Keywords
alkynes
bromoamines
bromohexamethyldisilazane
hyperconjugation
isomerism
orbital interactions
organosilicon compounds
polarizability
stretching vibration
Abstract
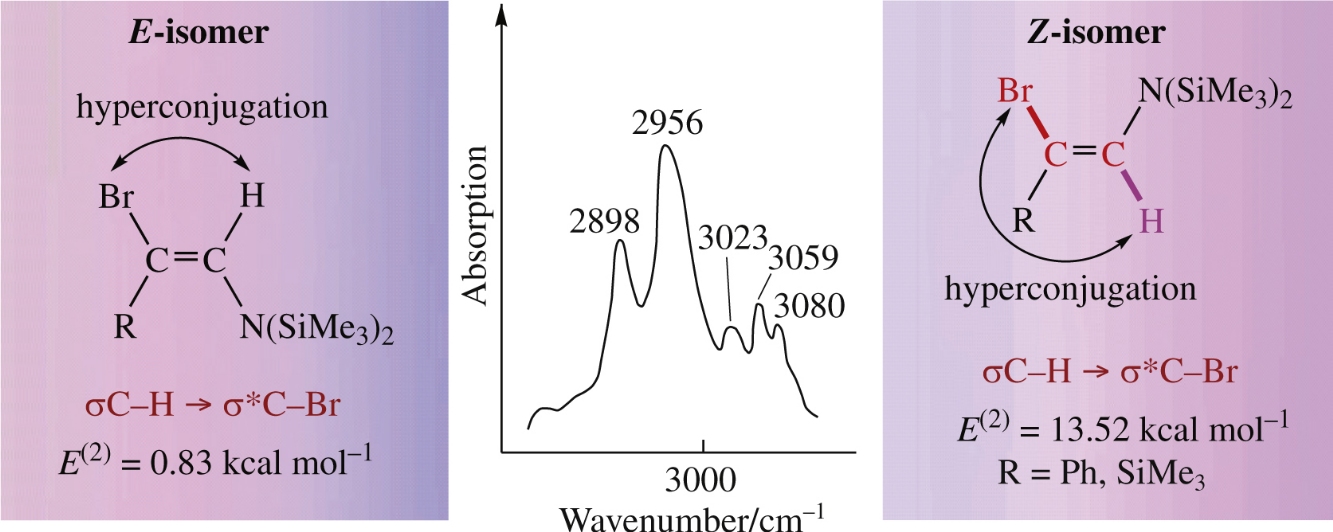
The conformational isomerism and orbital interactions of N-(2-bromo-2-phenylethenyl)-N,N-bis(trimethylsilyl)amine and N-(2-bromo-2-trimethylsilylethenyl)-N,N-bis(trimethylsilyl) amine were examined using NMR technique and quantum-chemical calculations. The IR criterion characterizing σ→σ* hyperconjugation is considered, and the linear dependence of σC–H→σ*C–X (X = F, Cl, Br) interaction energy on the polarizability of X is discussed.
References
1.
10.1016/j.mencom.2020.11.018_bib0005
Alabugin
Wiley Interdiscip. Rev.: Comput. Mol. Sci.,
2011
2.

Pophristic V., Goodman L.
Nature,
2001
3.

Mo Y., Wu W., Song L., Lin M., Zhang Q., Gao J.
Angewandte Chemie - International Edition,
2004
4.

Chen Z., Corminboeuf C., Mo Y.
Journal of Physical Chemistry A,
2013
5.

Pross A., Radom L., Riggs N.V.
Journal of the American Chemical Society,
1980
6.

Dorigo A.E., Pratt D.W., Houk K.N.
Journal of the American Chemical Society,
1987
7.

Lin B., Xie Z., Liu R., Liu L., Guo Q.
Journal of Molecular Structure THEOCHEM,
2003
8.

Rakhlin V.I., Mirskov R.G., Podgorbunskaya T.A., Voronkov M.G.
Russian Journal of General Chemistry,
2007
9.

Bailey R.E., West R.
Journal of Organometallic Chemistry,
1965
10.

Brand J.C., Cook M.D., Price A.J., Roberts B.P.
Journal of the Chemical Society Chemical Communications,
1982
11.

Cook M.D., Roberts B.P., Singh K.
Journal of the Chemical Society Perkin Transactions 2,
1983
12.

Roberts B.P., Wilson C.
Journal of the Chemical Society Chemical Communications,
1978
13.
Rusakova I.L., Krivdin L.B.
Mendeleev Communications,
2018
14.

Krivdin L.B.
Russian Chemical Reviews,
2020
15.
10.1016/j.mencom.2020.11.018_bib0075
Frisch
Gaussian 09, Revision C.01,
2010
16.

Reed A.E., Weinhold F.
Israel Journal of Chemistry,
1991
17.

Boganov S.E., Promyslov V.M., Rynin S.S., Krylova I.V., Zaitseva G.S., Egorov M.P.
Russian Chemical Bulletin,
2018
18.

Alabugin I.V., Zeidan T.A.
Journal of the American Chemical Society,
2002
19.

Nagle J.K.
Journal of the American Chemical Society,
1990
20.

Rakhlin V.I., Grigor'ev S.V., Mirskov R.G., Podgorbunskaya T.A., Voronkov M.G., Gendin D.V.
Russian Journal of General Chemistry,
2003