Keywords
1,2,5,6-tetrahydropyridine-2,6-dione
2H-pyrano[2,3-b]pyridinones
Erlenmeyer–Plöchl reaction
hippuric acid
ring switching
tricomponent condensation
triethyl orthoformate
Abstract
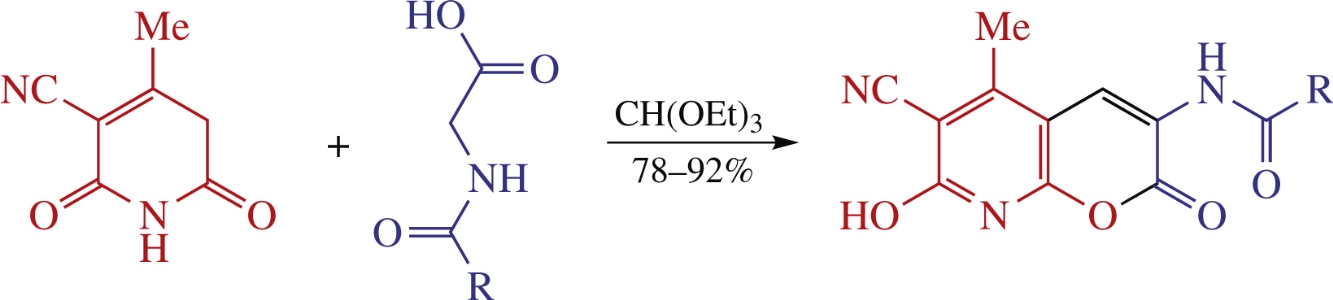
New 3-acylamino-7-hydroxy-5-methyl-2-oxo-2H-pyrano[2,3-b]pyridine-6-carbonitriles were synthesized via a tricomponent condensation of 3-cyano-4-methyl-1,2,5,6-tetrahydropyridine-2,6-dione, triethyl orthoformate and N-acylglycines under the Erlenmeyer–Plöchl reaction conditions. According to X-ray data, in the solid phase they exist as hydroxypyridine tautomer form.
References
1.

Takano H., Narumi T., Ohashi N., Suzuki A., Furuta T., Nomura W., Tamamura H.
Tetrahedron,
2014
2.

Takano H., Narumi T., Nomura W., Furuta T., Tamamura H.
Organic Letters,
2015
3.

Takano H., Narumi T., Nomura W., Tamamura H.
Journal of Organic Chemistry,
2017
4.

Stanovnik B., Svete J.
Chemical Reviews,
2004
5.

Svete J., Čadež Z., Stanovnik B., Tišler M.
Synthesis,
1990
6.

Toplak R., Svete J., Stanovnik B., Grdadolnik S.G.
Journal of Heterocyclic Chemistry,
1999
7.

Kralj L., Hvala A., Svete J., Golič L., Stanovnik B.
Journal of Heterocyclic Chemistry,
1997
8.

Kocevar M., Kepe V., Polanc S.
Heterocycles,
1995
9.

Kepe V., Kočevar M., Polanc S.
Journal of Heterocyclic Chemistry,
1996
10.

Kocevar M., Kepe V., Polanc S.
Heterocycles,
1998
11.
10.1016/j.mencom.2021.04.041_bib0055
Požgan
ARKIVOC,
2007
12.
Paponov B.V., Lvov S.V., Ichetovkina E.V., Panasenko I.A., Stepanian S.G.
Mendeleev Communications,
2012
13.
Oshega J.S., Paponov B.V., Omelchenko I.V., Shishkin O.V.
Mendeleev Communications,
2015
14.
10.1016/j.mencom.2021.04.041_bib0070
Erlenmeyer
Liebigs Ann. Chem.,
1893
15.

Dinsmore A., Doyle P.M., Young D.W.
Tetrahedron Letters,
1995
16.

WAMHOFF H., KORTE F.
Synthesis,
1972
17.

ZIMMER H., ROTHE J.
Journal of Organic Chemistry,
1959
18.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
19.
10.1016/j.mencom.2021.04.041_bib0095
Sheldrick
Acta Crystallogr.,
2015
20.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007