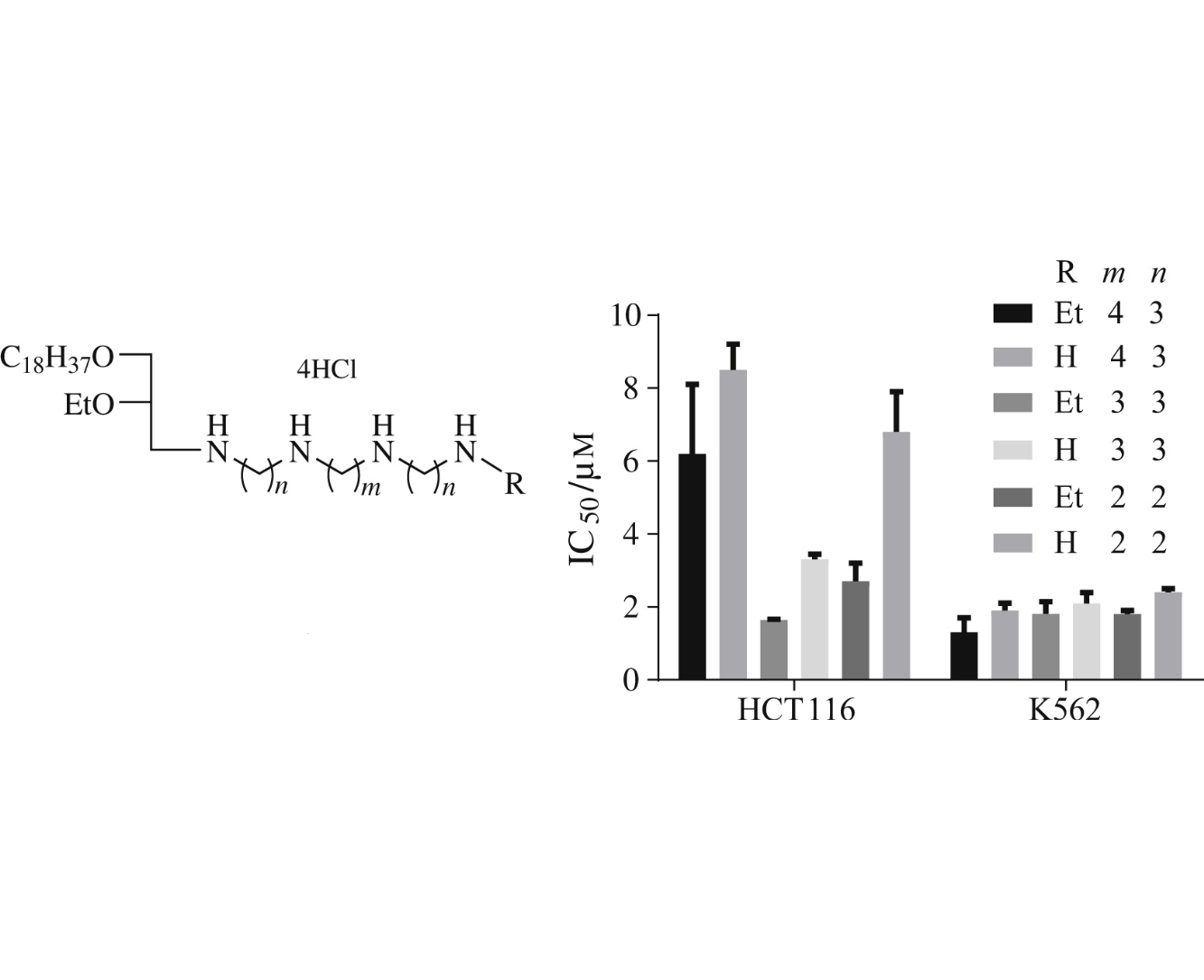
Abstract
New lipophilic polyamines in which natural or synthetic tetraamines were linked via the terminal NH2 groups to diglycerides and/or to short-chained aliphatic substituents were synthesized, and their cytotoxic activity was tested. Relationship between the structure of the synthesized compounds and their cytotoxicity and hemolytic effect was evaluated.
References
1.

2.

Ramani D., De Bandt J.P., Cynober L.
Clinical Nutrition,
2014
3.

Gerner E.W., Meyskens F.L.
Nature Reviews Cancer,
2004
4.

Basuroy U.K., Gerner E.W.
Journal of Biochemistry,
2006
5.

Casero R.A., Murray Stewart T., Pegg A.E.
Nature Reviews Cancer,
2018
6.

Murray-Stewart T.R., Woster P.M., Casero R.A.
Biochemical Journal,
2016
7.

Casero R.A., Woster P.M.
Journal of Medicinal Chemistry,
2009
8.

Nowotarski S.L., Woster P.M., Casero R.A.
Expert Reviews in Molecular Medicine,
2013
9.
Khomutov M.A., Hyvönen M.T., Simonian A.R., Weisell J., Vepsäläinen J., Alhonen L., Kochetkov S.N., Keinänen T.A., Khomutov A.R.
Mendeleev Communications,
2018
10.

Plyavnik N., Shtil A., Serebrennikova G.
Mini-Reviews in Medicinal Chemistry,
2006
11.

Mollinedo Faustino
Anti-Cancer Agents in Medicinal Chemistry
12.

Markova A.A., Plyavnik N.V., Tatarskii V.V., Shtil A.A., Serebrennikova G.A.
Russian Journal of Bioorganic Chemistry,
2010
13.

Markova A.A., Plyavnik N.V., Morozova N.G., Maslov M.A., Shtil A.A.
Russian Chemical Bulletin,
2014
14.

Kuksa V., Buchan R., Kong Thoo Lin P.
Synthesis,
2000
15.

Goldinga B.T., O'Sullivan M.C., Smith L.L.
Tetrahedron Letters,
1988
16.

Corcé V., Morin E., Guihéneuf S., Renault E., Renaud S., Cannie I., Tripier R., Lima L.M., Julienne K., Gouin S.G., Loréal O., Deniaud D., Gaboriau F.
Bioconjugate Chemistry,
2012
17.

Haussener T.J., Sebahar P.R., Kanna Reddy H.R., Williams D.L., Looper R.E.
Tetrahedron Letters,
2016
18.

Edwards M.L., Prakash N.J., Stemerick D.M., Sunkara S.P., Bitonti A.J., Davis G.F., Dumont J.A., Bey P.
Journal of Medicinal Chemistry,
1990
19.

Magoulas G.E., Tsigkou T., Skondra L., Lamprou M., Tsoukala P., Kokkinogouli V., Pantazaka E., Papaioannou D., Athanassopoulos C.M., Papadimitriou E.
Bioorganic and Medicinal Chemistry,
2017
20.

Panchenko S.P., Averin A.D., Lyakhovich M.S., Abel A.S., Maloshitskaya O.A., Beletskaya I.P.
Russian Chemical Bulletin,
2017
21.

Li M., Wang Y., Ge C., Chang L., Wang C., Tian Z., Wang S., Dai F., Zhao L., Xie S.
European Journal of Medicinal Chemistry,
2018
22.

Petukhov I.A., Maslov M.A., Morozova N.G., Serebrennikova G.A.
Russian Chemical Bulletin,
2010
23.

Puchkov P.A., Perevoshchikova K.A., Kartashova I.A., Luneva A.S., Kabilova T.O., Morozova N.G., Zenkova M.A., Maslov M.A.
Russian Journal of Bioorganic Chemistry,
2017
24.

Xu D., Prasad K., Repic O., Blacklock T.J.
Tetrahedron Letters,
1995
25.

Shmendel E.V., Perevoshchikova K.A., Shishova D.K., Kubasova T.S., Tyutyunnik L.L., Maslov M.A., Morozova N.G., Shtil A.A.
Russian Chemical Bulletin,
2015
26.

Linderoth L., Fristrup P., Hansen M., Melander F., Madsen R., Andresen T.L., Peters G.H.
Journal of the American Chemical Society,
2009
27.

Fukuyama T., Jow C., Cheung M.
Tetrahedron Letters,
1995
28.

Mosmann T.
Journal of Immunological Methods,
1983