Keywords
Bradford reagent
isopropanol
Nafion membrane
PDDA
proton conductivity
vanadyl permeability
Abstract
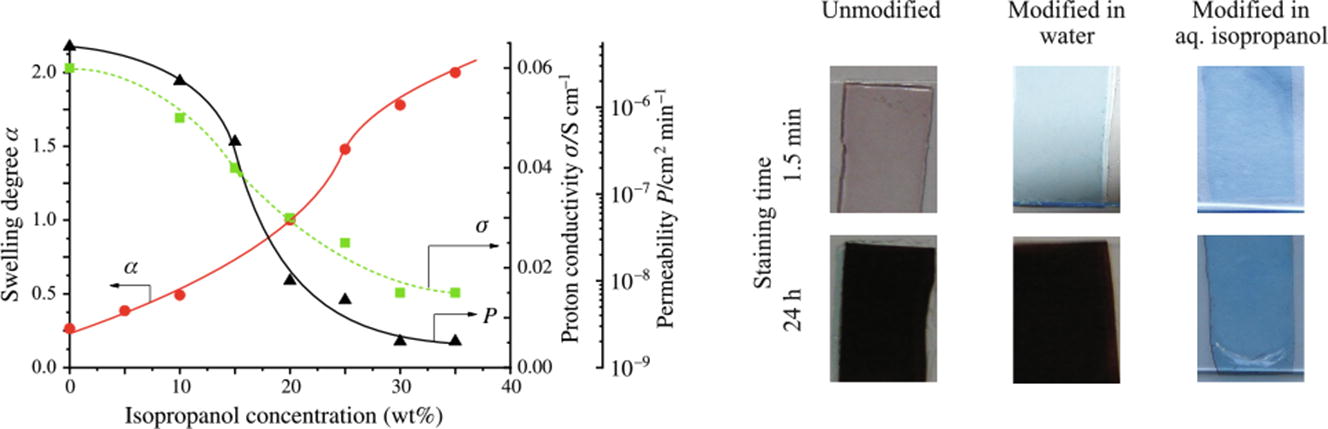
The modification of Nafion® membranes with poly(diallyldimethylammonium chloride) (PDDA) in aqueous solutions of isopropanol has been investigated. A remarkable decrease in vanadyl ion permeability (by 3.5 orders of magnitude), accompanied by a decrease in proton conductivity sixfold at most, was found for Nafion membranes modified in 30–35 wt% isopropanol solutions providing a high swelling of the membranes. The Bradford reagent has been used for the detection of PDDA on the membrane surface.
References
1.

Parasuraman A., Lim T.M., Menictas C., Skyllas-Kazacos M.
Electrochimica Acta,
2013
2.

Shi Y., Eze C., Xiong B., He W., Zhang H., Lim T.M., Ukil A., Zhao J.
Applied Energy,
2019
3.

4.

Gierke T.D., Munn G.E., Wilson F.C.
Journal of Polymer Science Part C: Polymer Letters,
1981
5.

Rubatat L., Gebel G., Diat O.
Macromolecules,
2004
6.

Young S.K., Trevino S.F., Beck Tan N.C.
Journal of Polymer Science, Part B: Polymer Physics,
2002
7.
10.1016/j.mencom.2021.11.023_b0035
Jeon
Energy Mater.,
2020
8.

Zakharova J.A., Novoskoltseva O.A., Pyshkina O.A., Karpushkin E.A., Sergeyev V.G.
Colloid and Polymer Science,
2018
9.

Wu J., Dai Q., Zhang H., Li X.
ChemSusChem,
2020
10.

Tempelman C.H., Jacobs J.F., Balzer R.M., Degirmenci V.
Journal of Energy Storage,
2020
11.

Machado C.A., Brown G.O., Yang R., Hopkins T.E., Pribyl J.G., Epps T.H.
ACS Energy Letters,
2020
12.

Apel P.Y., Bobreshova O.V., Volkov A.V., Volkov V.V., Nikonenko V.V., Stenina I.A., Filippov A.N., Yampolskii Y.P., Yaroslavtsev A.B.
Membranes and Membrane Technologies,
2019
13.

Xi J., Wu Z., Teng X., Zhao Y., Chen L., Qiu X.
Journal of Materials Chemistry A,
2008
14.

Fortier-McGill B., Reven L.
Macromolecules,
2008
15.

Pyshkina О.А., Novoskoltseva О.А., Zakharova J.A.
Colloid and Polymer Science,
2018
16.

Kujawski W., Poźniak G.
Separation Science and Technology,
2005
17.
10.1016/j.mencom.2021.11.023_b0085
Zawodzinski
J. Electrochem. Soc.,
1981
18.

Winardi S., Raghu S.C., Oo M.O., Yan Q., Wai N., Lim T.M., Skyllas-Kazacos M.
Journal of Membrane Science,
2014
19.

Bradford M.M.
Analytical Biochemistry,
1976
20.

Kusoglu A., Weber A.Z.
Chemical Reviews,
2017
21.

Compton S.J., Jones C.G.
Analytical Biochemistry,
1985
22.

Pontié M., Ben Rejeb S., Legrand J.
Separation and Purification Technology,
2012