Keywords
adamantane
carcinoma A549 cell line.
colchicine
colchicine binding site
dehydroadamantane
depolymerisation of microtubules
metabolic stability
tubulin
tubuloclustin
Abstract
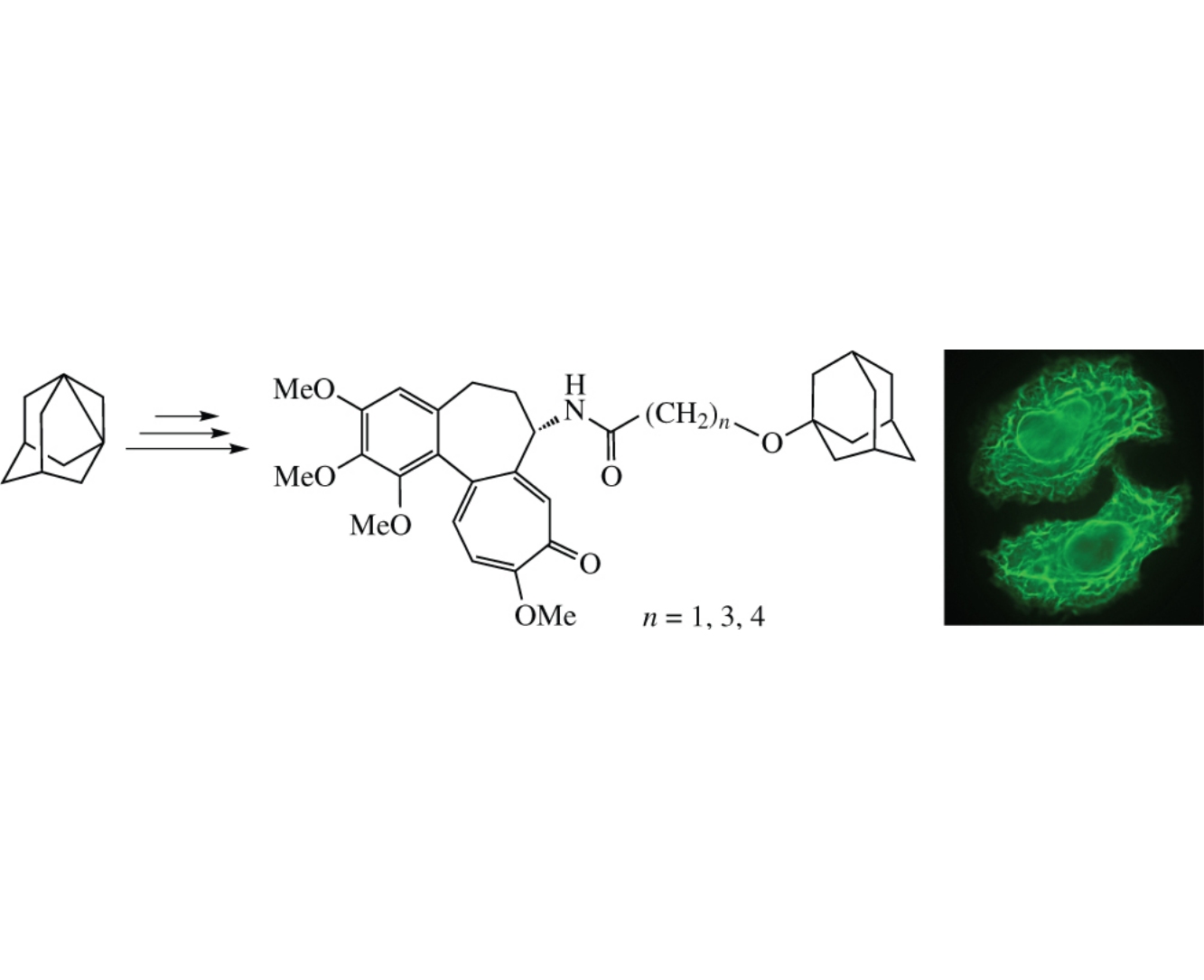
New analogues of anticancer agent tubuloclustin N-[7-(adamantan-1-yloxy)-7-heptanoyl]-N-deacetylcolchicine with ether moiety in the linker between colchicine and adamantine fragments were synthesized from w-(adamantan-1-yloxy)alkan- 1-ols. These compounds effectively inhibited growth of human lung carcinoma cell line A549 (IC50=5–15.5nM), induced both apoptosis and formation of tubulin clusters. The conjugates lacking ester carbonyl in the linker exhibit improved metabolic stability and are promising for further cytotoxicity studies in vivo.
References
1.

Kumar B., Kumar R., Skvortsova I., Kumar V.
Current Cancer Drug Targets,
2016
2.

Lu Y., Chen J., Xiao M., Li W., Miller D.D.
Pharmaceutical Research,
2012
3.

Vilanova C., Díaz-Oltra S., Murga J., Falomir E., Carda M., Marco J.A.
Bioorganic and Medicinal Chemistry Letters,
2015
4.

Thomopoulou P., Sachs J., Teusch N., Mariappan A., Gopalakrishnan J., Schmalz H.
ACS Medicinal Chemistry Letters,
2015
5.

Zefirova O.N., Lemcke H., Lantow M., Nurieva E.V., Wobith B., Onishchenko G.E., Hoenen A., Griffiths G., Zefirov N.S., Kuznetsov S.A.
ChemBioChem,
2013
6.

Zefirova O.N., Nurieva E.V., Wobith B., Gogol V.V., Zefirov N.A., Ogonkov A.V., Shishov D.V., Zefirov N.S., Kuznetsov S.A.
Molecular Diversity,
2017
7.

Zefirov N.A., Evteeva Y.A., Fatkulin A.R., Schulz S., Kuznetsov S.A., Zefirova O.N.
Pharmaceutical Chemistry Journal,
2019
8.

Zefirova O.N., Nurieva E.V., Glazkova Y.S., Zefirov N.A., Mamaeva A.V., Wobith B., Romanenko V.I., Lesnaya N.A., Treshchalina E.M., Kuznetsov S.A.
Pharmaceutical Chemistry Journal,
2014
9.
Zefirov N.A., Hoppe M., Kuznetsova I.V., Chernyshov N.A., Grishin Y.K., Maloshitskaya O.A., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2018
10.

Prota A.E., Danel F., Bachmann F., Bargsten K., Buey R.M., Pohlmann J., Reinelt S., Lane H., Steinmetz M.O.
Journal of Molecular Biology,
2014
11.

Trott O., Olson A.J.
Journal of Computational Chemistry,
2009
12.

Pettersen E.F., Goddard T.D., Huang C.C., Couch G.S., Greenblatt D.M., Meng E.C., Ferrin T.E.
Journal of Computational Chemistry,
2004
13.
![Chemical Transformations of Tetracyclo[3.3.1.13,7.01,3]decane (1,3-Dehydroadamantane): VII. Reaction of 1,3-Dehydroadamantane with Alkanediols and Amino Alcohols](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Butov G.M., Mokhov V.M.
Russian Journal of Organic Chemistry,
2018
14.

Bagnato J.D., Eilers A.L., Horton R.A., Grissom C.B.
Journal of Organic Chemistry,
2004
15.

Zefirov N.A., Evteeva Y.A., Wobith B., Kuznetsov S.A., Zefirova O.N.
Structural Chemistry,
2018
16.

Gerlier D., Thomasset N.
Journal of Immunological Methods,
1986
17.

Potten C.
British Journal of Cancer,
1996
18.