Keywords
3-carene
4,6-dibromoisatin
aldol reaction
convolutamydine A
isatin
α-pinene
β-amino alcohols
Abstract
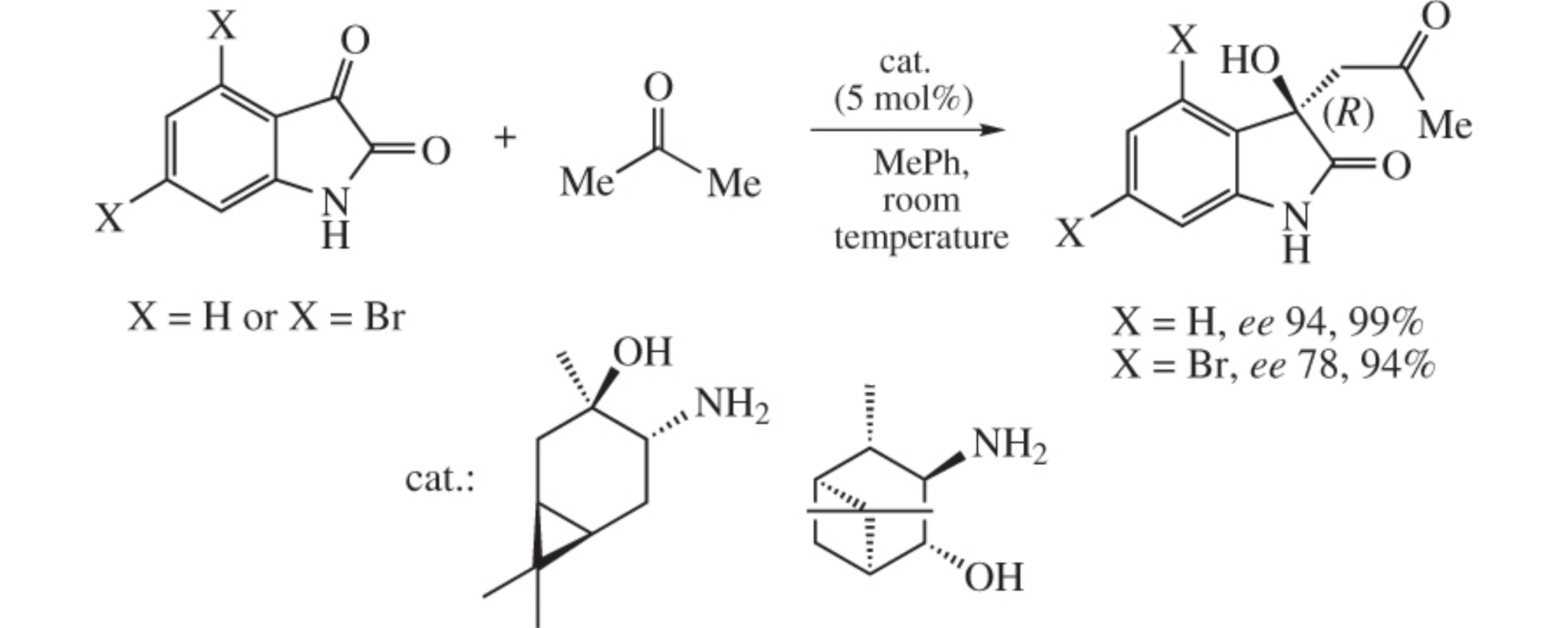
Asymmetric aldol reactions of isatin and 4,6-dibromoisatin with acetone are efficiently catalyzed by β-amino alcohols derived from α-pinene and 3-carene. The target compounds can be isolated by crystallization from toluene, which eliminates the need for using chromatography and makes the asymmetric synthesis of (R)-convolutamydine A (up to 94% ee and yield 75%) simple and convenient.
References
1.
10.1016/j.mencom.2020.03.005_bib0005
Modern Methods in Stereoselective Aldol Reactions,
2013
2.

Malkov A.V., Kabeshov M.A., Bella M., Kysilka O., Malyshev D.A., Pluháčková K., Kočovský P.
Organic Letters,
2007
3.

Zhong G., Fan J., Barbas C.F.
Tetrahedron Letters,
2004
4.

Sakthivel K., Notz W., Bui T., Barbas C.F.
Journal of the American Chemical Society,
2001
5.

Deng D., Liu P., Ji B., Fu W., Li L.
Catalysis Letters,
2010
6.

Gut B., Mlynarski J.
European Journal of Organic Chemistry,
2015
7.

Tzeng Z., Chen H., Huang C., Chen K.
Tetrahedron Letters,
2008
8.

VANLALDINPUIA K., BORA P., BEZ G.
Journal of Chemical Sciences,
2017
9.

Zalevskaya O.A., Gur'eva Y.A., Kutchin A.V.
Russian Chemical Reviews,
2019
10.

Marqués-López E., Herrera R.P., Christmann M.
Natural Product Reports,
2010
11.

Kinsella M., Duggan P.G., Lennon C.M.
Tetrahedron Asymmetry,
2011
12.

Allu S., Molleti N., Panem R., Singh V.K.
Tetrahedron Letters,
2011
13.

Guo Q., Bhanushali M., Zhao C.
Angewandte Chemie - International Edition,
2010
14.

Nakamura S., Hara N., Nakashima H., Kubo K., Shibata N., Toru T.
Chemistry - A European Journal,
2008
15.

Wan Y., Yuan R., Cui H., Zhang X., Li M., Xu J., Dou P., Zhang L., Wu H.
Research on Chemical Intermediates,
2018
16.

Yu B., Xing H., Yu D., Liu H.
Beilstein Journal of Organic Chemistry,
2016
17.

Ogasawara A., Subba Reddy U.V., Seki C., Okuyama Y., Uwai K., Tokiwa M., Takeshita M., Nakano H.
Tetrahedron Asymmetry,
2016
18.

Peddibhotla S.
Current Bioactive Compounds,
2009
19.

Hibino S., Choshi T.
Natural Product Reports,
2001
20.

Tang Y., Sattler I., Thiericke R., Grabley S., Feng X.
European Journal of Organic Chemistry,
2001
21.

KOGUCHI Y., KOHNO J., NISHIO M., TAKAHASHI K., OKUDA T., OHNUKI T., KOMATSUBARA S.
Journal of Antibiotics,
2012
22.

Labroo R.B., Cohen L.A.
Journal of Organic Chemistry,
1990
23.

Tokunaga T., Hume W.E., Umezome T., Okazaki K., Ueki Y., Kumagai K., Hourai S., Nagamine J., Seki H., Taiji M., Noguchi H., Nagata R.
Journal of Medicinal Chemistry,
2001
24.

Hewawasam* P., Meanwell N.A., Gribkoff V.K., Dworetzky S.I., Boissard C.G.
Bioorganic and Medicinal Chemistry Letters,
1997
25.

Boechat N., Kover W., Bongertz V., Bastos M., Romeiro N., Azevedo M., Wollinger W.
Medicinal Chemistry,
2007
26.

Hewawasam P., Erway M., Moon S.L., Knipe J., Weiner H., Boissard C.G., Post-Munson D.J., Gao Q., Huang S., Gribkoff V.K., Meanwell N.A.
Journal of Medicinal Chemistry,
2002
27.

Beloglazkina A.A., Karpov N.A., Mefedova S.R., Polyakov V.S., Skvortsov D.A., Kalinina M.A., Tafeenko V.A., Majouga A.G., Zyk N.V., Beloglazkina E.K.
Russian Chemical Bulletin,
2019
28.

Convolutamydine A, a novel bioactive hydroxyoxindole alkaloid from marine bryozoan Amathia convoluta
Kamano Y., Zhang H., Ichihara Y., Kizu H., Komiyama K., Pettit G.R.
Tetrahedron Letters,
1995
29.

Luppi G., Monari M., Corrêa R.J., de A. Violante F., Pinto A.C., Kaptein B., Broxterman Q.B., Garden S.J., Tomasini C.
Tetrahedron,
2006
30.

Banina O.A., Sudarikov D.V., Nigmatov A.G., Frolova L.L., Slepukhin P.A., Zlotin S.G., Kutchin A.V.
Russian Chemical Bulletin,
2017
31.

Frolova L.L., Sudarikov D.V., Alekseev I.N., Banina O.A., Slepukhin P.A., Kutchin A.V.
Russian Journal of Organic Chemistry,
2017
32.

Bagheri M., Azizi N., Saidi M.R.
Canadian Journal of Chemistry,
2005
33.

Chakraborty C., Layek A., Ray P.P., Malik S.
European Polymer Journal,
2014
34.

SHEPHERD R.G.
Journal of Organic Chemistry,
1947
35.

Jnaneshwara G.K., Bedekar A.V., Deshpande V.H.
Synthetic Communications,
1999
36.
Liang D., Wang Y.
Mendeleev Communications,
2019