Keywords
1,2,4-triazoles
amino acids
Boc-deprotection
cyanamides
heterocyclization
hydrazides
piperazines
pyrimidines
Abstract
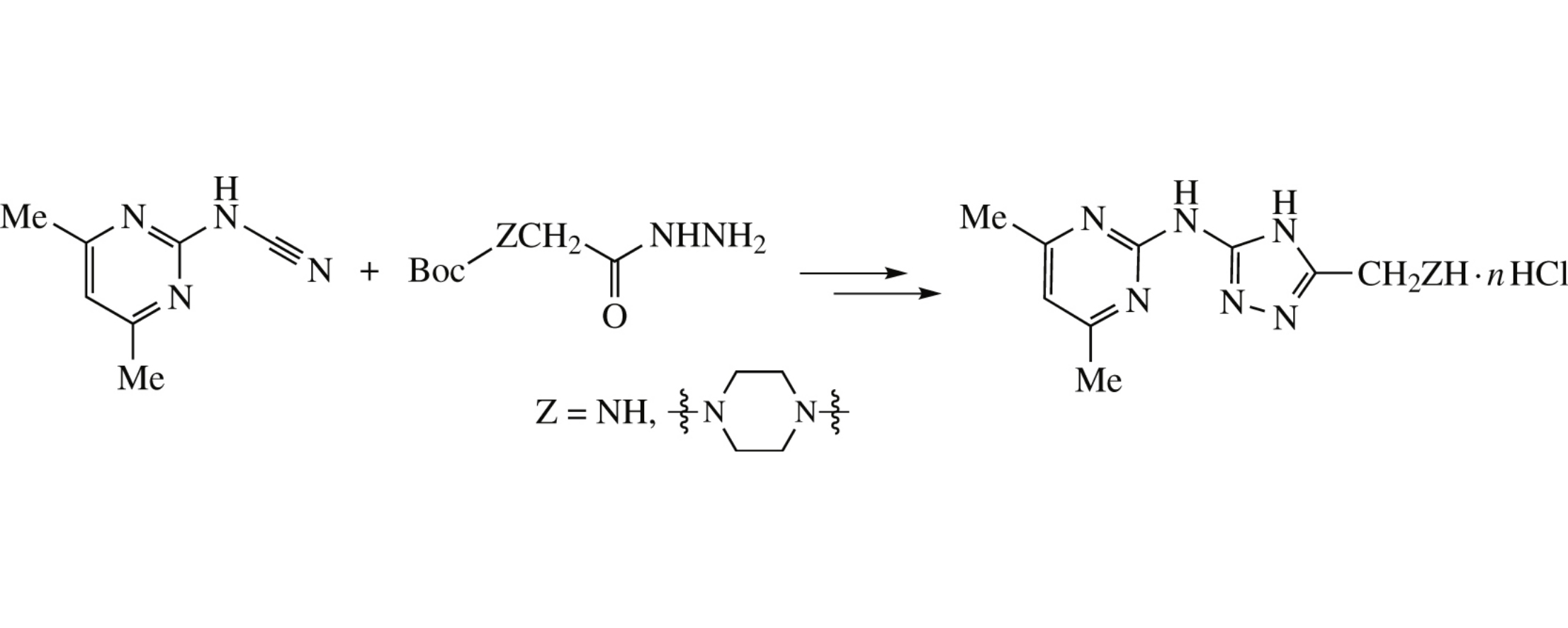
The Ni(acac)2-catalyzed 1,2,4-triazole formation from cyanamides and carbohydrazides was extended onto N-(4,6-dimethylpyrimidin-2-yl)cyanamide and glycine hydrazides. The obtained N-(5-aminomethyl-4H-1,2,4-triazol-3-yl)-4,6-dimethylpyrimidin-2-amines may be attractive for the estimation of their biological activity.
References
1.
Ananikov V.P., Khokhlova E.A., Egorov M.P., Sakharov A.M., Zlotin S.G., Kucherov A.V., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2015
2.
10.1016/j.mencom.2020.07.032_bib0010
Design of Hybrid Molecules for Drug Development,
2017
3.

Shaikh M.S., Palkar M.B., Patel H.M., Rane R.A., Alwan W.S., Shaikh M.M., Shaikh I.M., Hampannavar G.A., Karpoormath R.
RSC Advances,
2014
4.

Decker M.
Current Medicinal Chemistry,
2011
5.

B. Tsogoeva S.
Mini-Reviews in Medicinal Chemistry,
2010
6.
Sadchikova E.V., Alexeeva D.L., Nenajdenko V.G.
Mendeleev Communications,
2019
7.

Glanville A.R., Scott A.I., Morton J.M., Aboyoun C.L., Plit M.L., Carter I.W., Malouf M.A.
Journal of Heart and Lung Transplantation,
2005
8.
Chudinov M.V., Matveev A.V., Prutkov A.N., Konstantinova I.D., Fateev I.V., Prasolov V.S., Smirnova O.A., Ivanov A.V., Galegov G.A., Deryabin P.G.
Mendeleev Communications,
2016
9.

Cha R., Sobel J.D.
Expert Review of Anti-Infective Therapy,
2004
10.
Bel’skaya N.P., Demina M.A., Sapognikova S.G., Fan Z., Zhang H., Dehaen W., Bakulev V.A.
Arkivoc,
2008
11.
G. Mitchell, WO Patent 144234 A1, 2013.
12.
K. Baumann, A. Flohr, E. Goetschi, H. Jacobsen, S. Jolidon and T. Luebbers, US Patent 20090215759 A1, 2009.
13.

Bell K., Sunose M., Ellard K., Cansfield A., Taylor J., Miller W., Ramsden N., Bergamini G., Neubauer G.
Bioorganic and Medicinal Chemistry Letters,
2012
14.

Gregory T.F., Wright J.L., Wise L.D., Meltzer L.T., Serpa K.A., Konkoy C.S., Whittemore E.R., Woodward R.M.
Bioorganic and Medicinal Chemistry Letters,
2000
15.
Shagun L.G., Dorofeev I.A., Zhilitskaya L.V., Larina L.I., Yarosh N.O.
Mendeleev Communications,
2015
16.

Baumgarth M., Beier N., Gericke R.
Journal of Medicinal Chemistry,
1997
17.

Wustrow D., Belliotti T., Glase S., Kesten S.R., Johnson D., Colbry N., Rubin R., Blackburn A., Akunne H., Corbin A., Davis M.D., Georgic L., Whetzel S., Zoski K., Heffner T., et. al.
Journal of Medicinal Chemistry,
1998
18.
![4,5-Dihydro-1H-pyrazolo[3,4-d]pyrimidine containing phenothiazines as antitubercular agents](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Siddiqui A.B., Trivedi A.R., Kataria V.B., Shah V.H.
Bioorganic and Medicinal Chemistry Letters,
2014
19.

Cohen A., Suzanne P., Lancelot J., Verhaeghe P., Lesnard A., Basmaciyan L., Hutter S., Laget M., Dumètre A., Paloque L., Deharo E., Crozet M.D., Rathelot P., Dallemagne P., Lorthiois A., et. al.
European Journal of Medicinal Chemistry,
2015
20.

Antimalarial activity of new water-soluble dihydroartemisinin derivatives. 3. Aromatic amine analogs
Lin A.J., Li L.Q., Klayman D.L., George C.F., Flippen-Anderson J.L.
Journal of Medicinal Chemistry,
1990
21.
Verbitskiy E.V., Baskakova S.A., Gerasimova N.A., Evstigneeva N.P., Zil’berberg N.V., Kungurov N.V., Kravchenko M.A., Rusinov G.L., Chupakhina O.N., Charushin V.N.
Mendeleev Communications,
2018
22.

Mo C., Zhang Z., Guise C.P., Li X., Luo J., Tu Z., Xu Y., Patterson A.V., Smaill J.B., Ren X., Lu X., Ding K.
ACS Medicinal Chemistry Letters,
2017
23.

Paul R., Hallett W.A., Hanifin J.W., Reich M.F., Johnson B.D., Lenhard R.H., Dusza J.P., Kerwar S.S., Lin Y.I.
Journal of Medicinal Chemistry,
1993
24.

Koroleva E.V., Gusak K.N., Ignatovich Z.V.
Russian Chemical Reviews,
2010
25.

Rusinov V.L., Charushin V.N., Chupakhin O.N.
Russian Chemical Bulletin,
2018
26.

Dorokhov V.A., Gordeev M.F., Dem'yanets Z.K., Bochkareva M.N., Bogdanov V.S.
Russian Chemical Bulletin,
1989
27.

Dorokhov V.A., Gordeev M.F., Bogdanov V.S.
Russian Chemical Bulletin,
1988
28.

Dorokhov V.A., Gordeev M.F., Shashkova E.M., Komkov A.V., Bogdanov V.S.
Russian Chemical Bulletin,
1991
29.

Prezent M.A., Daeva E.D., Baranin S.V., Dorokhov V.A.
Russian Chemical Bulletin,
2015
30.
![A convenient route to the 1,2,5-oxadiazole-substituted 1,2,4-triazolo[1,5-a]pyrimidine derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Prezent M.A., Baranin S.V.
Chemistry of Heterocyclic Compounds,
2019
31.
Prezent M.A., Daeva E.D., Baranin S.V., Zavarzin I.V.
Mendeleev Communications,
2017
32.
Astakhov A.V., Suponitsky K.Y., Chernyshev V.M.
Mendeleev Communications,
2018
33.

Birtwell S.
Journal of the Chemical Society (Resumed),
1953
34.

Borg S., Estenne-Bouhtou G., Luthman K., Csoeregh I., Hesselink W., Hacksell U.
Journal of Organic Chemistry,
1995
35.

Jia J., Wang K., Shi W., Chen S., Li X., Ma H.
Chemistry - A European Journal,
2010