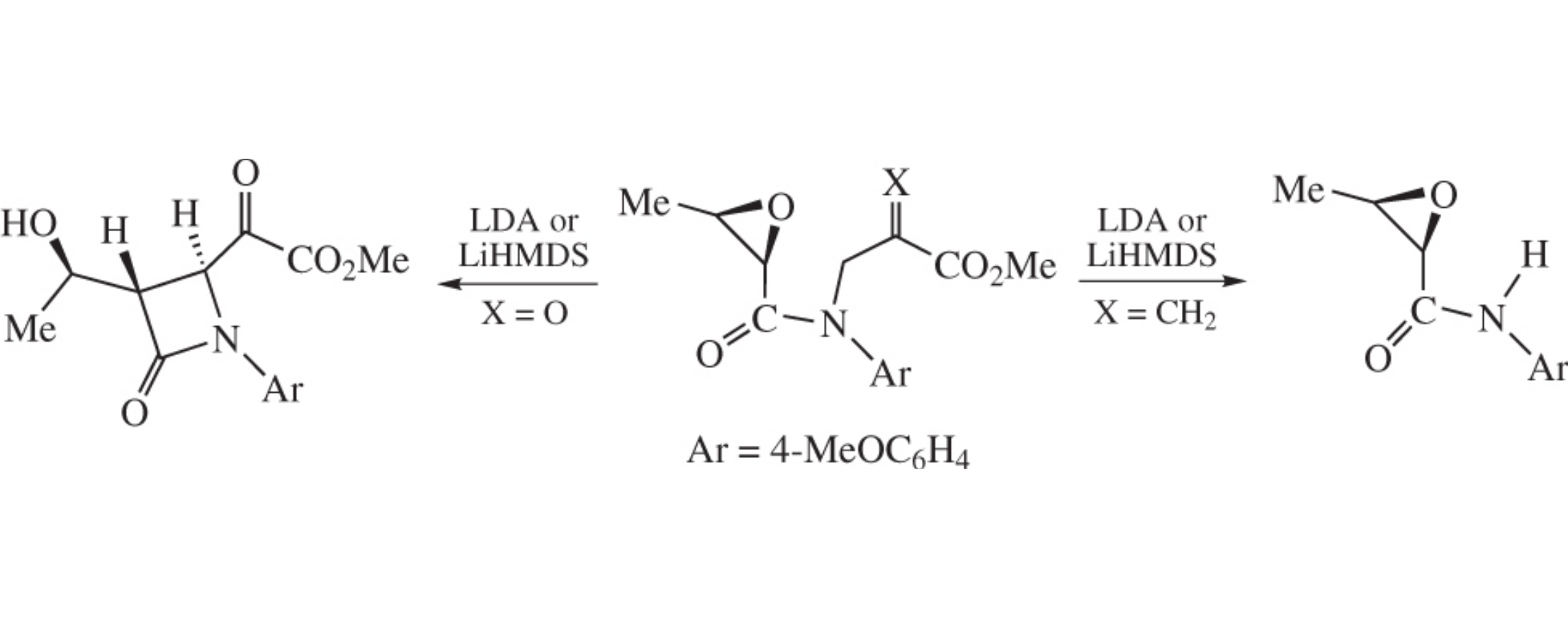
Abstract
(2R,3R)-N-(3-Methoxy-2,3-dioxopropyl)-N-(4-methoxy-phenyl)-2,3-epoxybutanamide on treatment with strong bases (LDA, LHMDS) gives 3-[(1R)-1-hydroxyethyl]azetidin-2-one derivative, while the N-(3-methoxy-2-methylidene-3-oxo-propyl) analogue undergoes fragmentation.
References
1.

Zhanel G.G., Wiebe R., Dilay L., Thomson K., Rubinstein E., Hoban D.J., Noreddin A.M., Karlowsky J.A.
Drugs,
2007
2.

Worthington R.J., Melander C.
Journal of Organic Chemistry,
2013
3.

4.

Kametani T., Nagahara T.
Heterocycles,
1987
5.

Cainelli G., Galletti P., Giacomini D.
Tetrahedron Letters,
1998
6.

Shiozaki M., Ishida N., Hiraoka T., Yanagisawa H.
Tetrahedron Letters,
1981
7.

Kugelman M., Gala D., Jaret R.S., Nyce P.L., McPhail A.T.
Synlett,
1990
8.

Niu C., Miller M.J.
Tetrahedron Letters,
1995
9.

Bose A.K., Manhas M.S., van der Veen J.M., Bari S.S., Wagle D.R., Hegde V.R., Krishnan L.
Tetrahedron Letters,
1985
10.

Hanessian S., Bedeschi A., Battistini C., Mongelli N.
Journal of the American Chemical Society,
1985
11.

Shiozaki M., Ishida N., Maruyama H., Hiraoka T.
Tetrahedron,
1983
12.

Shiozaki M., Ishida N., Hiraoka T., Maruyama H.
Tetrahedron,
1984
13.

Maruyama H., Shiozaki M., Hiraoka T.
Bulletin of the Chemical Society of Japan,
1985
14.

Shimohigashi Y., Waki M., Izumiya N.
Bulletin of the Chemical Society of Japan,
1979
15.
A. B. Baylis and M. E. D. Hillman, German Patent 2155113, 1972.(Chem. Abstr., 1973, 77, 34174q).
16.

Kruse P.F., Geurkink N., Grist K.L.
Journal of the American Chemical Society,
1954
17.
10.1016/j.mencom.2018.03.005_bib0045
Laurent
Eur. J. Org. Chem.,
2006
18.

Koch T., Hesse M.
Synthesis,
1992
19.

Davies S.G., Huckvale R., Lee J.A., Lorkin T.J., Roberts P.M., Thomson J.E.
Tetrahedron,
2012
20.

Kajihara K., Arisawa M., Shuto S.
Journal of Organic Chemistry,
2008
21.
10.1016/j.mencom.2018.03.005_bib0065
Barluenga
Chem. Commun.,
2005