Keywords
1,2,4-triazines
2-(indol-3-yl)pyridines
aza-Diels–Alder reaction
nucleophilic substitution of hydrogen
pyridines
Abstract
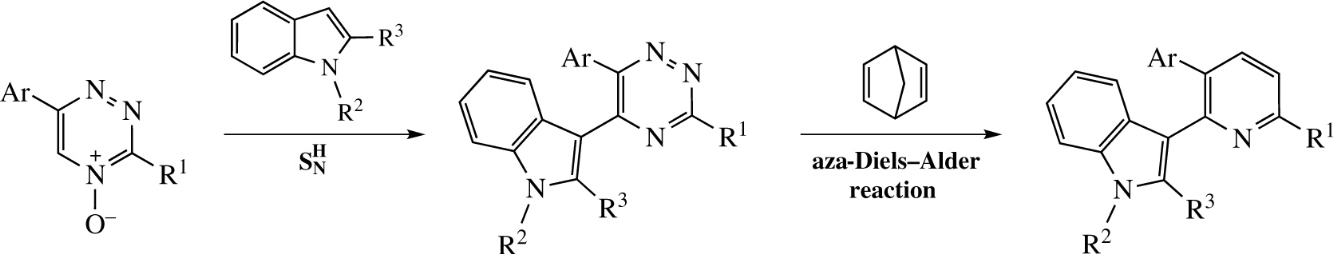
New access to substituted 2-(indol-3-yl)pyridines involves two stage protocol, namely, a reaction of deoxygenative nucleophilic substitution of hydrogen in 1,2,4-triazine-4-oxides under the action of indoles followed by aza-Diels–Alder reaction of thus obtained 5-indolyl-1,2,4-triazines with 2,5-norbornadiene in a pressure vessel. The reactions sequence provides good yields and is suitable for wide scope of substituted 1,2,4-triazines.
References
1.

Indole in the target-based design of anticancer agents: A versatile scaffold with diverse mechanisms
Dadashpour S., Emami S.
European Journal of Medicinal Chemistry,
2018
2.

Ölgen S., Altanlar N., Karataylı E., Bozdayı M.
Zeitschrift fur Naturforschung - Section C Journal of Biosciences,
2008
3.

Hong W., Li J., Chang Z., Tan X., Yang H., Ouyang Y., Yang Y., Kaur S., Paterson I.C., Ngeow Y.F., Wang H.
Journal of Antibiotics,
2017
4.

Kaushik N., Kaushik N., Attri P., Kumar N., Kim C., Verma A., Choi E.
Molecules,
2013
5.
10.1016/j.mencom.2020.11.007_bib0020
Altaf
J. Drug Design Med. Chem.,
2015
6.

Thirumurugan P., Mahalaxmi S., Perumal P.T.
Journal of Chemical Sciences,
2010
7.

Elshemy H.A., Zaki M.A., Mohamed E.I., Khan S.I., Lamie P.F.
Bioorganic Chemistry,
2020
8.

Kantevari S., Patpi S.R., Addla D., Putapatri S.R., Sridhar B., Yogeeswari P., Sriram D.
ACS Combinatorial Science,
2011
9.
H.-L. Wang, V.C. Cee, B.J. Herberich, C.L. M. Jackson, B.A. Lanman, T. Nixey, L.H. Pettus, A.B. Reed, B. Wu, R. Wurz and A. Tasker, Patent WO 2012/129338 A1, 2012.
10.
N. Gray, T. Zhang and N. P. Kwiatkowski, Patent WO 2014/63068 A1, 2014.
11.
C. Tang, Q. Ren, J. Yin, K. Yi and Y. Zhang, Patent WO 2018/41091 A1, 2018.
12.

Gong X., Song G., Zhang H., Li X.
Organic Letters,
2011
13.

Muthusubramanian S., Suresh R., Senthilkumaran R.
Synlett,
2014
14.

Wu N., Song F., Yan L., Li J., You J.
Chemistry - A European Journal,
2014
15.

Wang Z., Song F., Zhao Y., Huang Y., Yang L., Zhao D., Lan J., You J.
Chemistry - A European Journal,
2012
16.

Rocca P., Marsais F., Godard A., Quéguiner G., Adams L., Alo B.
Tetrahedron Letters,
1995
17.

Yu J., Moon H.R., Kim S.Y., Kim J.N.
Bulletin of the Korean Chemical Society,
2015
18.

Brasse M., Ellman J.A., Bergman R.G.
Chemical Communications,
2011
19.

Shiri M., Zolfigol M.A., Pirveysian M., Ayazi-Nasrabadi R., Kruger H.G., Naicker T., Mohammadpoor-Baltork I.
Tetrahedron,
2012
20.

Gao Q., Wang Y., Wang Q., Zhu Y., Liu Z., Zhang J.
Organic and Biomolecular Chemistry,
2018
21.
G. D. Cuny, J.R. Hauske, M.Z. Hoemann and I. Chopra, Patent US 6376670 B1, 2002.
22.

Hoemann M.Z., Kumaravel G., Xie R.L., Rossi R.F., Meyer S., Sidhu A., Cuny G.D., Hauske J.R.
Bioorganic and Medicinal Chemistry Letters,
2000
23.
A. R. Gangloff, J. Nowakowaski, B.R. Paraselli, J.A. Stafford and M. G. Tennant, Patent WO 2005/61519 A1, 2005.
24.

Ma Y., Zhou Z., Zhao Y., Luo Y., Zhu Y., Wang H., Zhou H., Tan H., Xie W., Tang Z.
Synlett,
2018
25.

Cacchi S., Fabrizi G., Parisi L.
Synthesis,
2004
26.

Pfu¨ller O.C., Sauer J.
Tetrahedron Letters,
1998
27.

Foster R.A., Willis M.C.
Chemical Society Reviews,
2013
28.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
29.

Kozhevnikov V.N., Shabunina O.V., Kopchuk D.S., Ustinova M.M., König B., Kozhevnikov D.N.
Tetrahedron,
2008
30.
10.1016/j.mencom.2020.11.007_sbref0125a
Metal Free C–H Functionalization of Aromatics: Nucleophilic Displacement of Hydrogen,
2014
31.

Chupakhin O.N., Charushin V.N.
Pure and Applied Chemistry,
2017
32.

Charushin V.N., Chupakhin O.N.
Russian Chemical Bulletin,
2019
33.

Kozhevnikov D.N., Kozhevnikov V.N., Prokhorov A.M., Ustinova M.M., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G., König B.
Tetrahedron Letters,
2006
34.

Savchuk M.I., Khasanov A.F., Kopchuk D.S., Krinochkin A.P., Nikonov I.L., Starnovskaya E.S., Shtaitz Y.K., Kovalev I.S., Zyryanov G.V., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2019
35.

Moseev T.D., Varaksin M.V., Gorlov D.A., Nikiforov E.A., Kopchuk D.S., Starnovskaya E.S., Khasanov A.F., Zyryanov G.V., Charushin V.N., Chupakhin O.N.
Journal of Fluorine Chemistry,
2019
36.
Fatykhov R.F., Savchuk M.I., Starnovskaya E.S., Bobkina M.V., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Khalymbadzha I.A., Chupakhin O.N., Charushin V.N., Kartsev V.G.
Mendeleev Communications,
2019
37.

Krinochkin A.P., Kopchuk D.S., Kovalev I.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
ChemistrySelect,
2020
38.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Giri K., Santra S., Kovalev I.S., Nosova E.V., Gundala S., Venkatapuram P., Zyryanov G.V., Majee A., Chupakhin O.N.
Organic and Biomolecular Chemistry,
2018
39.
10.1016/j.mencom.2020.11.007_bib0140
Rusinov
Russ. J. Org. Chem.,
2000
40.
10.1016/j.mencom.2020.11.007_bib0145
Kozhevnikov
Russ. J. Org. Chem.,
1998
41.
Neunhoeffer H., Weischedel F., Böhnisch V.
1971
42.

Prokhorov A.M., Kozhevnikov D.N.
Chemistry of Heterocyclic Compounds,
2012
43.

Savchuk M.I., Starnovskaya E.S., Shtaitz Y.K., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Russian Journal of General Chemistry,
2018