Keywords
[1,3]dioxolo[4,5-d]imidazoles
aminals
benzil
imidazolidines
thioureas
ureas
α-diketones
Abstract
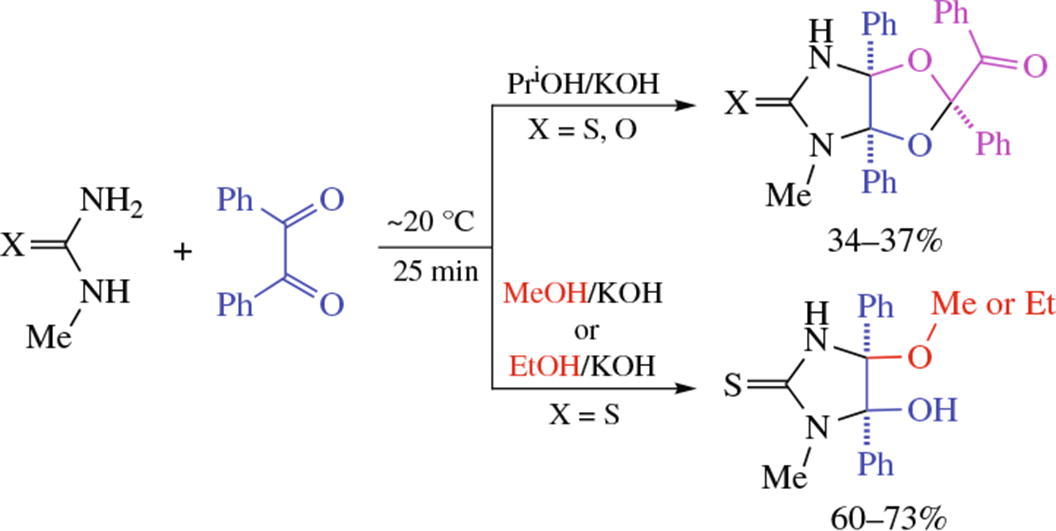
New pathways of reaction between 1-methylthiourea or 1-methylurea and benzil bring about new derivatives of (2S*,3aR*,6aS*)-perhydro-3aH-[1,3]dioxolo[4,5-d]imidazole and racemic (4S*,5R*)-4-alkoxy-5-hydroxy-1-methyl-4,5-diphenylimidazolidine-2-thiones. Some of the obtained urea-and thiourea derivatives were characterized by X-ray diffraction, which showed their supramolecular organization governed by the directionality of hydrogen bonds at the acceptor side C=O or C=S groups.
References
1.

Makhova N.N., Belen’kii L.I., Gazieva G.A., Dalinger I.L., Konstantinova L.S., Kuznetsov V.V., Kravchenko A.N., Krayushkin M.M., Rakitin O.A., Starosotnikov A.M., Fershtat L.L., Shevelev S.A., Shirinian V.Z., Yarovenko V.N.
Russian Chemical Reviews,
2020
2.

Baranov V.V., Galochkin A.A., Nelyubina Y.V., Kravchenko A.N., Makhova N.N.
Synthesis,
2020
3.

Kravchenko A.N., Baranov V.V., Gazieva G.A.
Russian Chemical Reviews,
2018
4.
(a) V. V. Baranov, M. M. Antonova, E. K. Melnikova and A. N. Kravchenko, Russ. Chem. Bull., Int. Ed., 2020, 69, 401 (Izv. Akad. Nauk, Ser. Khim., 2020, 401)
5.

Karnoukhova V.A., Baranov V.V., Vologzhanina A.V., Kravchenko A.N., Fedyanin I.V.
CrystEngComm,
2021
6.

Anikina L.V., Gazieva G.A., Kravchenko A.N.
Russian Chemical Bulletin,
2020
7.

Muccioli G.G., Fazio N., Scriba G.K., Poppitz W., Cannata F., Poupaert J.H., Wouters J., Lambert D.M.
Journal of Medicinal Chemistry,
2005
8.

Neville R.
Journal of Organic Chemistry,
1958
9.

Lee C.K., Kim S.H., Kim Y.B.
Journal of Heterocyclic Chemistry,
1996
10.

Butler A.R., Leitch E.
Journal of the Chemical Society Perkin Transactions 2,
1980
11.

Broan C.J., Butler A.R., Reed D., Sadler I.H.
Journal of the Chemical Society Perkin Transactions 2,
1989
12.
10.1016/j.mencom.2021.09.027_b0055
Broan
J. Chem. Soc., Perkin Trans.,
1501
13.
Alexeev A.A., Nurieva E.V., Trofimova T.P., Chesnakova E.A., Grishin Y.K., Lyssenko K.A., Filimonova M.V., Zefirova O.N.
Mendeleev Communications,
2019
14.
Kalistratova A.V., Kovalenko L.V., Oshchepkov M.S., Gamisoniya A.M., Gerasimova T.S., Demidov Y.A., Akimov M.G.
Mendeleev Communications,
2020
15.
Baranov V.V., Ivanova A.B., Kolotyrkina N.G., Kravchenko A.N.
Mendeleev Communications,
2019
16.

Baranov V., Antonova M., Kolotyrkina N., Zanin I., Kravchenko A.
Synlett,
2018
17.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
18.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
19.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
20.

Muccioli G.G., Poupaert J.H., Wouters J., Norberg B., Poppitz W., Scriba G.K., Lambert D.M.
Tetrahedron,
2003
21.

Muccioli G.G., Martin D., Scriba G.K., Poppitz W., Poupaert J.H., Wouters J., Lambert D.M.
Journal of Medicinal Chemistry,
2005
22.

Trišović N., Valentić N., Ušćumlić G.
Chemistry Central Journal,
2011
23.

Nelyubina Y.V., Gazieva G.A., Baranov V.V., Belyakov P.A., Chizhov A.O., Lyssenko K.A., Kravchenko A.N.
Russian Chemical Bulletin,
2009
24.

Allen F.H., Bird C.M., Rowland R.S., Raithby P.R.
Acta Crystallographica Section B Structural Science,
1997
25.

Guihéneuf S., Paquin L., Carreaux F., Durieu E., Meijer L., Bazureau J.P.
Organic and Biomolecular Chemistry,
2012
26.
T. L. Boehm, L. C. Christie, B. Devadas, H. M. Madsen, L. Marrufo and S. Selness, Patent WO 2006/040666, 2006.
27.
Kravchenko A.N., Chikunov I.E., Gazieva G.A., Makhova N.N.
Mendeleev Communications,
2016
28.

Raux B., Voitovich Y., Derviaux C., Lugari A., Rebuffet E., Milhas S., Priet S., Roux T., Trinquet E., Guillemot J., Knapp S., Brunel J., Fedorov A.Y., Collette Y., Roche P., et. al.
Journal of Medicinal Chemistry,
2016