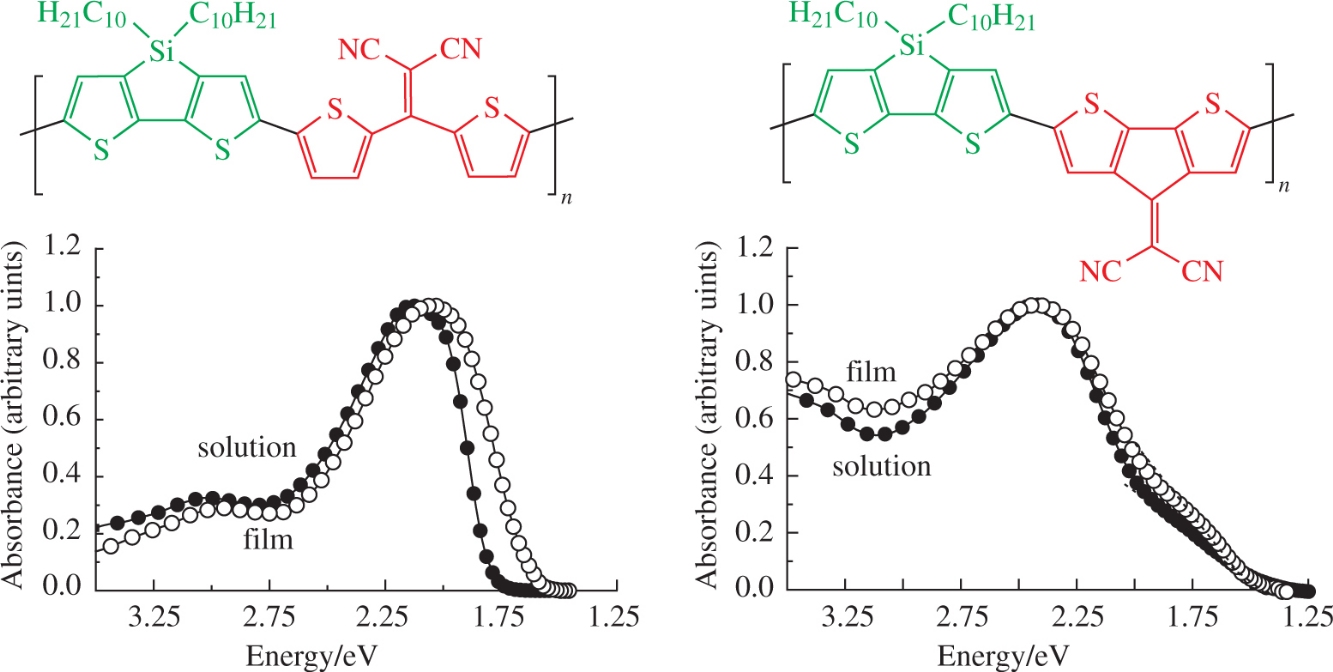
Abstract
Novel bifunctional acceptor monomer, 2-[bis(5-bromothiophen-2-yl)methylidene]malononitrile, is obtained in three steps in a total yield of 79%. The Stille cross-coupling between this monomer and 2,6-ditin derivative of 4,4-didecyl-4H-silolo[3,2-b: 4,5-b′]dithiophene afforded donor–acceptor polymer whose properties were compared to the analogue containing a planar cyclopenta[2,1-b: 3,4-b′]dithiophene acceptor block. The copolymers have low narrow optical band gaps and effectively absorb visible light, however the former possesses improved solubility and thermal stability as compared to the latter.
References
1.

Kim T., Lee K.
Macromolecular Rapid Communications,
2015
2.
Solodukhin A.N., Luponosov Y.N., Buzin M.I., Peregudova S.M., Svidchenko E.A., Ponomarenko S.A.
Mendeleev Communications,
2018
3.

Wu Y., Zhu W.
Chemical Society Reviews,
2013
4.

Jiang H., Wu Y., Islam A., Wu M., Zhang W., Shen C., Zhang H., Li E., Tian H., Zhu W.
ACS applied materials & interfaces,
2018
5.

Zhang S., Sun T., Xu Z., Li T., Li Y., Niu Q., Liu H.
Tetrahedron Letters,
2017
6.

Zhang S., Niu Q., Sun T., Li Y., Li T., Liu H.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2017
7.

Kumar C.V., Cabau L., Koukaras E.N., Sharma A., Sharma G.D., Palomares E.
Journal of Materials Chemistry A,
2015
8.
![Development of Spiro[cyclopenta[1,2-b:5,4-b′]dithiophene-4,9′-fluorene]-Based A-π-D-π-A Small Molecules with Different Acceptor Units for Efficient Organic Solar Cells](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Wang W., Shen P., Dong X., Weng C., Wang G., Bin H., Zhang J., Zhang Z., Li Y.
ACS applied materials & interfaces,
2017
9.

Hou M., Wang H., Miao Y., Xu H., Guo Z., Chen Z., Liao X., Li L., Li J., Guo K.
ACS Applied Energy Materials,
2018
10.
![D-A-D-A-D push pull organic small molecules based on 5,10-dihydroindolo[3,2-b]indole (DINI) central core donor for solution processed bulk heterojunction solar cells](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Sim J., Do K., Song K., Sharma A., Biswas S., Sharma G.D., Ko J.
Organic Electronics,
2016
11.

Patil Y., Misra R., Sharma A., Sharma G.D.
Physical Chemistry Chemical Physics,
2016
12.

Gautam P., Misra R., Siddiqui S.A., Sharma G.D.
ACS applied materials & interfaces,
2015
13.

Li C., Li M., Li Y., Shi Z., Li Z., Wang X., Sun J., Sun J., Zhang D., Cui Z.
Journal of Materials Chemistry C,
2016
14.

Wu Y., Huo J., Cao L., Ding S., Wang L., Cao D., Wang Z.
Sensors and Actuators, B: Chemical,
2016
15.

Starikova T.Y., Surin N.M., Borshchev O.V., Pisarev S.A., Svidchenko E.A., Fedorov Y.V., Ponomarenko S.A.
Journal of Materials Chemistry C,
2016
16.

Luo Z., Xiong W., Liu T., Cheng W., Wu K., Sun Y., Yang C.
Organic Electronics,
2017
17.

Luponosov Y.N., Solodukhin A.N., Mannanov A.L., Trukhanov V.A., Peregudova S.M., Pisarev S.A., Bakirov A.V., Shcherbina M.A., Chvalun S.N., Paraschuk D.Y., Ponomarenko S.A.
Organic Electronics,
2017
18.

Luponosov Y.N., Min J., Solodukhin A.N., Bakirov A.V., Dmitryakov P.V., Shcherbina M.A., Peregudova S.M., Cherkaev G.V., Chvalun S.N., Brabec C.J., Ponomarenko S.A.
Journal of Materials Chemistry C,
2016
19.

Mitchell V.D., Jones D.J.
Polymer Chemistry,
2018
20.

Nakabayashi K., Mori H.
Materials,
2014
21.

Liu D., Zhao W., Zhang S., Ye L., Zheng Z., Cui Y., Chen Y., Hou J.
Macromolecules,
2015
22.

Po R., Bianchi G., Carbonera C., Pellegrino A.
Macromolecules,
2015
23.

Leclerc N., Chávez P., Ibraikulov O., Heiser T., Lévêque P.
Polymers,
2016
24.

Tan S.E., Sarjadi M.S.
Polymer Science - Series B,
2017
25.

Deshmukh K.D., Matsidik R., Prasad S.K., Chandrasekaran N., Welford A., Connal L.A., Liu A.C., Gann E., Thomsen L., Kabra D., Hodgkiss J.M., Sommer M., McNeill C.R.
ACS applied materials & interfaces,
2017
26.

Revoju S., Biswas S., Eliasson B., Sharma G.D.
Dyes and Pigments,
2018
27.

Lee K., Lee H., Morino K., Sudo A., Endo T.
Journal of Polymer Science, Part A: Polymer Chemistry,
2011
28.
![4H-cyclopenta[2,1-b:3,4-b′]dithiophen-4-one (CPDTO) homopolymer with side chains on every other CPDTO](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Sun J., Venkatesan S., Dubey A., Qiao Q., Zhang C.
Journal of Polymer Science, Part A: Polymer Chemistry,
2017
29.

Brisset H., Thobie-Gautier C., Jubault M., Gorgues A., Roncali J.
Journal of the Chemical Society Chemical Communications,
1994
30.

Shi J., Zhao W., Xu L., Kan Y., Li C., Song J., Wang H.
Journal of Physical Chemistry C,
2014
31.
![The Use of Cyclopenta[2,1‐b;3,4‐b′]dithiophene Analogues for the Development of Low‐Bandgap Materials](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Willot P., De Cremer L., Koeckelberghs G.
Macromolecular Chemistry and Physics,
2012
32.
![Low band gap dithieno[3,2-b:2',3'-d]silole-containing polymers, synthesis, characterization and photovoltaic application.](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Huo L., Chen H., Hou J., Chen T.L., Yang Y.
Chemical Communications,
2009