Keywords
chromenes.
chromeno[3′,4′:5,6]pyrano[2,3-c]pyridines
electrophilic substitution
furo[3,4-c]pyridines
heterocyclization
pyridoxal
Abstract
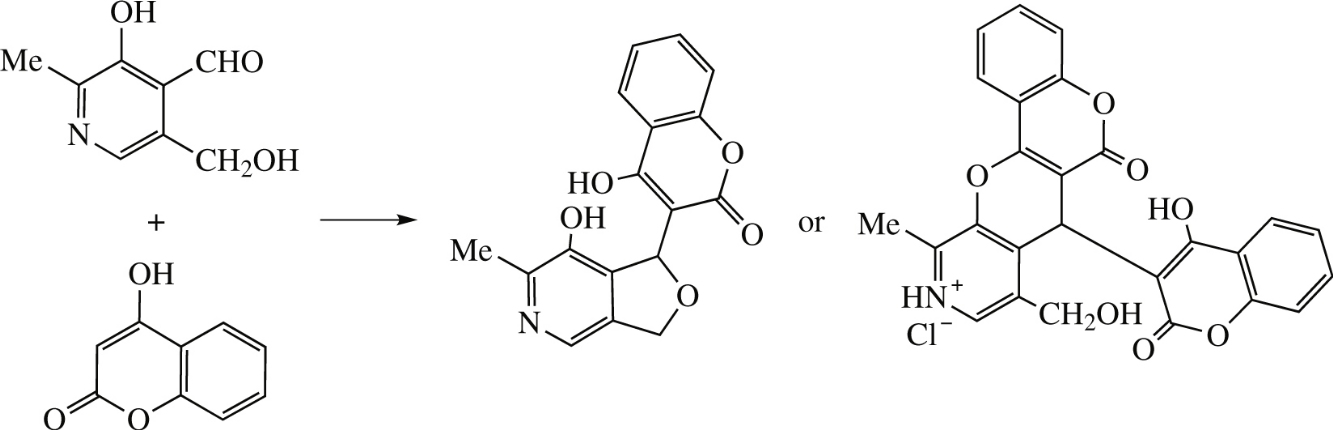
Pyridoxal in the absence of catalyst forms furo[3,4-c]pyridine species (ortho aldehyde and hydroxymethyl groups are involved) which is further transformed into C–C hybrid with 4-hydroxycoumarin. Different products of chromeno-[3′,4′:5,6]pyrano[2,3-c]pyridine type are formed when pyridoxal hydrochloride or more sophisticated 2,4-dihydroxybenzaldehyde derivative are applied (herein, ortho aldehyde and hydroxy groups are involved into heterocyclization).
References
1.

Müller-Schiffmann A., Sticht H., Korth C.
BioDrugs,
2012
2.

Cerecetto H., González M.
Topics in Heterocyclic Chemistry,
2007
3.

Chugunova E., Burilov A.
Current Topics in Medicinal Chemistry,
2016
4.

Kibardina L.K., Trifonov A.V., Burilov A.R., Gazizov A.S., Pudovik M.A.
Russian Journal of General Chemistry,
2018
5.
Kibardina L.K., Trifonov A.V., Dobrynin A.B., Pudovik M.A., Burilov A.R.
Mendeleev Communications,
2018
6.

Kibardina L.K., Trifonov A.V., Burilov A.R., Pudovik M.A.
Russian Journal of General Chemistry,
2018
7.

Manolov I., Raleva S., Genova P., Savov A., Froloshka L., Dundarova D., Argirova R.
Bioinorganic Chemistry and Applications,
2006
8.

Manolov I., Maichle-Moessmer C., Nicolova I., Danchev N.
Archiv der Pharmazie,
2006
9.

Khan K.M., Iqbal S., Lodhi M.A., Maharvi G.M., Ullah Z., Choudhary M.I., Rahman A., Perveen S.
Bioorganic and Medicinal Chemistry,
2004
10.

Lehmann J.
The Lancet,
1943
11.

Stern P., Deželić M., Košak R.
Naunyn-Schmiedeberg's Archives of Pharmacology,
1957
12.

Mueller R.L.
Best Practice and Research in Clinical Haematology,
2004
13.

Smolobochkin A.V., Gazizov A.S., Burilov A.R., Pudovik M.A., Sinyashin O.G.
Russian Chemical Reviews,
2019
14.

Rezaei R., Moezzi F., Doroodmand M.M.
Chinese Chemical Letters,
2014
15.

Zolfigol M.A., Moosavi-Zare A.R., Zarei M.
Comptes Rendus Chimie,
2014
16.

Zahiri S., Mokhtary M.
Journal of Taibah University for Science,
2015
17.

Zhu A., Bai S., Li L., Wang M., Wang J.
Catalysis Letters,
2015
18.

Chang X., Zhang X., Chen Z.
Organic and Biomolecular Chemistry,
2018
19.
KHODABAKHSHI S., KARAMI B., ESKANDARI K., RASHIDI A.
South African Journal of Chemistry,
2015
20.

Han H., Zhang Z., Zhang J., Zhang B.
Main Group Chemistry,
2019
21.

Khan K.M., Iqbal S., Lodhi M.A., Maharvi G.M., Perveen S., Choudhary M.I., Atta-ur-Rahman, Chohan Z.H., Supuran C.T.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2004
22.

Synthesis of Biscoumarin and Dihydropyran Derivatives and Evaluation of Their Antibacterial Activity
Li J., Lv C., Li X., Qu D., Hou Z., Jia M., Luo X., Li X., Li M.
Molecules,
2015
23.
APEX2 (Version 2.1), SAINTPlus, Data Reduction and Correction Program (Version 7.31A), Bruker Advanced X-ray Solutions, Bruker AXS, Madison, Wisconsin, USA, 2006.
24.
G. M. Sheldrick, SADABS, Program for Empirical X-ray Absorption Correction, Bruker-Nonius, 1990-2004.
25.
10.1016/j.mencom.2020.11.025_bib0125
Sheldrick
Acta Crystallogr.,
2015
26.

Farrugia L.J.
Journal of Applied Crystallography,
1999
27.
10.1016/j.mencom.2020.11.025_bib0135
Spek
Acta Crystallogr.,
2009
28.

Macrae C.F., Bruno I.J., Chisholm J.A., Edgington P.R., McCabe P., Pidcock E., Rodriguez-Monge L., Taylor R., van de Streek J., Wood P.A.
Journal of Applied Crystallography,
2008