Abstract
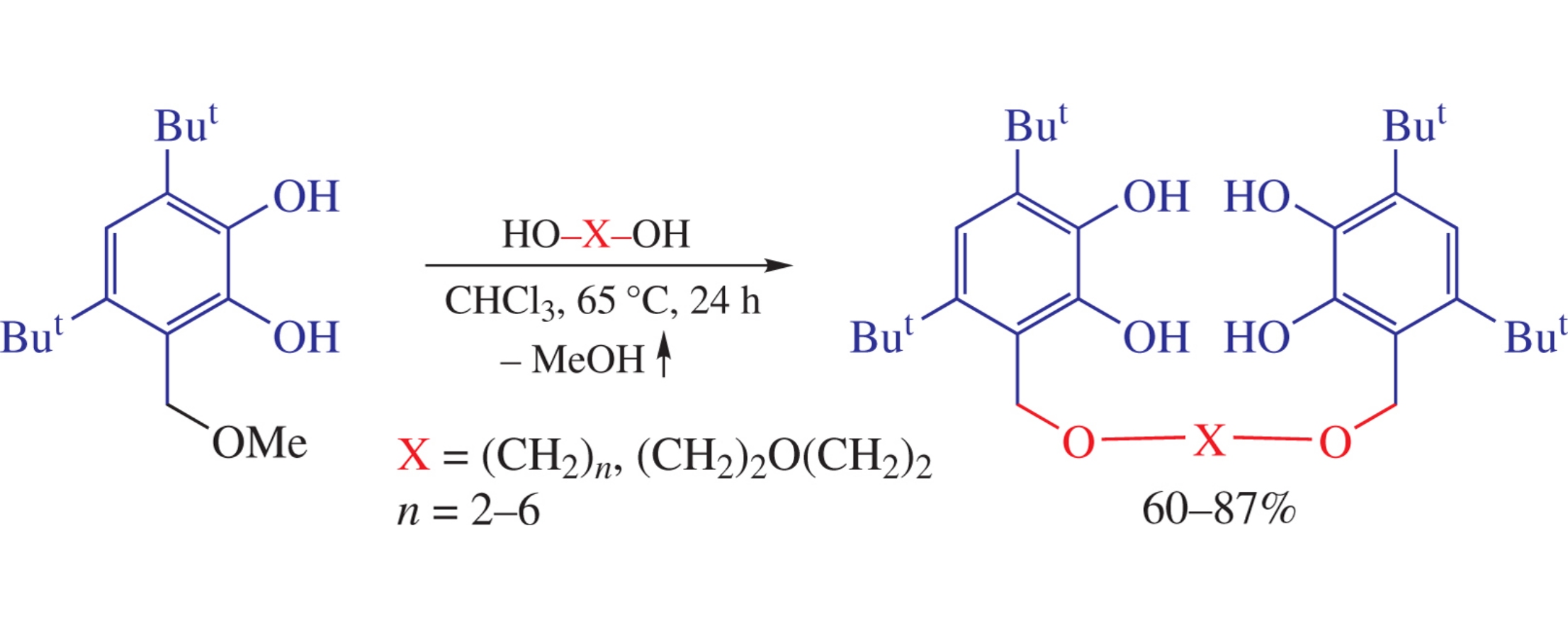
The trans-etherification of 4,6-di-tert-butyl-2,3-dihydroxybenzyl methyl ether with α,ω-alkanediols under mild conditions (CHCl3, 65°C) leads to new sterically hindered biscatechols in high yields. The molecular structures of three of them was determined by single-crystal X-ray diffraction.
References
1.

Thulasiraman P., Newton S.M., Xu J., Raymond K.N., Mai C., Hall A., Montague M.A., Klebba P.E.
Journal of Bacteriology,
1998
2.

Durbin P.W., Kullgren B., Ebbe S.N., Xu J., Raymond K.N.
Health Physics,
2003
3.

Sprencel C., Cao Z., Qi Z., Scott D.C., Montague M.A., Ivanoff N., Xu J., Raymond K.M., Newton S.M., Klebba P.E.
Journal of Bacteriology,
2000
4.

Ni C., Shuh D.K., Raymond K.N.
Chemical Communications,
2011
5.

Pham T.A., Xu J., Raymond K.N.
Journal of the American Chemical Society,
2014
6.
![Triaryl- and trialkylantimony(V) Bis(catecholates) based on 1,1′-spirobis[3,3-dimethylindanequinone-5,6]: Spectroscopic and electrochemical studies](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Poddel’sky A.I., Smolyaninov I.V., Vavilina N.N., Kurskii Y.A., Berberova N.T., Cherkasov V.K., Abakumov G.A.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2012
7.

Arsen’ev M.V., Okhlopkova L.S., Poddel’skii A.I., Fukin G.K.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2018
8.

Abakumov G.A., Piskunov A.V., Cherkasov V.K., Fedushkin I.L., Ananikov V.P., Eremin D.B., Gordeev E.G., Beletskaya I.P., Averin A.D., Bochkarev M.N., Trifonov A.A., Dzhemilev U.M., D'yakonov V.A., Egorov M.P., Vereshchagin A.N., et. al.
Russian Chemical Reviews,
2018
9.

Albrecht M.
Chemistry - A European Journal,
2000
10.

Albrecht M., Janser I., Fröhlich R.
Chemical Communications,
2005
11.

Janser I., Albrecht M., Hunger K., Burk S., Rissanen K.
European Journal of Inorganic Chemistry,
2005
12.

Albrecht M., Shang Y., Hasui K., Gossen V., Raabe G., Tahara K., Tobe Y.
Dalton Transactions,
2012
13.

Metelitza D.I., Karasyova E.I.
Applied Biochemistry and Microbiology,
2007
14.

Perron N.R., Brumaghim J.L.
Cell Biochemistry and Biophysics,
2009
15.

Zhao J., Lu H., Cao J., Jiang Y., Chen C.
Tetrahedron Letters,
2009
16.

Loughrey J.J., Sproules S., McInnes E.J., Hardie M.J., Halcrow M.A.
Chemistry - A European Journal,
2014
17.

Smolyaninov I.V., Poddel’skiy A.I., Berberova N.T., Cherkasov V.K., Abakumov G.A.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2010
18.

Poddel'sky A.I., Cherkasov V.K., Abakumov G.A.
Coordination Chemistry Reviews,
2009
19.

Pierpont C.
Coordination Chemistry Reviews,
2001
20.

Sato O., Tao J., Zhang Y.
Angewandte Chemie - International Edition,
2007
21.

Chesnokov S.A., Cherkasov V.K., Abakumov G.A., Mamysheva O.N., Chechet Y.V., Nevodchikov V.I.
Russian Chemical Bulletin,
2001
22.

Chesnokov S.A., Lenshina N.A., Arsenyev M.V., Kovylin R.S., Baten'kin M.A., Poddel'sky A.I., Abakumov G.A.
Applied Organometallic Chemistry,
2016
23.

Suenaga Y., Hirano Y., Umehata Y., Minematsu T.
Inorganica Chimica Acta,
2011
24.

Albrecht M., Stortz P., Runsink J., Weis P.
Chemistry - A European Journal,
2004
25.

Albrecht M., Mirtschin S., de Groot M., Janser I., Runsink J., Raabe G., Kogej M., Schalley C.A., Fröhlich R.
Journal of the American Chemical Society,
2005
26.
![6,6'-[piperazine-1,4-diylbis(methylene)]bis[3,5-di(tert-butyl)-1,2- benzoquinone]: Synthesis and properties](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Sayapin Y.A., Tupaeva I.O., Tkachev V.V., Shilov G.V.
Russian Journal of Organic Chemistry,
2016
27.
Arsenyev M.V., Baranov E.V., Fedorov A.Y., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2015
28.

Arsenyev M.V., Baranov E.V., Chesnokov S.A., Cherkasov V.K., Abakumov G.A.
Russian Chemical Bulletin,
2013
29.
Arsenyev M.V., Astafeva T.V., Baranov E.V., Poddelsky A.I., Chesnokov S.A.
Mendeleev Communications,
2018
30.

Poddel'sky A.I., Arsenyev M.V., Astaf'eva T.V., Chesnokov S.A., Fukin G.K., Abakumov G.A.
Journal of Organometallic Chemistry,
2017
31.
Arsenyev M.V., Baranov E.V., Shurygina M.P., Chesnokov S.A., Abakumov G.A.
Mendeleev Communications,
2016
32.

Osipov D.V., Osyanin V.A., Klimochkin Y.N.
Russian Chemical Reviews,
2017
33.

Poddel'sky A.I., Astaf'eva T.V., Smolyaninov I.V., Arsenyev M.A., Fukin G.K., Berberova N.T., Cherkasov V.K., Abakumov G.A.
Journal of Organometallic Chemistry,
2018
34.
SAINT. Data Reduction and Correction Program, v. 8.37A, Bruker AXS, Madison, WI, USA, 2012.
35.
Data Collection, Reduction and Correction Program CrysAlisPro – Software Package, Agilent Technologies, Yarnton, UK, 2012.
36.
SADABS. Bruker/Siemens Area Detector Absorption Correction Program, v. 2016/2, Bruker AXS, Madison, WI, USA, 2016.
37.
SCALE3 ABSPACK: Empirical Absorption Correction, CrysAlisPro – Software Package, Agilent Technologies, Yarnton, UK, 2012.
38.
10.1016/j.mencom.2019.01.031_bib0095
Sheldrick
Acta Crystallogr.,
2015
39.
10.1016/j.mencom.2019.01.031_bib0100
Sheldrick
SHELXTL. Structure Determination Software Suite, v. 6.14,
2003