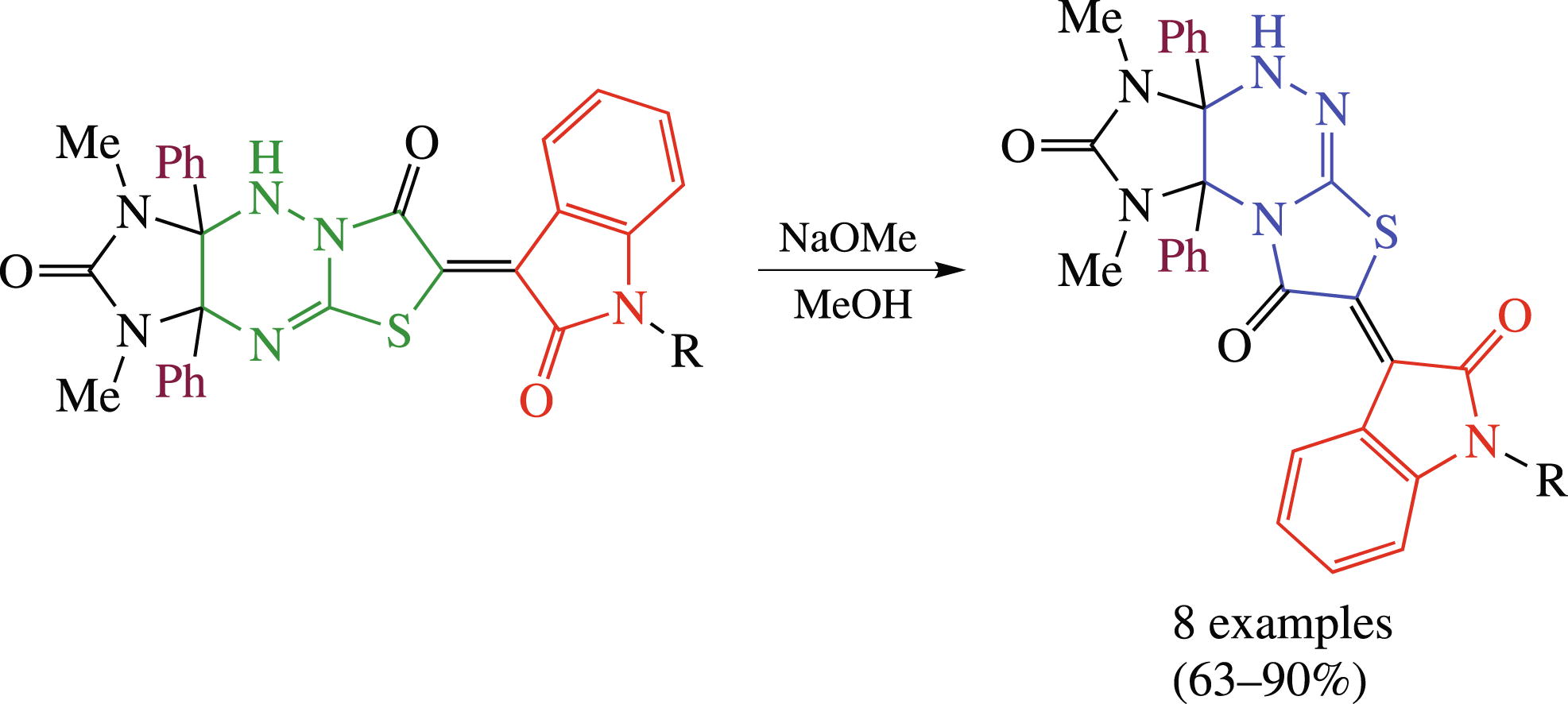
Previously unavailable 7-oxindolylidene-1,3-dimethyl-3a,9adiphenylimidazo[4,5-e]thiazolo[2,3-c][1,2,4]triazines were obtained in 63–90% yields by skeletal amidine rearrangement of the thiazolo-triazine system in the corresponding imidazo[4,5-e]thiazolo[3,2-b][1,2,4]triazines on treatment with sodium methoxide.
References
1.

Singh S.P., Parmar S.S., Raman K., Stenberg V.I.
Chemical Reviews,
1981
2.

Trotsko N.
European Journal of Medicinal Chemistry,
2021
3.

Kolos N.N., Nazarenko N.V., Shishkina S.V., Doroshenko A.O., Shvets E.G., Kolosov M.A., Yaremenko F.G.
Chemistry of Heterocyclic Compounds,
2020
4.

Chadha N., Bahia M.S., Kaur M., Silakari O.
Bioorganic and Medicinal Chemistry,
2015
5.

Nirwan S., Chahal V., Kakkar R.
Journal of Heterocyclic Chemistry,
2019
6.

Gazieva G.A., Izmest’ev A.N.
Chemistry of Heterocyclic Compounds,
2015
7.

Kaminskyy D., Kryshchyshyn A., Lesyk R.
European Journal of Medicinal Chemistry,
2017
8.

Tomasic T., Masic L.
Current Medicinal Chemistry,
2009
9.

Kaur Manjal S., Kaur R., Bhatia R., Kumar K., Singh V., Shankar R., Kaur R., Rawal R.K.
Bioorganic Chemistry,
2017
10.

Izmest'ev A.N., Streltsov A.A., Karnoukhova V.A., Kolotyrkina N.G., Strelenko Y.A., Kravchenko A.N., Gazieva G.A.
ChemistrySelect,
2022
11.

Makhova N.N., Belen’kii L.I., Gazieva G.A., Dalinger I.L., Konstantinova L.S., Kuznetsov V.V., Kravchenko A.N., Krayushkin M.M., Rakitin O.A., Starosotnikov A.M., Fershtat L.L., Shevelev S.A., Shirinian V.Z., Yarovenko V.N.
Russian Chemical Reviews,
2020
12.

Jossang A., Jossang P., Hadi H.A., Sevenet T., Bodo B.
Journal of Organic Chemistry,
1991
13.

Anderton N., Cockrum P.A., Colegate S.M., Edgar J.A., Flower K., Vit I., Willing R.I.
Phytochemistry,
1998
14.

Stuppner H., Sturm S., Konwalinka G.
Chromatographia,
1992
15.

Galliford C., Scheidt K.
Angewandte Chemie - International Edition,
2007
16.

Yu B., Yu D., Liu H.
European Journal of Medicinal Chemistry,
2015
17.
10.1016/j.mencom.2022.09.037_b0085
Zhao
BioDiscovery,
2013
18.

Bora D., Kaushal A., Shankaraiah N.
European Journal of Medicinal Chemistry,
2021
19.

Gazieva G.A., Izmest’ev A.N., Anikina L.V., Pukhov S.A., Meshchaneva M.E., Khakimov D.V., Kolotyrkina N.G., Kravchenko A.N.
Molecular Diversity,
2018
20.
![Synthesis and biological activity of oxindolylidene derivatives of imidazo[4,5-e]thiazolo[3,2-b]-1,2,4-triazin-7-ones and imidazo[4,5-e]thiazolo[2,3-N]-1,2,4-triazin-8-ones](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Izmest’ev A.N., Gazieva G.A., Kulikov A.S., Anikina L.V., Kolotyrkina N.G., Kravchenko A.N.
Russian Journal of Organic Chemistry,
2017
21.

Izmest'ev A.N., Gazieva G.A., Anikina L.V., Pukhov S.A., Karnoukhova V.A., Kolotyrkina N.G., Kravchenko A.N.
New Journal of Chemistry,
2021
22.

Izmest'ev A.N., Gazieva G.A., Karnoukhova V.A., Kravchenko A.N.
Organic and Biomolecular Chemistry,
2020
23.

Izmest'ev A.N., Kim N.A., Karnoukhova V.A., Kolotyrkina N.G., Kravchenko A.N., Gazieva G.A.
ChemistrySelect,
2019
24.
![Diastereoselective synthesis of (Z)-6-(2-oxo-1,2-dihydro-3H-indol-3- ylidene)-3,3a,9,9a-tetrahydroimidazo[4,5-e]thiazolo[3,2-b]-1,2,4-triazin-2,7(1H, 6H)-diones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gazieva G.A., Shishkova E.A., Kulikova L.B., Kolotyrkina N.G., Sigay N.V., Kravchenko A.N.
Journal of Heterocyclic Chemistry,
2013
25.

Evdokimov N.M., Magedov I.V., McBrayer D., Kornienko A.
Bioorganic and Medicinal Chemistry Letters,
2016
26.

George G., Auti P.S., Paul A.T.
New Journal of Chemistry,
2021
27.
![Structure of reaction products of substituted [1,3]thiazolo[3,2-b][1,2,4]triazol-6(5H)-ones with amines and hydrazines](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Rzhevskii A.A., Gerasimova N.P., Chirkova Z.V., Filimonov S.I., Firgang S.I., Stashina G.A., Suponitsky K.Y.
Russian Journal of Organic Chemistry,
2013
28.
Egyptian Journal of Chemistry,
2016