Keywords
benzothiadiazoles
cross-coupling
luminophores
photophysical properties
synthesis
Abstract
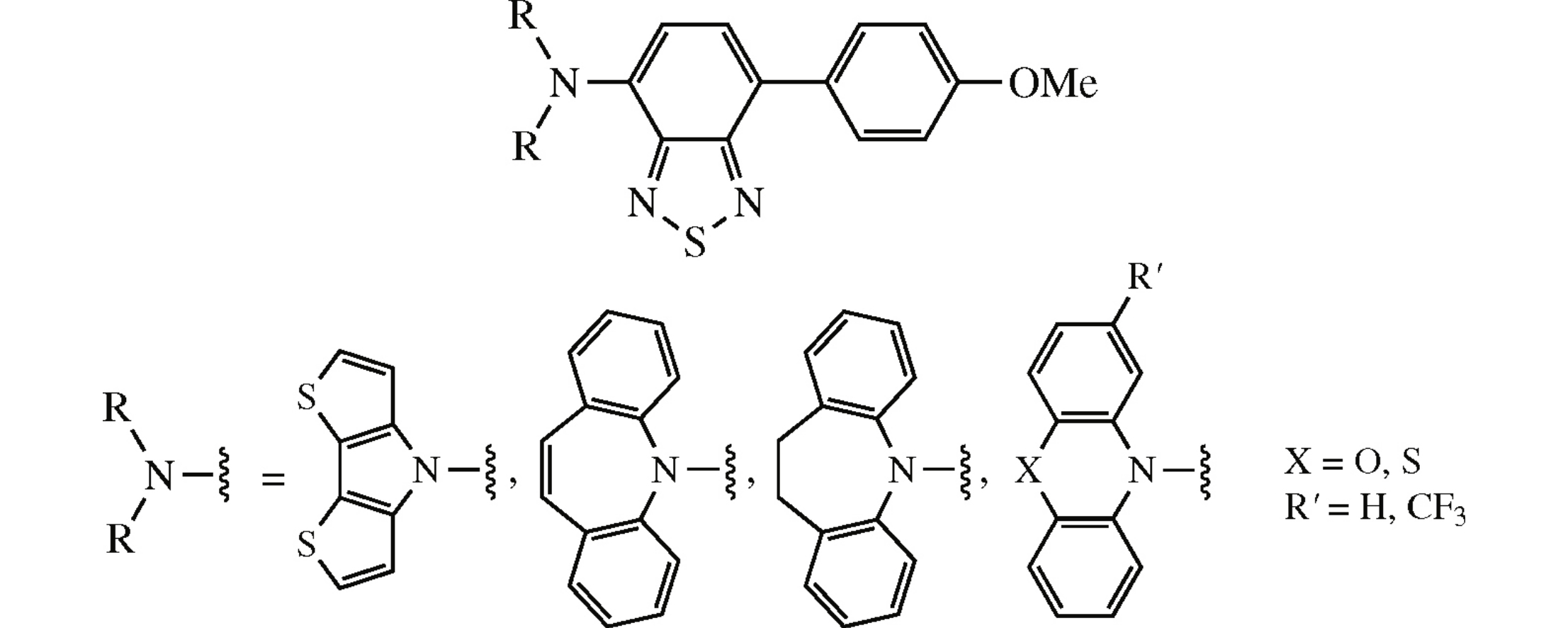
New unsymmetrically substituted benzothiadiazoles were synthesized from 4,7-dibromo-2,1,3-benzothiadiazole via the sequence of Pd-catalyzed Suzuki and Buchwald–Hartwig cross-coupling reactions with 4-methoxyphenylboronic acid and heterocyclic amines, respectively. Based on initially performed photophysical study as well as DFT calculation, these compounds, in particular with dibenzoazepine core, can be selected as promising scaffolds for further fine-tuning of their properties to be used in optoelectronics including OLED technologies.
References
1.

Sukhikh T.S., Ogienko D.S., Bashirov D.A., Konchenkoa S.N.
Russian Chemical Bulletin,
2019
2.

Müller C.D., Falcou A., Reckefuss N., Rojahn M., Wiederhirn V., Rudati P., Frohne H., Nuyken O., Becker H., Meerholz K.
Nature,
2003
3.

Tsao H.N., Cho D.M., Park I., Hansen M.R., Mavrinskiy A., Yoon D.Y., Graf R., Pisula W., Spiess H.W., Müllen K.
Journal of the American Chemical Society,
2011
4.

Zhang M., Tsao H.N., Pisula W., Yang C., Mishra A.K., Müllen K.
Journal of the American Chemical Society,
2007
5.

Sonar P., Singh S.P., Li Y., Soh M.S., Dodabalapur A.
Advanced Materials,
2010
6.

Li Y.
Accounts of Chemical Research,
2012
7.

Du J., Biewer M.C., Stefan M.C.
Journal of Materials Chemistry A,
2016
8.

Liu Q., Zhan H., Ho C., Dai F., Fu Y., Xie Z., Wang L., Li J., Yan F., Huang S., Wong W.
Chemistry - An Asian Journal,
2013
9.

Neto B.A., Carvalho P.H., Correa J.R.
Accounts of Chemical Research,
2015
10.

Carvalho P.H., Correa J.R., Guido B.C., Gatto C.C., De Oliveira H.C., Soares T.A., Neto B.A.
Chemistry - A European Journal,
2014
11.

Wu J., Lai G., Li Z., Lu Y., Leng T., Shen Y., Wang C.
Dyes and Pigments,
2016
12.

Kimura M., Karasawa M., Sasagawa N., Takemoto K., Goto R., Mori S.
Chemistry Letters,
2012
13.

Ni F., Wu Z., Zhu Z., Chen T., Wu K., Zhong C., An K., Wei D., Ma D., Yang C.
Journal of Materials Chemistry C,
2017
14.

Peng Z., Wang Z., Huang Z., Liu S., Lu P., Wang Y.
Journal of Materials Chemistry C,
2018
15.

Vasil’ev A.A., Struchkova M.I., Sheremetev A.B., Levinson F.S., Varganov R.V., Lyssenko K.A.
Russian Chemical Bulletin,
2011
16.

Flourescence self-quenching of the molecular forms of Rhodamine B in aqueous and ethanolic solutions
Arbeloa F.L., Ojeda P.R., Arbeloa I.L.
Journal of Luminescence,
1989
17.

Karstens T., Kobs K.
The Journal of Physical Chemistry,
1980
18.

Molotkov A.P., Arsenov M.A., Kapustin D.A., Muratov D.V., Shepel' N.E., Fedorov Y.V., Smol'yakov A.F., Knyazeva E.I., Lypenko D.A., Dmitriev A.V., Aleksandrov A.E., Maltsev E.I., Loginov D.A.
ChemPlusChem,
2020
19.

Kharitonov V.B., Nelyubina Y.V., Muratov D.V., Shepel' N.E., Loginov D.A.
Journal of Organometallic Chemistry,
2020