Keywords
cancer cell lines
cytotoxic activity
heterocyclization
isothiouronic salts
nitriles
pyrimidines
sulfones
Abstract
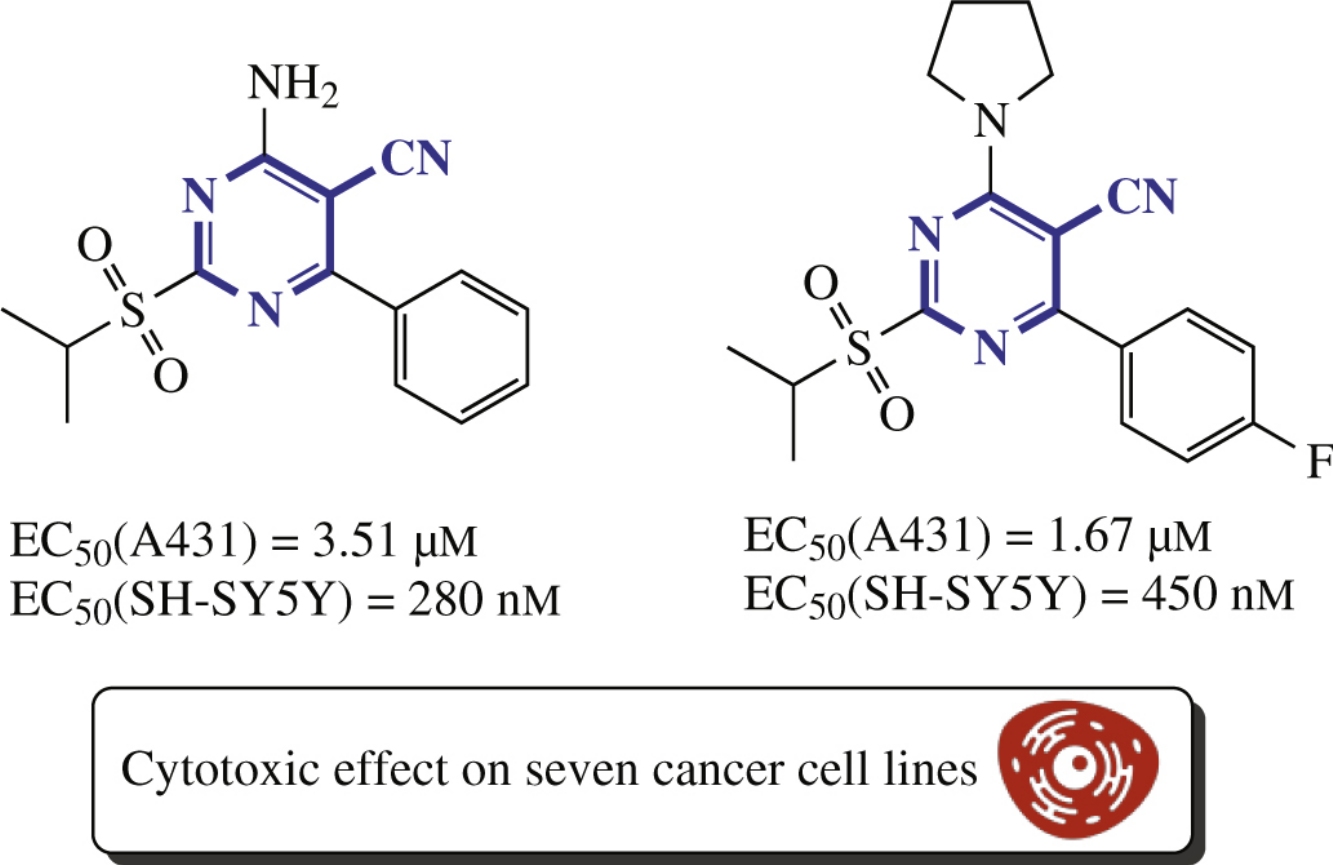
Novel 4-amino-5-cyano-2-sulfonylpyrimidines were prepared based on three-component cyclization between isothiouronium salts, benzaldehydes and malononitrile, followed by oxidation of the sulfide moiety with Oxone. The cytotoxic activity of the synthesized compounds, as well as the induction of apoptosis, inhibition of the cell cycle and proliferation tests were performed on selected cancer cell lines A431, A549, A375, HCT 116, MCF7, LNCap and SH-SY5Y.
References
1.

Fabbro D.
Molecular Pharmacology,
2014
2.

Jiao Q., Bi L., Ren Y., Song S., Wang Q., Wang Y.
Molecular Cancer,
2018
3.

Ma D., Wang L., Lai Q., Peng K., Li X., Li Z., Liu L., Luo Z., Liu S.
Bioorganic and Medicinal Chemistry Letters,
2018
4.
Egorov V.A., Gimalova F.A., Zileeva Z.R., Zainullina L.F., Vakhitova Y.V., Miftakhov M.S.
Mendeleev Communications,
2019
5.

Botta L., Filippi S., Bizzarri B.M., Meschini R., Caputo M., Proietti-De-Santis L., Iside C., Nebbioso A., Gualandi G., Saladino R.
Bioorganic and Medicinal Chemistry Letters,
2019
6.
Bagautdinova R.K., Vagapova L.I., Smolobochkin A.V., Gazizov A.S., Burilov A.R., Pudovik M.A., Voloshina A.D.
Mendeleev Communications,
2019
7.
Vostrikov N.S., Spirikhin L.V., Lobov A.N., Gimazetdinov A.M., Zileeva Z.R., Vakhitova Y.V., Macaev Z.R., Pivnitsky K.K., Miftakhov M.S.
Mendeleev Communications,
2019
8.

Uecker A., Sicker M., Beckers T., Mahboobi S., Hägerstrand D., Östman A., Böhmer F.
Anti-Cancer Drugs,
2010
9.
Zorina A.D., Nikiforova N.S., Zarubaev V.V., Marchenko S.A., Selivanov S.I., Starova G.L., Mehtiev A.R., Rodionov E.I., Rodionova A.A., Trifonov R.E.
Mendeleev Communications,
2019
10.

Rusinov V.L., Charushin V.N., Chupakhin O.N.
Russian Chemical Bulletin,
2018
11.

Ranson M., Mansoor W., Jayson G.
Expert Review of Anticancer Therapy,
2002
12.

Cross D.A., Ashton S.E., Ghiorghiu S., Eberlein C., Nebhan C.A., Spitzler P.J., Orme J.P., Finlay M.R., Ward R.A., Mellor M.J., Hughes G., Rahi A., Jacobs V.N., Brewer M.R., Ichihara E., et. al.
Cancer Discovery,
2014
13.

Deininger M.W., Druker B.J.
Pharmacological Reviews,
2003
14.

Ghodasara H.B., Trivedi A.R., Kataria V.B., Patel B.G., Shah V.H.
Medicinal Chemistry Research,
2013
15.

Stella A., Van Belle K., De Jonghe S., Louat T., Herman J., Rozenski J., Waer M., Herdewijn P.
Bioorganic and Medicinal Chemistry,
2013
16.

Patel L., Chandrasekhar J., Evarts J., Haran A.C., Ip C., Kaplan J.A., Kim M., Koditek D., Lad L., Lepist E., McGrath M.E., Novikov N., Perreault S., Puri K.D., Somoza J.R., et. al.
Journal of Medicinal Chemistry,
2016
17.

Liu C., Wrobleski S.T., Lin J., Ahmed G., Metzger A., Wityak J., Gillooly K.M., Shuster D.J., McIntyre K.W., Pitt S., Shen D.R., Zhang R.F., Zhang H., Doweyko A.M., Diller D., et. al.
Journal of Medicinal Chemistry,
2005
18.

Myers S.M., Bawn R.H., Bisset L.C., Blackburn T.J., Cottyn B., Molyneux L., Wong A., Cano C., Clegg W., Harrington R.W., Leung H., Rigoreau L., Vidot S., Golding B.T., Griffin R.J., et. al.
ACS Combinatorial Science,
2016
19.

Anderson L., Zhou M., Sharma V., McLaughlin J.M., Santiago D.N., Fronczek F.R., Guida W.C., McLaughlin M.L.
Journal of Organic Chemistry,
2010
20.

Ding L., Wang Z., Sun X., Yang J., Ma C., Li W., Liu H.
Bioorganic and Medicinal Chemistry Letters,
2017