Abstract
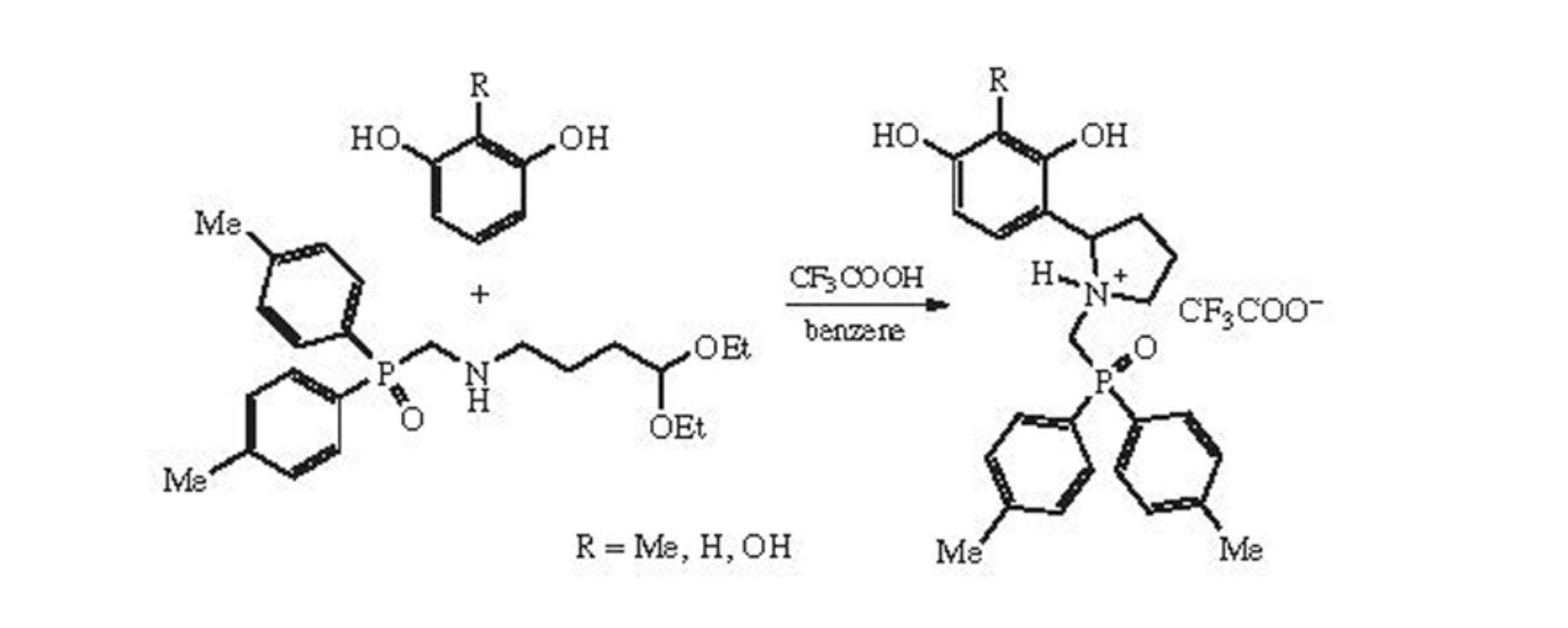
2-Aryl-substituted pyrrolidines containing phosphine oxide group have been obtained by the reaction of P-(4,4-diethoxybutylaminomethyl)-P,P-di-p-tolylphosphine oxide with polyatomic phenols.
References
1.

Martin O.
Annales Pharmaceutiques Francaises,
2007
2.

Banerjee S., Hussain M., Wang Z., Saliganan A., Che M., Bonfil D., Cher M., Sarkar F.H.
Cancer Research,
2007
3.

Johari S.A., Mohtar M., Syed Mohammad S.A., Sahdan R., Shaameri Z., Hamzah A.S., Mohammat M.F.
BioMed Research International,
2015
4.

Hensler M.E., Bernstein G., Nizet V., Nefzi A.
Bioorganic and Medicinal Chemistry Letters,
2006
5.
Sukhanova A.A., Nelyubina Y.V., Zlotin S.G.
Mendeleev Communications,
2016
6.

Coelho F., da Silva E., Yamakawa N., Dos Santos A.
Synthesis,
2017
7.
Khasiyatullina N.R., Vazykhova A.M., Mironov V.F., Krivolapov D.B., Voronina Y.K., Voloshina A.D., Kulik N.V., Strobykina A.S.
Mendeleev Communications,
2017
8.

Tatarinov D.A., Kuznetsov D.M., Voloshina A.D., Lyubina A.P., Strobykina A.S., Mukhitova F.K., Polyancev F.M., Mironov V.F.
Tetrahedron,
2016
9.
D. Haebich, J. Hansen and A. Paessens, EP0472077, 1992.
10.

Kočalka P., Rejman D., Vaněk V., Rinnová M., Tomečková I., Králíková Š., Petrová M., Páv O., Pohl R., Buděšínský M., Liboska R., Točík Z., Panova N., Votruba I., Rosenberg I., et. al.
Bioorganic and Medicinal Chemistry Letters,
2010
11.

Rejman D., Panova N., Klener P., Maswabi B., Pohl R., Rosenberg I.
Journal of Medicinal Chemistry,
2012
12.

Keough D.T., Hocková D., Rejman D., Špaček P., Vrbková S., Krečmerová M., Eng W.S., Jans H., West N.P., Naesens L.M., de Jersey J., Guddat L.W.
Journal of Medicinal Chemistry,
2013
13.

Şener Ş., Mete A.
Synthetic Communications,
1997
14.

Koizumi T., Kobayashi Y., Amitani H., Yoshii E.
Journal of Organic Chemistry,
1977
15.

Ma X., Zhao Y.
Journal of Organic Chemistry,
1989
16.

Nakayama K., Thompson W.J.
Journal of the American Chemical Society,
1990
17.
10.1016/j.mencom.2018.07.019_sbref0065a
Kowalczyk
Synthesis,
2017
18.
10.1016/j.mencom.2018.07.019_sbref0065b
Subotkowski
Pol. J. Chem,
1980
19.

Rejman D., Pohl R., Kočalka P., Masojídková M., Rosenberg I.
Tetrahedron,
2009
20.

Belyaev A., Borloo M., Augustyns K., Lambeir A., De Meester I., Scharpe S., Blaton N., Peeters O.M., De Ranter C., Haemers A.
Tetrahedron Letters,
1995
21.

Belyaev A., Zhang X., Augustyns K., Lambeir A., De Meester I., Vedernikova I., Scharpé S., Haemers A.
Journal of Medicinal Chemistry,
1999
22.

Frejaville C., Karoui H., Tuccio B., le Moigne F., Culcasi M., Pietri S., Lauricella R., Tordo P.
Journal of the Chemical Society Chemical Communications,
1994
23.

van Assche I., Soroka M., Haemers A., Hooper M., Blanot D., van Heijenoort J.
European Journal of Medicinal Chemistry,
1991
24.

Kaboudin B., Karami L., Kato J., Aoyama H., Yokomatsu T.
Tetrahedron Letters,
2013
25.

Martirosyan A.H., Gasparyan S.P., Alexanyan M.V., Harutyunyan G.K., Panosyan H.A., Schinazi R.F.
Medicinal Chemistry Research,
2016
26.
Matyugina E.S., Valuev-Elliston V.T., Chizhov A.O., Kochetkov S.N., Khandazhinskaya A.L.
Mendeleev Communications,
2016
27.
Gaman M.S., Matyugina E.S., Novikov M.S., Babkov D.A., Solyev P.N., Kochetkov S.N., Khandazhinskaya A.L.
Mendeleev Communications,
2017
28.

Smolobochkin A.V., Gazizov A.S., Vagapova L.I., Burilov A.R., Pudovik M.A.
Russian Chemical Bulletin,
2014
29.

Gazizov A.S., Kharitonova N.I., Smolobochkin A.V., Syakaev V.V., Burilov A.R., Pudovik M.A.
Monatshefte fur Chemie,
2015
30.

Vagapova L.I., Amirova L.R., Burilov A.R., Garifzyanov A.R., Pudovik M.A., Kharlampidi K.E.
Russian Journal of Organic Chemistry,
2014