Abstract
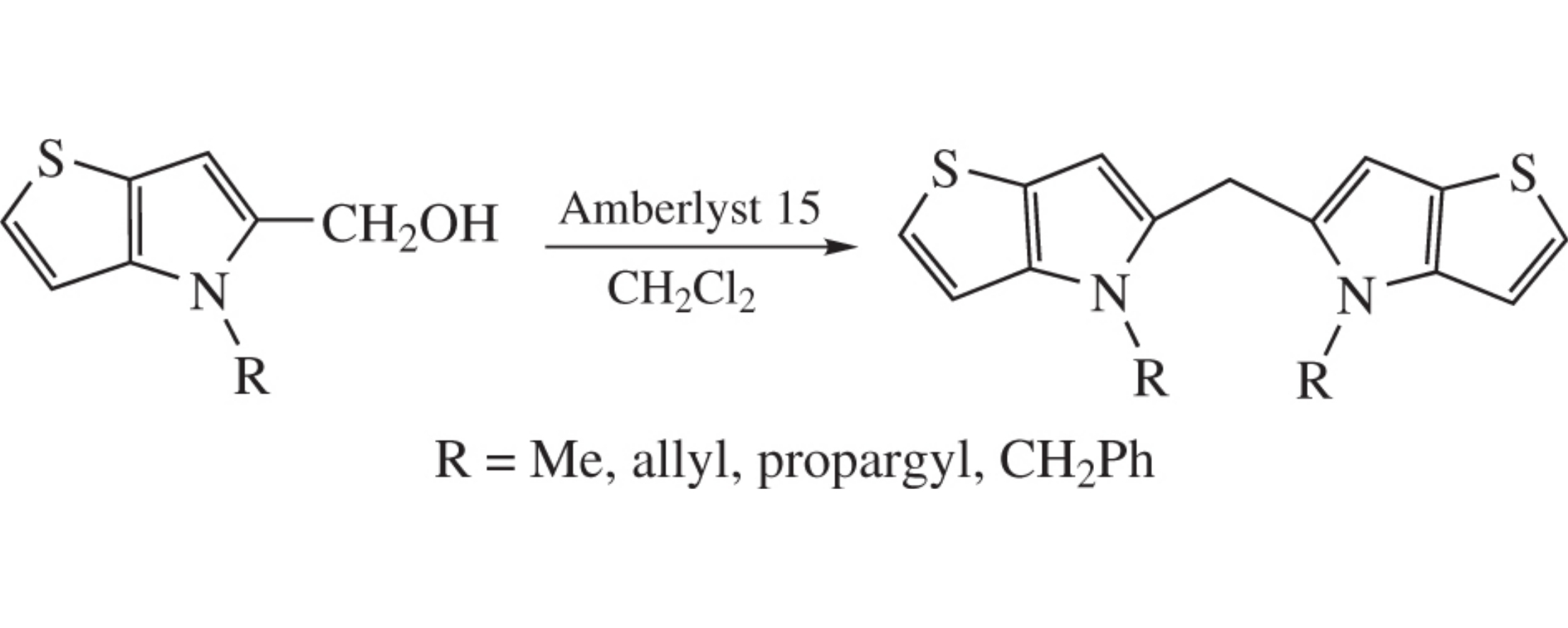
N-Substituted (4H-thieno[3,2-b]pyrrol-5-yl)methanols were obtained by alkylation of methyl 4H-thieno[3,2-b]pyrrole-5-carboxylate followed by reduction with LiAlH4. These compounds on contact with Amberlyst 15 (H-form) in CH2Cl2 undergo self-condensation to produce bis(4H-thieno[3,2-b]-pyrrol-5-yl)methanes.
References
1.

Hemetsberger H., Knittel D.
Monatshefte fur Chemie,
1972
2.
![Regioselective acylation of methyl 2-methyl-4H-thieno[3,2-b]pyrrole-5-carboxylate](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Yarovenko V.N.
Russian Chemical Bulletin,
2003
3.
![Trisubstituted Thieno[3,2-b]pyrrole 5-Carboxamides as Potent Inhibitors of Alphaviruses](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ching K., Kam Y., Merits A., Ng L.F., Chai C.L.
Journal of Medicinal Chemistry,
2015
4.
![Structural Optimizations of Thieno[3,2-b]pyrrole Derivatives for the Development of Metabolically Stable Inhibitors of Chikungunya Virus](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ching K., Tran T.N., Amrun S.N., Kam Y., Ng L.F., Chai C.L.
Journal of Medicinal Chemistry,
2017
5.
![Thieno[3,2-b]pyrrole-5-carboxamides as New Reversible Inhibitors of Histone Lysine Demethylase KDM1A/LSD1. Part 1: High-Throughput Screening and Preliminary Exploration](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sartori L., Mercurio C., Amigoni F., Cappa A., Fagá G., Fattori R., Legnaghi E., Ciossani G., Mattevi A., Meroni G., Moretti L., Cecatiello V., Pasqualato S., Romussi A., Thaler F., et. al.
Journal of Medicinal Chemistry,
2017
6.
10.1016/j.mencom.2018.03.028_bib0030
Ashok
Indian J. Heterocycl. Chem.,
2010
7.
![Synthesis of Heterocyclic Compounds Possessing the 4H-Thieno[3,2-b]Pyrrole Moiety](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Ilyin A.P., Dmitrieva I.G., Kustova V.A., Manaev A.V., Ivachtchenko A.V.
Journal of Combinatorial Chemistry,
2006
8.

Sindac J.A., Yestrepsky B.D., Barraza S.J., Bolduc K.L., Blakely P.K., Keep R.F., Irani D.N., Miller D.J., Larsen S.D.
Journal of Medicinal Chemistry,
2012
9.

Verbitskiy E.V., Slepukhin P.A., Valova M.S., Cheprakova E.M., Schepochkin A.V., Rusinov G.L., Charushin V.N.
European Journal of Organic Chemistry,
2014
10.

Krayushkin M.M., Yarovenko V.N., Semenov S.L., Zavarzin I.V., Ignatenko A.V., Martynkin A.Y., Uzhinov B.M.
Organic Letters,
2002
11.

Synthesis and Properties of Semiconducting Bispyrrolothiophenes for Organic Field-Effect Transistors
Jones C., Boudinet D., Xia Y., Denti M., Das A., Facchetti A., Driver T.G.
Chemistry - A European Journal,
2014
12.

Jiang X., Zhang H., Zhang Y., Zhao W.
Tetrahedron,
2012
13.

Burns D.H., Li Y.H., Shi D.C., Caldwell T.M.
Journal of Organic Chemistry,
2002
14.

Bari S.E., Iturraspe J., Frydman B.
Tetrahedron,
1995
15.

Jackson A.H., Pandey R.K., Nagaraja Rao K.R., Roberts E.
Tetrahedron Letters,
1985
16.

17.

Singh K., Sharma S., Sharma A.
Synthetic Communications,
2011
18.

Khan T.K., Jana S.K., Rao M.R., Shaikh M.S., Ravikanth M.
Inorganica Chimica Acta,
2012
19.

Wood T.E., Thompson A.
Chemical Reviews,
2007
20.

Mikhalitsyna E.A., Tyurin V.S., Nefedov S.E., Syrbu S.A., Semeikin A.S., Koifman O.I., Beletskaya I.P.
European Journal of Inorganic Chemistry,
2012
21.

Lash T.D.
Chemistry - A European Journal,
1996
22.

Fang Z., Liu B.
Tetrahedron Letters,
2008
23.

Nguyen L.T., Senge M.O., Smith K.M.
Journal of Organic Chemistry,
1996
24.

Wu L., Burgess K.
Chemical Communications,
2008
25.

Lindsey J.S.
Accounts of Chemical Research,
2009
26.
10.1016/j.mencom.2018.03.028_sbref0085b
Sharma
Der Chemica Sinica,
2015
27.

Zhao W., Carreira E.M.
Chemistry - A European Journal,
2006
28.

Pereira N.A., Pinho e Melo T.M.
Organic Preparations and Procedures International,
2014
29.

Landmana M., Görls H., Crause C., Nienaber H., Olivier A., Lotz S.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
2007