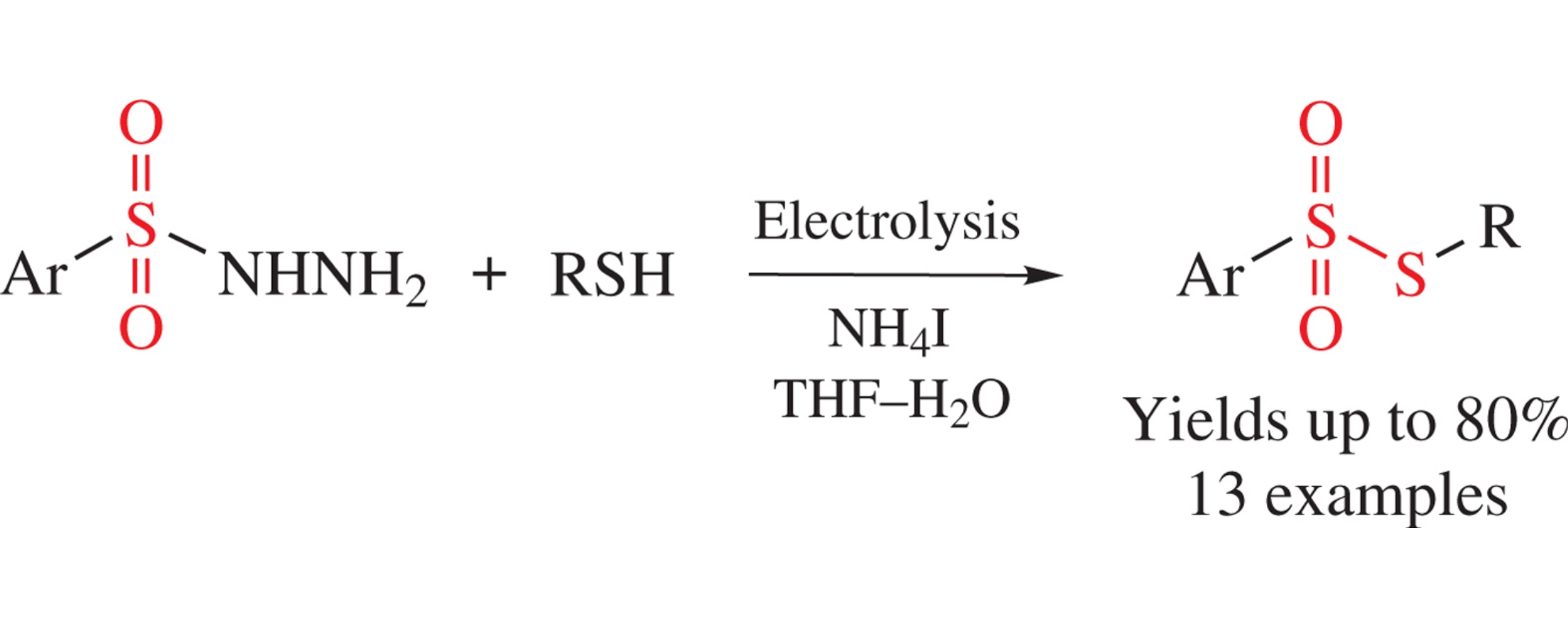
Abstract
Unsymmetrical thiosulfonates were synthesized from thiols and arenesulfonohydrazides by their electrolysis in undivided cell equipped with graphite anode and stainless steel cathode under high current density applying NH4I both as a redox catalyst and a supporting electrolyte. In the course of the process, oxidative formation of S–S bond occurs with the loss of hydrazine moiety.
References
1.

Mahieu J., Gosselet M., Sebille B., Beuzard Y.
Synthetic Communications,
1986
2.

Pham H.T., Nguyen N.T., Duus F., Luu T.X.
Phosphorus, Sulfur and Silicon and the Related Elements,
2015
3.
10.1016/j.mencom.2019.01.027_sbref0010a
Lei
Oxidative Cross-Coupling Reactions,
2016
4.

Zhang T., Wang N., Xing Y.
Journal of Organic Chemistry,
2018
5.

Liu Y., Yi H., Lei A.
Chinese Journal of Chemistry,
2018
6.

Antonchick A., Murarka S.
Synthesis,
2018
7.

Zhang H., Lei A.
Asian Journal of Organic Chemistry,
2018
8.

Mulina O.M., Ilovaisky A.I., Terent'ev A.O.
European Journal of Organic Chemistry,
2018
9.

Liang S., Xu K., Zeng C., Tian H., Sun B.
Advanced Synthesis and Catalysis,
2018
10.

Fujiki K., Tanifuji N., Sasaki Y., Yokoyama T.
Synthesis,
2002
11.

Peng Z., Zheng X., Zhang Y., An D., Dong W.
Green Chemistry,
2018
12.

Liang G., Liu M., Chen J., Ding J., Gao W., Wu H.
Chinese Journal of Chemistry,
2012
13.

Yadav L., Keshari T., Kapoorr R.
Synlett,
2016
14.

Taniguchi N.
European Journal of Organic Chemistry,
2014
15.

Taniguchi N.
Journal of Organic Chemistry,
2015
16.

Zhang G., Lv S., Shoberu A., Zou J.
Journal of Organic Chemistry,
2017
17.

Chen Q., Huang Y., Wang X., Wu J., Yu G.
Organic and Biomolecular Chemistry,
2018
18.

Cao L., Luo S., Jiang K., Hao Z., Wang B., Pang C., Wang Z.
Organic Letters,
2018
19.

Jiang Y., Xu K., Zeng C.
Chemical Reviews,
2017
20.

Möhle S., Zirbes M., Rodrigo E., Gieshoff T., Wiebe A., Waldvogel S.R.
Angewandte Chemie - International Edition,
2018
21.

Liu K., Song C., Lei A.
Organic and Biomolecular Chemistry,
2018
22.

Pletcher D., Green R.A., Brown R.C.
Chemical Reviews,
2017
23.

Wiebe A., Gieshoff T., Möhle S., Rodrigo E., Zirbes M., Waldvogel S.R.
Angewandte Chemie - International Edition,
2018
24.

Cardoso D.S., Šljukić B., Santos D.M., Sequeira C.A.
Organic Process Research and Development,
2017
25.

Yan M., Kawamata Y., Baran P.S.
Chemical Reviews,
2017
26.
Mendkovich A.S., Syroeshkin M.A., Mitina K.R., Mikhailov M.N., Gultyai V.P., Pechennikov V.M.
Mendeleev Communications,
2017
27.

Yuan Y., Cao Y., Lin Y., Li Y., Huang Z., Lei A.
ACS Catalysis,
2018
28.

Zhao H., Xu P., Song J., Xu H.
Angewandte Chemie - International Edition,
2018
29.
Lyalin B.V., Sigacheva V.L., Fershtat L.L., Makhova N.N., Petrosyan V.A.
Mendeleev Communications,
2018
30.

Luo S., Li L., Yang Q., Jia Z.
Synthesis,
2018
31.
Terent’ev A.O., Mulina O.M., Pirgach D.A., Syroeshkin M.A., Glinushkin A.P., Nikishin G.I.
Mendeleev Communications,
2016
32.

Terent'ev A.O., Mulina O.M., Pirgach D.A., Ilovaisky A.I., Syroeshkin M.A., Kapustina N.I., Nikishin G.I.
Tetrahedron,
2017
33.
O. M. Mulina and A. O. Terent’ev, in Proceedings of the International Conference ‘Frontiers in Chemistry’, Armenia, 2018, p. PS 073.
34.

Pan X., Gao J., Yuan G.
Tetrahedron,
2015
35.

Huang H., Yuan G., Li X., Jiang H.
Tetrahedron Letters,
2013
36.

Gao X., Yuan G., Chen H., Jiang H., Li Y., Qi C.
Electrochemistry Communications,
2013
37.

Yuan G., Zhu Z., Gao X., Jiang H.
RSC Advances,
2014
38.

Qu Q., Gao X., Gao J., Yuan G.
Science China Chemistry,
2015
39.

Liang S., Zeng C., Luo X., Ren F., Tian H., Sun B., Little R.D.
Green Chemistry,
2016
40.

Luo Y., Pan X., Yuan G.
Tetrahedron,
2015
41.

Emmanuvel L., Shukla R.K., Sudalai A., Gurunath S., Sivaram S.
Tetrahedron Letters,
2006
42.

Li W., Zeng C., Hu L., Tian H., Little R.D.
Advanced Synthesis and Catalysis,
2013
43.
C. L. Mehltretter, US Patent 2770589A, 1956.
44.
C. L. Mehltretter, US Patent 2830941A, 1958.
45.

Aiya Y., Fujii S., Sugino K., Shirai K.
Journal of the Electrochemical Society,
2007
46.

Yamada S., Morizono D., Yamamoto K.
Tetrahedron Letters,
1992
47.

Lavekar A.G., Equbal D., Saima, Sinha A.K.
Advanced Synthesis and Catalysis,
2017
48.

Alabugin I.V., Timokhin V.I., Abrams J.N., Manoharan M., Abrams R., Ghiviriga I.
Journal of the American Chemical Society,
2008
49.

Gilmore K., Gold B., Clark R.J., Alabugin I.V.
Australian Journal of Chemistry,
2013
50.

Guo Z., Wei W., Zhou G., Xu X., Chen G.
Synlett,
2018
51.

Do Q.T., Elothmani D., Le Guillanton G., Simonet J.
Tetrahedron Letters,
1997
52.

53.

Hirano M., Yakabe S., Ando K., Morimoto T.
Journal of Chemical Research,
1998
54.

Zeynizadeh B.
Journal of Chemical Research,
2002
55.

Dreyer D.R., Jia H., Todd A.D., Geng J., Bielawski C.W.
Organic and Biomolecular Chemistry,
2011