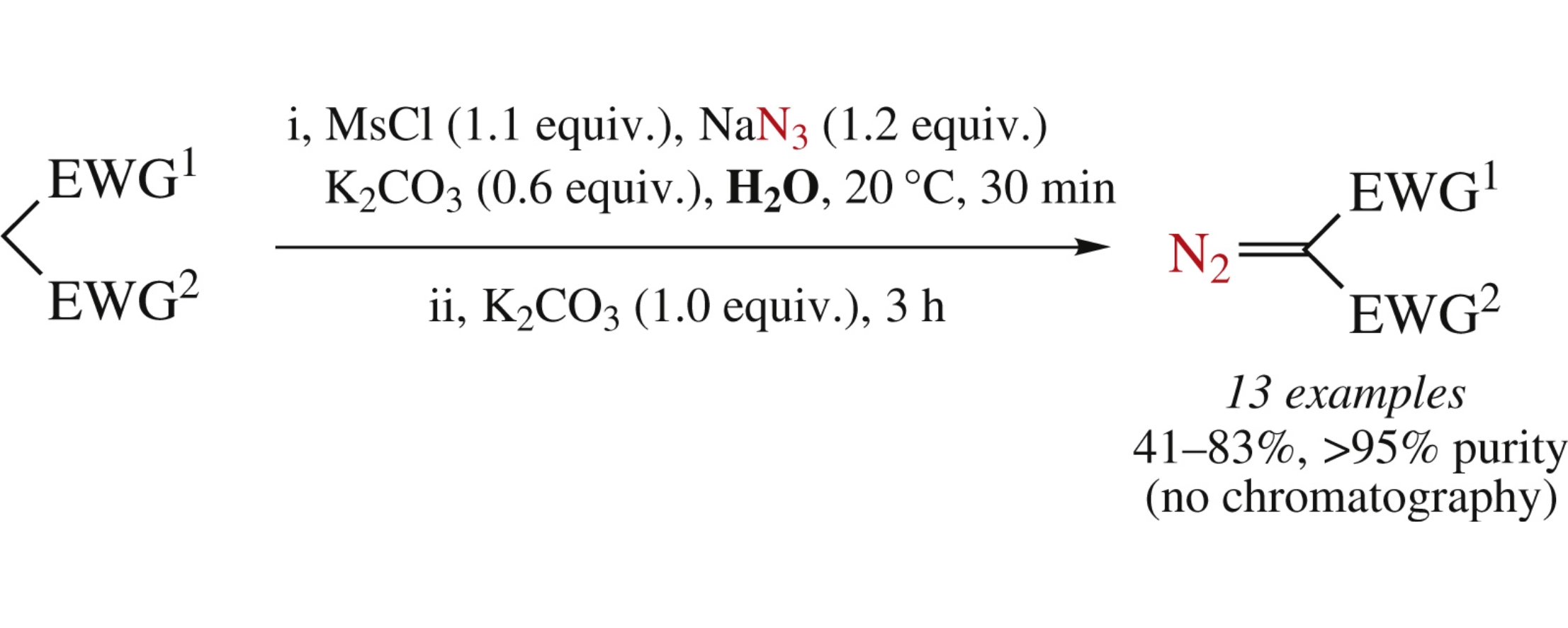Keywords
in situ generation
active methylene compounds
aqueous medium
diazo transfer
mesyl azide
sulfonyl azides
Abstract
Mesyl azide generated in situ in aqueous medium converted a range of active methylene substrates into the corresponding diazo compounds in good yields and high purity with no need for chromatographic purification. The products thus obtained are suitable for the subsequent RhII-catalyzed O–H insertions with no need for chromatography in the interim.
References
1.

Ford A., Miel H., Ring A., Slattery C.N., Maguire A.R., McKervey M.A.
Chemical Reviews,
2015
2.

Kirmse W.
European Journal of Organic Chemistry,
2002
3.
Sadchikova E.V., Alexeeva D.L., Nenajdenko V.G.
Mendeleev Communications,
2019
4.
10.1016/j.mencom.2020.05.037_bib0020
Doyle
Modern Catalytic Methods for Organic Synthesis with Diazo Compounds: From Cyclopropanes to Ylides,
1998
5.
6.

7.

Curphey T.J.
Organic Preparations and Procedures International,
1981
8.

Sattely E.S., Meek S.J., Malcolmson S.J., Schrock R.R., Hoveyda A.H.
Journal of the American Chemical Society,
2008
9.

O'Mahony R.M., Lynch D., Hayes H.L., Ní Thuama E., Donnellan P., Jones R.C., Glennon B., Collins S.G., Maguire A.R.
European Journal of Organic Chemistry,
2017
10.

Deadman B.J., O'Mahony R.M., Lynch D., Crowley D.C., Collins S.G., Maguire A.R.
Organic and Biomolecular Chemistry,
2016
11.

Dar’in D., Kantin G., Krasavin M.
Chemical Communications,
2019
12.
![A novel approach to biologically relevant oxazolo[5,4-d]pyrimidine-5,7-diones via readily available diazobarbituric acid derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Gecht M., Kantin G., Dar'in D., Krasavin M.
Tetrahedron Letters,
2019
13.

Shershnev I., Dar'in D., Chuprun S., Kantin G., Bakulina O., Krasavin M.
Tetrahedron Letters,
2019
14.

Dar’in D., Kantin G., Krasavin M.
Synthesis,
2019