Abstract
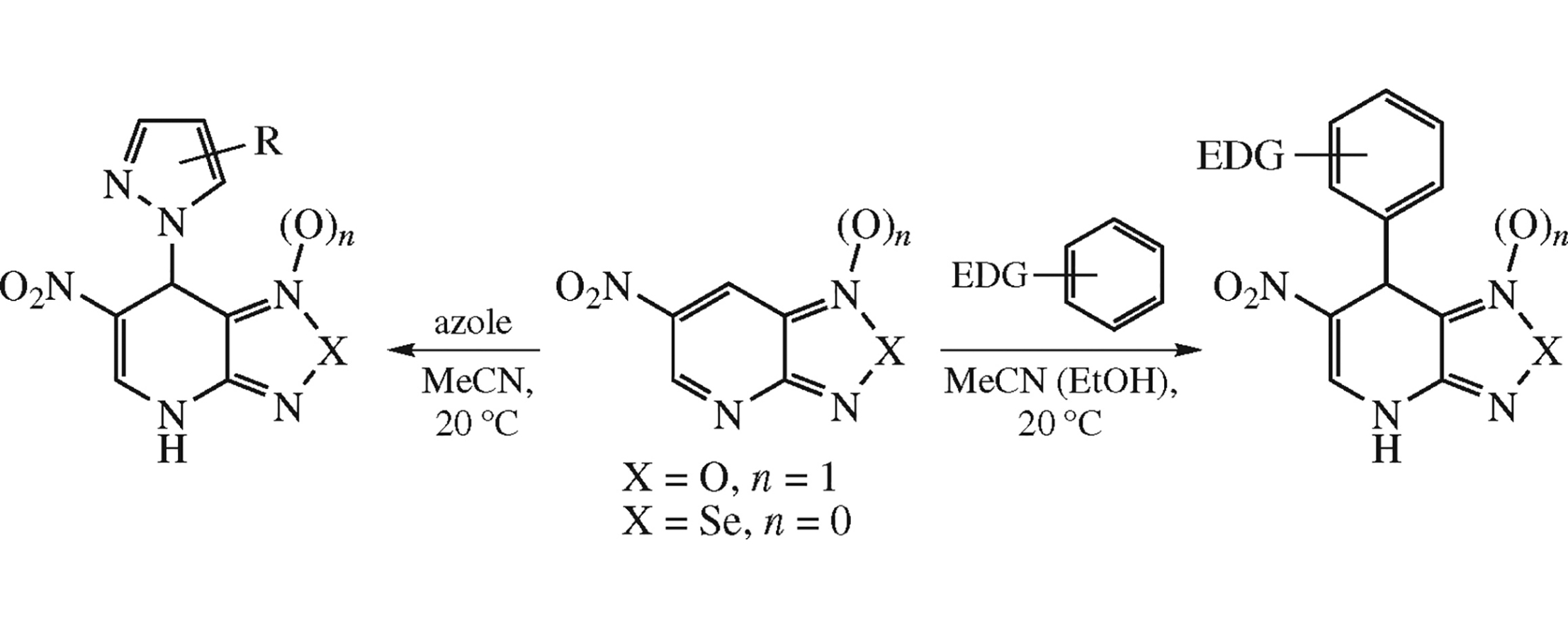
Highly electrophilic 6-nitro-4-azabenzofuroxan and 6-nitro-4-azabenzo[1,2,5]selenadiazole add π-excessive (het)arenes and other neutral nucleophiles at 7-position to give C–C and N–C-bonded adducts, 1,4-dihydropyridines fused with furoxan or selenadiazole ring.
References
1.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
2.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
3.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
4.
![Synthesis of novel polycyclic heterosystems from 5-nitro[1,2,5]selenadiazolo[3,4-e]benzofuroxans](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Pavlov A.A., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2016
5.
Starosotnikov A.M., Bastrakov M.A., Pavlov A.A., Fedyanin I.V., Dalinger I.L., Shevelev S.A.
Mendeleev Communications,
2016
6.
![3-R-4-Nitro-6,7-furoxanobenzo[d]isoxazoles – a new type of condensed nitroarenes capable of Diels–Alder reaction](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bastrakov M.A., Starosotnikov A.M., Kachala V.V., Dalinger I.L., Shevelev S.A.
Chemistry of Heterocyclic Compounds,
2015
7.
10.1016/j.mencom.2018.11.025_sbrefe0020a
Nitric Oxide Donors: For Pharmaceutical and Biological Applications,
2005
8.

Miller M.R., Megson I.L.
British Journal of Pharmacology,
2007
9.

Granik V.G., Ryabova S.Y., Grigoriev N.B.
Russian Chemical Reviews,
1997
10.

Feelisch M.
Journal of Cardiovascular Pharmacology,
2008
11.

12.

Feelisch M., Schönafingeri K., Noack H.
Biochemical Pharmacology,
1992
13.

Medana C., Ermondi G., Fruttero R., Di Stilo A., Ferretti C., Gasco A.
Journal of Medicinal Chemistry,
1994
14.
Fershtat L.L., Epishina M.A., Kulikov A.S., Makhova N.N.
Mendeleev Communications,
2015
15.

Fershtat L.L., Epishina M.A., Kulikov A.S., Struchkova M.I., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
16.

Fershtat L.L., Larin A.A., Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Ananyev I.V., Makhova N.N.
RSC Advances,
2016
17.

Fershtat L.L., Epishina M.A., Ovchinnikov I.V., Kachala V.V., Makhova N.N.
Chemistry of Heterocyclic Compounds,
2015
18.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
19.

Starosotnikov A.M., Shkaev D.V., Bastrakov M.A., Fedyanin I.V., Shevelev S.A., Dalinger I.L.
Beilstein Journal of Organic Chemistry,
2017
20.
![Study of the reactions of 3-chloro-4-cyanobenzo[b][1,6]naphthyridine with nucleophilic reagents](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Ivanov A.S., Tugusheva N.Z., Solov"eva N.P., Granik V.G.
Russian Chemical Bulletin,
2002
21.

Cai S.X., Huang J., Espitia S.A., Tran M., Ilyin V.I., Hawkinson J.E., Woodward R.M., Weber E., Keana J.F.
Journal of Medicinal Chemistry,
1997
22.

Shealy Y.F., Clayton J.D., Dixon G.J., Dulmadge E.A., Pittillo R.F., Hunt D.E.
Biochemical Pharmacology,
1966
23.
10.1016/j.mencom.2018.11.025_bibe0045
Huang
Chem. J. Chin. Univ.,
2012
24.

Plano D., Moreno E., Font M., Encío I., Palop J.A., Sanmartín C.
Archiv der Pharmazie,
2010
25.

Tan H., Zhao Z., Ma Z., Yan H.
Tetrahedron,
2018
26.

OHSUMI K., OHISHI K., MORINAGA Y., NAKAGAWA R., SUGA Y., SEKIYAMA T., AKIYAMA Y., TSUJI T., TSURUO T.
Chemical and Pharmaceutical Bulletin,
2011
27.

Goba I., Turovska B., Belyakov S., Liepinsh E.
Journal of Molecular Structure,
2014
28.

Guanaes L.D., Ducatti D.R., Duarte M.E., Barreira S.M., Noseda M.D., Gonçalves A.G.
Tetrahedron Letters,
2015
29.

Zhang B., Cui Y., Jiao N.
Chemical Communications,
2012
30.

Pintér Á., Sud A., Sureshkumar D., Klussmann M.
Angewandte Chemie - International Edition,
2010
31.
10.1016/j.mencom.2018.11.025_bibe0070
Sheldrick
Acta Crystallogr.,
2005
32.
10.1016/j.mencom.2018.11.025_bibe0075
Sheldrick
C71,
2005