Keywords
cyclic voltammetry
electrochemical reduction
heterocyclization
nitroarenes
piperidines
pyrido[1,2-a]benzimidazoles
pyrido[3′,2′:4,5]imidazo[1,2-a]pyridines
Abstract
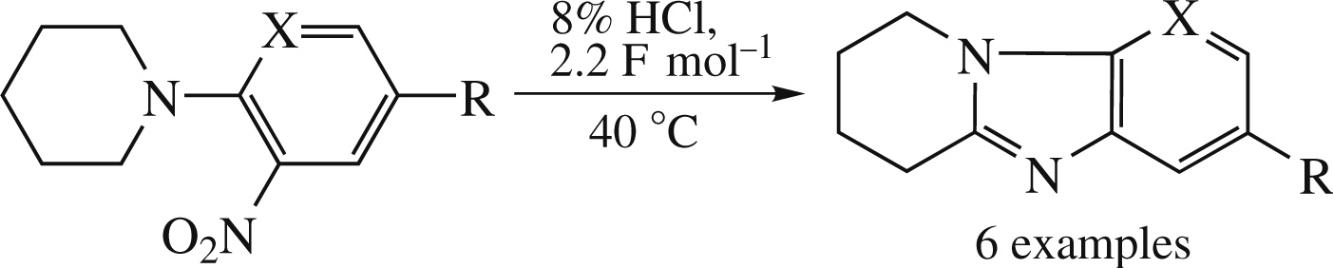
Electrochemical reduction of ortho-piperidino substituted nitro(het)arenes in an undivided cell on a lead cathode in 8% HCl gave either 1,2,3,4-tetrahydropyrido[1,2-a]-benzimidazoles or 6,7,8,9-tetrahydropyrido[3′,2′:4,5]- imidazo[1,2-a]pyridines. The reductive heterocyclization mechanism involves the initial formation of a nitroso derivative followed by the formation of an imidazole ring.
References
1.
R. P. Alexander, M.D. Calmiano, S. Defays, V. Durieu, M. Deligny, J.P. Heer, V.E. Jackson, J. Keyaerts, B. Kroeplien, C.M. Mac, Y.A. Sabnis, M.D. Selby, D.L. L. Swinnen, H.N. Van and Z. Zhu, Patent WO 2015/86525A1, 2015.
2.
H. Uchida, A. Asagarasu and T. Matsui, Patent US 9200008B2, 2015.
3.
A. Kazuyoshi, K. Kobayashi, T. Takai, T. Kojima, K. Tokumaru and M. Mochizuki, Patent US 8901141B2, 2014.
4.
![Synthesis and Toxicity of New Ring-Fused Imidazo[5,4-f]benzimidazolequinones and Mechanism Using Amine N-Oxide Cyclizations](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Fagan V., Bonham S., McArdle P., Carty M.P., Aldabbagh F.
European Journal of Organic Chemistry,
2012
5.
![Synthesis by Radical Cyclization and Cytotoxicity of Highly Potent Bioreductive Alicyclic Ring Fused [1,2-a]Benzimidazolequinones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Lynch M., Hehir S., Kavanagh P., Leech D., O'Shaughnessy J., Carty M., Aldabbagh F.
Chemistry - A European Journal,
2007
6.

Moriarty E., Carr M., Bonham S., Carty M.P., Aldabbagh F.
European Journal of Medicinal Chemistry,
2010
7.

Fagan V., Bonham S., Carty M.P., Aldabbagh F.
Organic and Biomolecular Chemistry,
2010
8.
![COMPARE analysis of the toxicity of an iminoquinone derivative of the imidazo[5,4-f]benzimidazoles with NAD(P)H:quinone oxidoreductase 1 (NQO1) activity and computational docking of quinones as NQO1 substrates](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fagan V., Bonham S., Carty M.P., Saenz-Méndez P., Eriksson L.A., Aldabbagh F.
Bioorganic and Medicinal Chemistry,
2012
9.

Gurry M., Aldabbagh F.
Organic and Biomolecular Chemistry,
2016
10.

Liubchak K., Nazarenko K., Tolmachev A.
Tetrahedron,
2012
11.
![Metal-free construction of tricyclic or tetracyclic compounds—acid-promoted synthesis of benzo[4,5]imidazo[2,1-a]isoindole and 1,2-dialkyl-2,3-dihydrobenzimidazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chen J., Qu J., Zhang Y., Chen Y., Liu N., Chen B.
Tetrahedron,
2013
12.

Selivanova G.A., Tretyakov E.V.
Russian Chemical Bulletin,
2020
13.
![Furan ring transformation as key stage in the synthesis of 5H,12H-benzo[4,5]imidazo[1,2-a]pyrrolo[1,2-d]pyrazines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Stroganova T.A., Vasilin V.K., Krapivin G.D.
Chemistry of Heterocyclic Compounds,
2018
14.

Xue D., Long Y.
Journal of Organic Chemistry,
2014
15.

Gurry M., Sweeney M., McArdle P., Aldabbagh F.
Organic Letters,
2015
16.

Sweeney M., Gurry M., Keane L.J., Aldabbagh F.
Tetrahedron Letters,
2017
17.

Sweeney M., Keane L.J., Gurry M., McArdle P., Aldabbagh F.
Organic Letters,
2018
18.

Suschitzky H., Sutton M.E.
Tetrahedron,
1968
19.

Shawcross A.P., Stanforth S.P.
Journal of Heterocyclic Chemistry,
1990
20.

Hubbard J.W., Piegols A.M., Söderberg B.C.
Tetrahedron,
2007
21.

Das S., Joardar S., Bhattacharyya A.
Synthesis,
2014
22.

Nguyen T.B., Ermolenko L., Al-Mourabit A.
Green Chemistry,
2016
23.

Suschitzky H., Sutton M.E.
Tetrahedron Letters,
1967
24.

Fielden R., Meth-Cohn O., Suschitzky H.
Tetrahedron Letters,
1970
25.

Sbei N., Listratova A.V., Titov A.A., Voskressensky L.G.
Synthesis,
2019
26.
Sokolov A.A., Syroeshkin M.A., Begunov R.S., Rusakova N.N., Gultyai V.P.
Mendeleev Communications,
2012
27.
![Efficient electrochemical synthesis of pyrido[1,2-a]benzimidazoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sokolov A.A., Syroeshkin M.A., Solkan V.N., Shebunina T.V., Begunov R.S., Mikhal’chenko L.V., Leonova M.Y., Gultyai V.P.
Russian Chemical Bulletin,
2014
28.
Begunov R.S., Sokolov A.A., Fedyanin I.V., Sakulina V.O., Syroeshkin M.A.
Mendeleev Communications,
2019
29.

Cardoso D.S., Šljukić B., Santos D.M., Sequeira C.A.
Organic Process Research and Development,
2017
30.

Shatskiy A., Lundberg H., Kärkäs M.D.
ChemElectroChem,
2019
31.
10.1016/j.mencom.2020.09.027_bib0155
Organic Electrochemistry,
2001
32.

Siqueira R.P., Barbosa É.D., Polêto M.D., Righetto G.L., Seraphim T.V., Salgado R.L., Ferreira J.G., Barros M.V., de Oliveira L.L., Laranjeira A.B., Almeida M.R., Júnior A.S., Fietto J.L., Kobarg J., de Oliveira E.B., et. al.
PLoS ONE,
2015
33.
Syroeshkin M.A., Mendkovich A.S., Mikhalchenko L.V., Rusakov A.I., Gul’tyai V.P.
Mendeleev Communications,
2009
34.
Electrochemically initiated transformation of 4-nitrophenylhydroxylamine into 4,4′-dinitroazobenzene
Syroeshkin M.A., Mikhalchenko L.V., Leonova M.Y., Mendkovich A.S., Rusakov A.I., Gul’tyai V.P.
Mendeleev Communications,
2011
35.

Mendkovich A.S., Syroeshkin M.A., Mikhalchenko L.V., Rusakov A.I., Gultyai V.P.
Russian Chemical Bulletin,
2008
36.

Syroeshkin M.A., Mendkovich A.S., Mikhal’chenko L.V., Rusakov A.I., Gul’tyai V.P.
Russian Chemical Bulletin,
2009
37.
Mikhalchenko L.V., Mendkovich A.S., Syroeshkin M.A., Gul’tyai V.P.
Mendeleev Communications,
2009
38.
Bruker APEX3, Bruker AXS, Madison, WI, USA, 2018.
39.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
40.
10.1016/j.mencom.2020.09.027_bib0195
Sheldrick
Acta Crystallogr.,
2015
41.
10.1016/j.mencom.2020.09.027_bib0200
Sheldrick
Acta Crystallogr.,
2015