Abstract
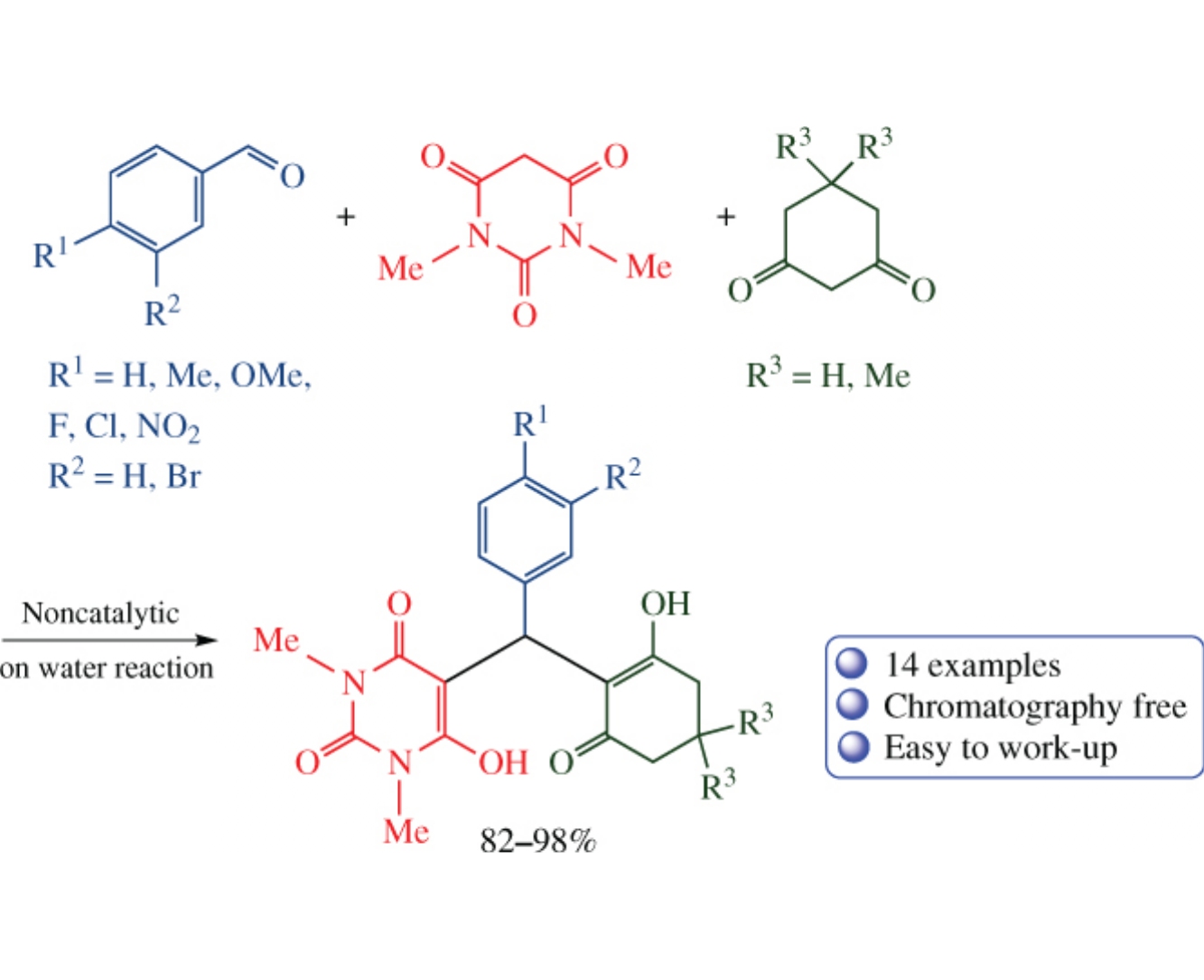
Heating aldehydes, N,N’-dimethylbarbituric acid and cyclohexane-1,3-diones in water results in fast formation of Knoevenagel–Michael adducts in 82–98% yields. This multicomponent process opens facile, efficient and environmentally benign way to the new functionalized [(2-hydroxy-6-oxocyclohex-1-en-1-yl)(aryl)methyl]pyrimidine-2,4(1H,3H)-diones bearing both barbituric acid and cyclohexane-1,3-dionemoieties separated by arylmethylene spacer, which are promising compounds for biomedical applications including analeptics, anti-AIDS and anticancer remedies.
References
1.
10.1016/j.mencom.2020.01.005_bib0005
Organic Reactions in Water: Principles, Strategies and Applications,
2007
2.

Simon M., Li C.
Chemical Society Reviews,
2012
3.

Narayan S., Muldoon J., Finn M.G., Fokin V.V., Kolb H.C., Sharpless K.B.
Angewandte Chemie - International Edition,
2005
4.

Butler R.N., Coyne A.G., Cunningham W.J., Moloney E.M.
Journal of Organic Chemistry,
2013
5.

6.

7.
10.1016/j.mencom.2020.01.005_bib0035
Multicomponent Reactions,
2005
8.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Goloveshkin A.S., Ushakov I.E., Egorov M.P.
Mendeleev Communications,
2018
9.
Vereshchagin A.N., Karpenko K.A., Elinson M.N., Dorofeeva E.O., Goloveshkin A.S., Egorov M.P.
Mendeleev Communications,
2018
10.
Elinson M.N., Vereshchagin A.N., Anisina Y.E., Krymov S.K., Fakhrutdinov A.N., Egorov M.P.
Mendeleev Communications,
2019
11.

Butler R.N., Coyne A.G.
Organic and Biomolecular Chemistry,
2016
12.
Demchuk D.V., Elinson M.N., Nikishin G.I.
Mendeleev Communications,
2011
13.

Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Egorov M.P.
Comptes Rendus Chimie,
2013
14.

Wender P.A.
Natural Product Reports,
2014
15.

Benign by design: catalyst-free in-water, on-water green chemical methodologies in organic synthesis
Gawande M.B., Bonifácio V.D., Luque R., Branco P.S., Varma R.S.
Chemical Society Reviews,
2013
16.

Evans B.E., Rittle K.E., Bock M.G., DiPardo R.M., Freidinger R.M., Whitter W.L., Lundell G.F., Veber D.F., Anderson P.S., Chang R.S., Lotti V.J., Cerino D.J., Chen T.B., Kling P.J., Kunkel K.A., et. al.
Journal of Medicinal Chemistry,
1988
17.

18.

Uhlmann C., Fröscher W.
CNS Neuroscience and Therapeutics,
2009
19.

Naguib F.N., Levesque D.L., Eng-Chi Wang, Panzica R.P., El Kouni M.H.
Biochemical Pharmacology,
1993
20.

Pyrimidine-2,4,6-Triones: A New Effective and Selective Class of Matrix Metalloproteinase Inhibitors
Grams F., Brandstetter H., DAlò S., Geppert D., Krell H., Leinert H., Livi V., Menta E., Oliva A., Zimmermann G.
Biological Chemistry,
2001
21.

Maquoi E., Sounni N.E., Devy L., Olivier F., Frankenne F., Krell H., Grams F., Foidart J., Noël A.
Clinical Cancer Research,
2004
22.
![The first example of the cascade assembly of a spirocyclopropane structure: direct transformation of benzylidenemalononitriles and N,N′-dialkylbarbituric acids into substituted 2-aryl-4,6,8-trioxo-5,7-diazaspiro[2.5]octane-1,1-dicarbonitriles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Elinson M.N., Vereshchagin A.N., Stepanov N.O., Zaimovskaya T.A., Merkulova V.M., Nikishin G.I.
Tetrahedron Letters,
2010
23.
Elinson M.N., Ilovaisky A.I., Merkulova V.M., Zaimovskaya T.A., Nikishin G.I.
Mendeleev Communications,
2011
24.

Elinson M.N., Merkulova V.M., Ilovaisky A.I., Barba F., Batanero B.
Electrochimica Acta,
2011
25.
![Solvent-free and ‘on-water’ multicomponent assembling of aldehydes, 3-methyl-2-pyrazoline-5-one, and malononitrile: fast and efficient approach to medicinally relevant pyrano[2,3-c]pyrazole scaffold](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Elinson M.N., Nasybullin R.F., Ryzhkov F.V., Zaimovskaya T.A., Nikishin G.I.
Monatshefte fur Chemie,
2015
26.

Elinson M.N., Nasybullin R.F., Sokolova O.O., Zaimovskaya T.A., Egorov M.P.
Monatshefte fur Chemie,
2015
27.

Sheikhhosseini E., A. Bigdeli M., Habibi A., Balalaie S.
Heterocycles,
2011
28.

Barakat A., Soliman S.M., Elshaier Y.A., Ali M., Al-Majid A.M., Ghabbour H.A.
Journal of Molecular Structure,
2017
29.

Moosavi-Zare A.R., Zolfigol M.A., Zarei M., Zare A., Khakyzadeh V., Hasaninejad A.
Applied Catalysis A: General,
2013
30.

Moosavi-Zare A.R., Zolfigol M.A., Khaledian O., Khakyzadeh V., Farahani M.D., Kruger H.G.
New Journal of Chemistry,
2014