Abstract
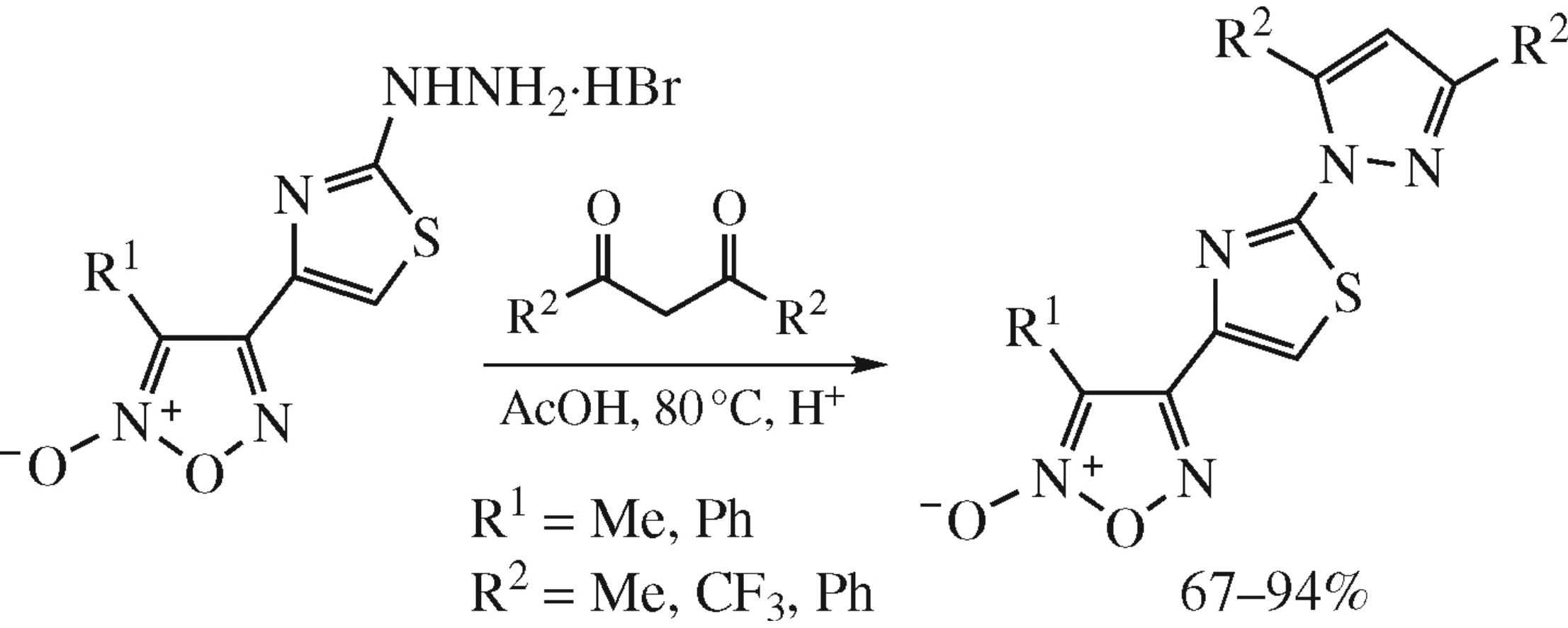
The effective synthesis of pharmacologically oriented heterocyclic ensembles, [2-(1H-pyrazol-1-yl)thiazol-4-yl]furoxans, comprising furoxan moiety as NO-donor and pharmacophoric pyrazolylthiazole fragment is based on the condensation of (2-hydrazinylthiazol-4-yl)furoxan hydrobromides with linear 1,3-diketones. The reaction proceeds through hydroxypyrazoline intermediate.
References
1.

Vitaku E., Smith D.T., Njardarson J.T.
Journal of Medicinal Chemistry,
2014
2.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
3.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
4.
10.1016/j.mencom.2019.05.015_sbref0010a
Nikonov
2008
5.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
6.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
7.
Larin A.A., Fershtat L.L., Ustyuzhanina N.E., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
8.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
9.

Makhova N.N., Fershtat L.L.
Tetrahedron Letters,
2018
10.

Fershtat L.L., Larin A.A., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016
11.

Fershtat L.L., Epishina M.A., Ovchinnikov I.V., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016
12.
![Versatile approach to heteroarylfuroxan derivatives from oximinofuroxans via a one-pot, nitration/thermolysis/[3+2]-cycloaddition cascade](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Larin A.A., Fershtat L.L., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2017
13.

Fershtat L.L., Larin A.A., Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Ananyev I.V., Makhova N.N.
RSC Advances,
2016
14.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
15.

Sanfilippo P.J., Andrade-Gordon P., Urbanski M.J., Beers K.N., Eckardt A., Falotico R., Ginsberg M.H., Offord S., Press J.B.
Journal of Medicinal Chemistry,
1995
16.
Prokopp C.R., Rubin M.A., Sauzem P.D., de Souza A.H., Berlese D.B., Lourega R.V., Muniz M.N., Bonacorso H.G., Zanatta N., Martins M.A., Mello C.F.
Brazilian Journal of Medical and Biological Research,
2006
17.

Aggarwal R., Kumar R., Kumar S., Garg G., Mahajan R., Sharma J.
Journal of Fluorine Chemistry,
2011
18.

Kumar S., Aggarwal R., Sharma C.
Synthetic Communications,
2015
19.

Donohue B.A., Michelotti E.L., Reader J.C., Reader V., Stirling M., Tice C.M.
Journal of Combinatorial Chemistry,
2001
20.
Kulikov A.S., Epishina M.A., Churakov A.I., Anikina L.V., Fershtat L.L., Makhova N.N.
Mendeleev Communications,
2018
21.

Mahajan M., Sondhi S., Ralhan N.
Australian Journal of Chemistry,
1977
22.
10.1016/j.mencom.2019.05.015_bib0060
Alaka
J. Indian Chem. Soc.,
1982
23.

Sobiesiak M., Muzioł T., Rozalski M., Krajewska U., Budzisz E.
New Journal of Chemistry,
2014
24.
10.1016/j.mencom.2019.05.015_bib0070
Singh
Indian J. Chem., Sect. B,
1995
25.

Sharma P.K., Chandna N., Kumar S., Kumar P., Kumar S., Kaushik P., Kaushik D.
Medicinal Chemistry Research,
2011
26.
10.1016/j.mencom.2019.05.015_bib0080
Ponzio
Gazz. Chim. Ital.,
1937
27.
10.1016/j.mencom.2019.05.015_bib0085
The Quantum Theory of Atoms in Molecules: From Solid State to DNA and Drug Design,
2007
28.

Romanova A., Lyssenko K., Ananyev I.
Journal of Computational Chemistry,
2018
29.

Blanco M.A., Martín Pendás A., Francisco E.
Journal of Chemical Theory and Computation,
2005
30.
10.1016/j.mencom.2019.05.015_bib0100
Groom
Acta Crystallogr.,
2016
31.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
32.
10.1016/j.mencom.2019.05.015_sbref0105b
Sheldrick
Acta Crystallogr.,
2015