Keywords
aldehydes
arenecarboxylic acids
C–O cleavage
dimethyl dicarbonate
hydrosilanes
nickel catalysis
Abstract
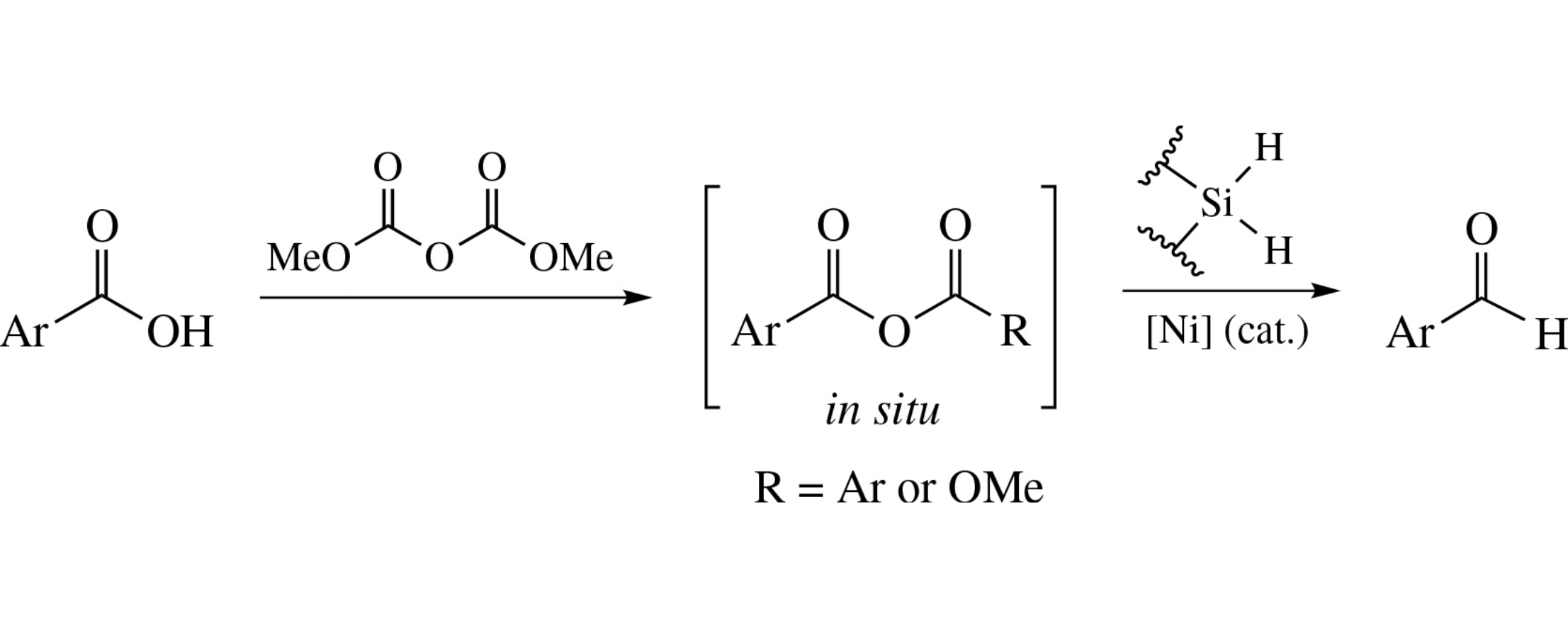
Nickel-catalyzed reduction of (het)arenecarboxylic acids with hydrosilanes in the presence of dimethyl dicarbonate as the activator affords the corresponding aldehydes. The role of the activator is the conversion of the acids into their anhydrides undergoing C–O cleavage. The good yields were achieved in case of substrates bearing electron-donating and electron-neutral groups.
References
1.
10.1016/j.mencom.2021.03.043_bib0005
Johnstone
1991
2.

Brown H.C., Cha J.S., Nazer B., Yoon N.M.
Journal of the American Chemical Society,
1984
3.

Miyamoto K., Motoyama Y., Nagashima H.
Chemistry Letters,
2012
4.

Misal Castro L.C., Li H., Sortais J., Darcel C.
Chemical Communications,
2012
5.

Zheng J., Chevance S., Darcel C., Sortais J.
Chemical Communications,
2013
6.

Bézier D., Park S., Brookhart M.
Organic Letters,
2013
7.

Zhang M., Li N., Tao X., Ruzi R., Yu S., Zhu C.
Chemical Communications,
2017
8.

Stache E.E., Ertel A.B., Rovis T., Doyle A.G.
ACS Catalysis,
2018
9.

Iosub A.V., Wallentin C., Bergman J.
Nature Catalysis,
2018
10.

Nagayama K., Shimizu I., Yamamoto A.
Chemistry Letters,
1998
11.

Direct Hydrogenation of Carboxylic Acids to Corresponding Aldehydes Catalyzed by Palladium Complexes
Nagayama K., Shimizu I., Yamamoto A.
Bulletin of the Chemical Society of Japan,
2001
12.

Gooßen L.J., Ghosh K.
Chemical Communications,
2002
13.

Fujihara T., Cong C., Terao J., Tsuji Y.
Advanced Synthesis and Catalysis,
2013
14.

Bercot E.A., Rovis T.
Journal of the American Chemical Society,
2001
15.

O'Brien E.M., Bercot E.A., Rovis T.
Journal of the American Chemical Society,
2003
16.

Kajita Y., Kurahashi T., Matsubara S.
Journal of the American Chemical Society,
2008
17.

Ochi Y., Kurahashi T., Matsubara S.
Organic Letters,
2011
18.

Zhao C., Jia X., Wang X., Gong H.
Journal of the American Chemical Society,
2014
19.

Lin T., Mi J., Song L., Gan J., Luo P., Mao J., Walsh P.J.
Organic Letters,
2018
20.

He J., Song P., Xu X., Zhu S., Wang Y.
ACS Catalysis,
2019
21.

Le C.“., MacMillan D.W.
Journal of the American Chemical Society,
2015
22.

Joe C.L., Doyle A.G.
Angewandte Chemie - International Edition,
2016
23.

Stache E.E., Rovis T., Doyle A.G.
Angewandte Chemie - International Edition,
2017
24.

Badir S.O., Dumoulin A., Matsui J.K., Molander G.A.
Angewandte Chemie - International Edition,
2018
25.

Iosub A.V., Moravčík Š., Wallentin C., Bergman J.
Organic Letters,
2019
26.

Nickel-Catalyzed Barton Decarboxylation and Giese Reactions: A Practical Take on Classic Transforms.
Qin T., Malins L.R., Edwards J.T., Merchant R.R., Novak A.J., Zhong J.Z., Mills R.B., Yan M., Yuan C., Eastgate M.D., Baran P.S.
Angewandte Chemie - International Edition,
2016
27.

Simmons B.J., Hoffmann M., Hwang J., Jackl M.K., Garg N.K.
Organic Letters,
2017
28.

Johnson J.B., Bercot E.A., Rowley J.M., Coates G.W., Rovis T.
Journal of the American Chemical Society,
2007
29.

Revunova K., Nikonov G.I.
Chemistry - A European Journal,
2013
30.

Guo L., Srimontree W., Zhu C., Maity B., Liu X., Cavallo L., Rueping M.
Nature Communications,
2019