Keywords
charge-discharge cycling
crown ethers
electrochemical impedance
lithium battery
lithium ferro phosphate cathode.
lithium ions
organic electrolyte
quantum-chemical modeling
Abstract
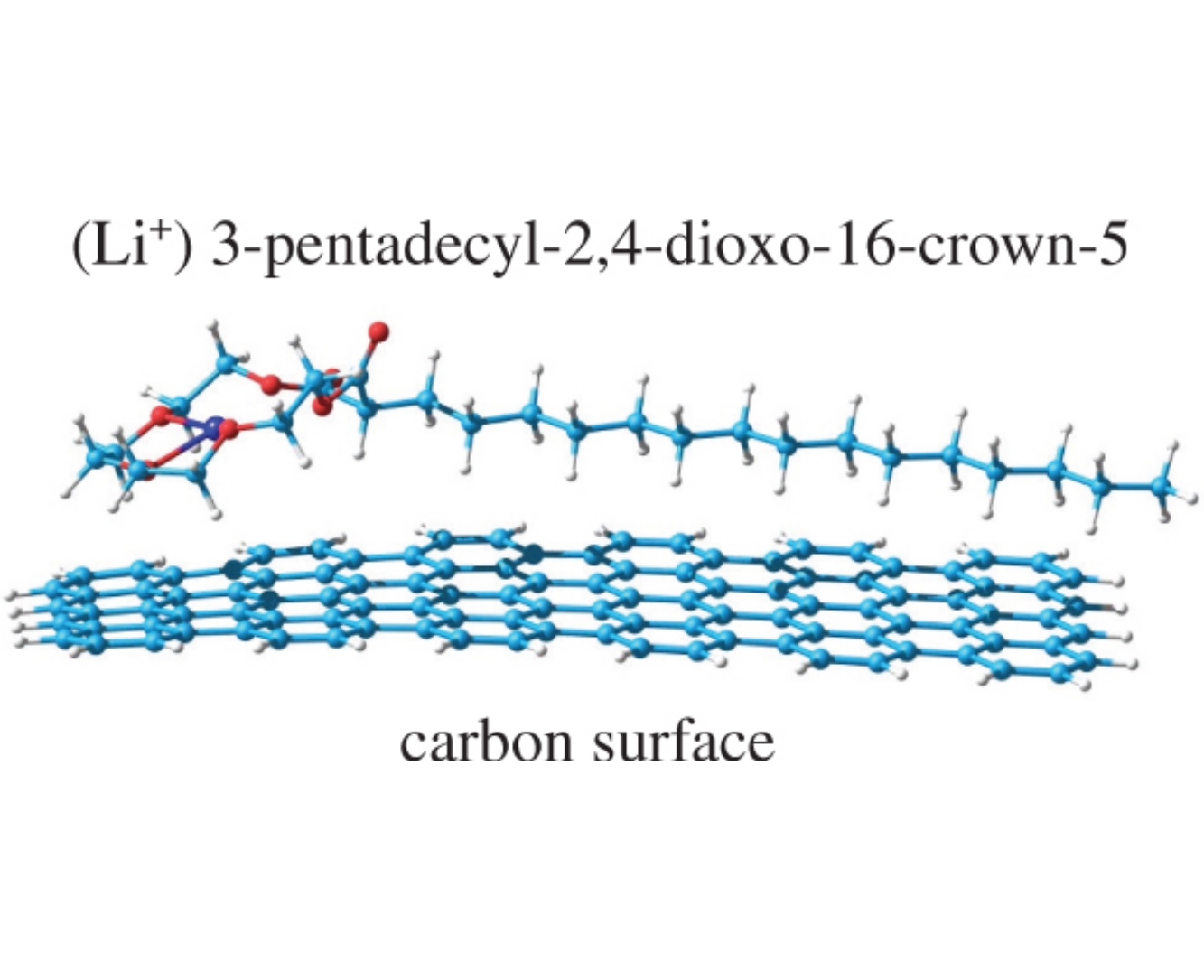
A new crown ether, 3-pentadecyl-2,4-dioxo-16-crown-5, with surfactant properties has been synthesized in three simple steps from diethyl pentadecylmalonate. Quantum chemical calculations show that this crown ether is capable of adsorbing both on the surface of the lithium anode and on the surface of carbon. The effectiveness of its application as a functional additive in organic electrolyte for lithium batteries has been experimentally confirmed.
References
1.

Thompson M.D., Bradshaw J.S., Nielsen S.F., Bishop C.T., Cox F.T., Fore P.E., Maas G.E., Izatt R.M., Christensen J.J.
Tetrahedron,
1977
2.

Geneste P., Guida A., Reminiac C., Amblard G., Gavach C.
Tetrahedron Letters,
1981
3.

Solov’ev V.P., Tsivadze A.Y.
Protection of Metals and Physical Chemistry of Surfaces,
2015
4.
![Optimizing Lithium Ion Conduction through Crown Ether-Based Cylindrical Channels in [Ni(dmit)2]− Salts](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ichihashi K., Konno D., Date T., Nishimura T., Maryunina K.Y., Inoue K., Nakaya T., Toyoda K., Tatewaki Y., Akutagawa T., Nakamura T., Nishihara S.
Chemistry of Materials,
2018
5.
Yarmolenko O.V., Tulibaeva G.Z., Khatmullina K.G., Bogdanova L.M., Shestakov A.F.
Mendeleev Communications,
2016
6.

Ignatova A.A., Yarmolenko O.V., Tulibaeva G.Z., Shestakov A.F., Fateev S.A.
Journal of Power Sources,
2016
7.
A.-C. Gentschev, H. Hain, S. Scharner and B. Stiaszny, Patent WO 2017216149A1, 2017.
8.
J. Y. Hwang and H. Y. Ha, Patent KR 2018001967, 2018.
9.

Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1996
10.

Laikov D.N.
Chemical Physics Letters,
1997
11.

Sharma B.D., Biswas A.B.
Analytical Chemistry,
1958
12.

Pugia M.J., Knudsen B.E., Bartsch R.A.
Journal of Organic Chemistry,
1987
13.

Bradshaw J.S., Hansen L.D., Nielsen S.F., Thompson M.D., Reeder R.A., Izatt R.M., Christensen J.J.
Journal of the Chemical Society Chemical Communications,
1975
14.

Bradshaw J.S., Jolley S.T., Jones B.A.
Journal of Heterocyclic Chemistry,
1980
15.

Baskakova Y.V., Yarmolenko O.V., Shuvalova N.I., Tulibaeva G.Z., Efimov O.N.
Russian Journal of Electrochemistry,
2006
16.

Tulibaeva G.Z., Yarmolenko O.V., Shestakov A.F.
Russian Chemical Bulletin,
2009
17.

Baymuratova G.R., Slesarenko A.A., Yudina A.V., Yarmolenko O.V.
Russian Chemical Bulletin,
2018