Abstract
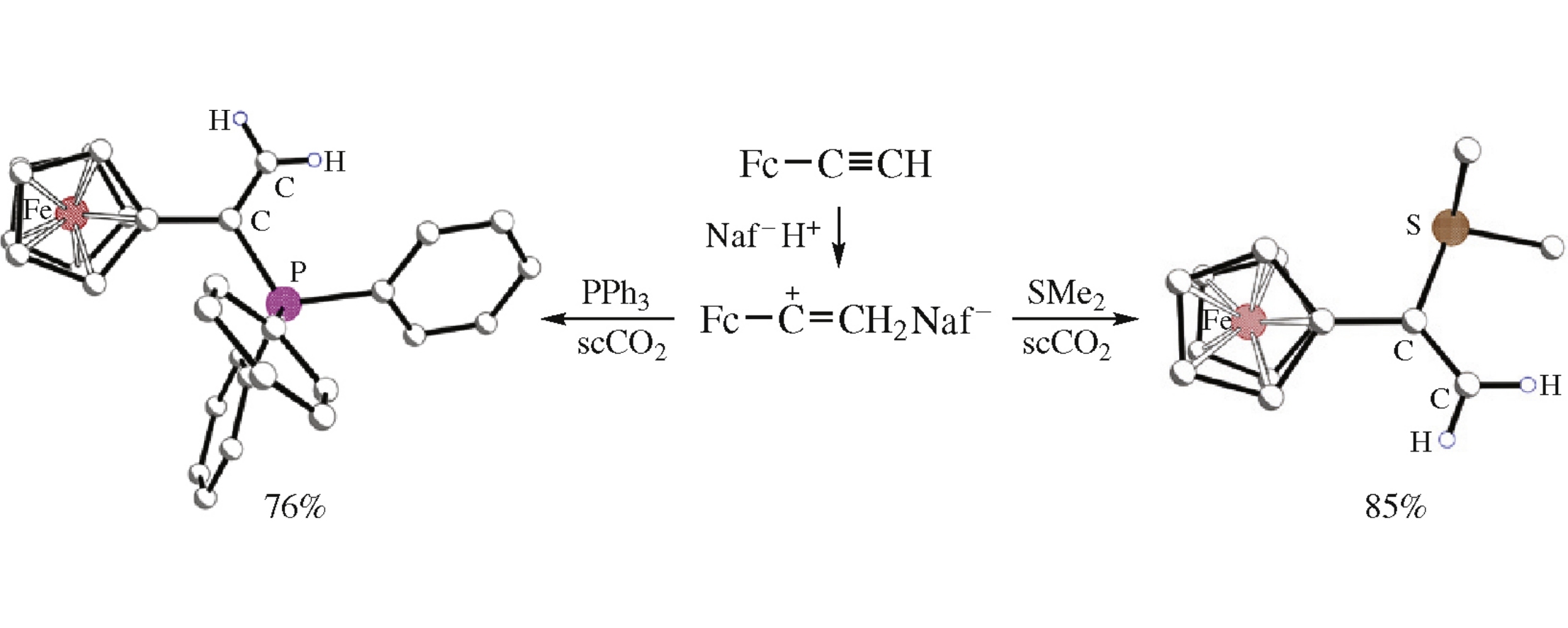
Cation CpFeC5H4–C+=CH2 was obtained by protonation of FcC≡CH with Nafion superacid in DMF or scCO2 and characterized by NMR spectroscopy. The protonation in the presence of SMe2 or PPh3 affords new onium derivatives, which were isolated as the tetrafluoroborate salts.
References
1.
10.1016/j.mencom.2017.07.016_sbref0005a
Deno
1970
2.
10.1016/j.mencom.2017.07.016_sbref0005b
Richey
1970
3.

Abram T.S., Watts W.E.
Journal of the Chemical Society Perkin Transactions 1,
1977
4.

Abram T.S., Watts W.E.
Journal of the Chemical Society Perkin Transactions 1,
1977
5.

Abram T.S., Watts W.E.
Journal of the Chemical Society Perkin Transactions 1,
1977
6.
10.1016/j.mencom.2017.07.016_sbref0015a
Gelbard
Chem. Res.,
2005
7.
10.1016/j.mencom.2017.07.016_sbref0015b
El–Kattan
Encyclopedia of Reagents for Organic Synthesis,
2001
8.

Elliott J.A., Hanna S., Elliott A.M., Cooley G.E.
Polymer,
2001
9.
10.1016/j.mencom.2017.07.016_sbref0015d
Sokolov
Chem.,
2010
10.

Kizas O.A., Antonov D.Y., Vopilov Y.E., Godovikov I.A., Peregudov A.S., Kagramanov N.D., Bulatnikova L.N., Nikitin L.N., Khokhlov A.R.
Journal of Supercritical Fluids,
2013
11.

Rosenblum M., Brawn N., Papenmeier J., Applebaum M.
Journal of Organometallic Chemistry,
1966
12.

Ivanchev S.S., Myakin S.V.
Russian Chemical Reviews,
2010
13.
10.1016/j.mencom.2017.07.016_sbref0025b
Kreure
2003
14.

15.

Jessop P.G., Ikariya T., Noyori R.
Chemical Reviews,
1999
16.

Darr J.A., Poliakoff M.
Chemical Reviews,
1999
17.

Beckman E.J.
Journal of Supercritical Fluids,
2004
18.

Jessop P.G.
Journal of Supercritical Fluids,
2006
19.

Rayner C.M.
Organic Process Research and Development,
2006
20.

Morita D.K., David S.A., Tumas W., Morita D.K., Pesiri D.R., Glaze W.H.
Chemical Communications,
1998
21.

Gordon R.S., Holmes A.B.
Chemical Communications,
2002
22.
10.1016/j.mencom.2017.07.016_sbref0035c
Gallyamov
Chem. Phys.,
2008
23.

Lyubimov S.E., Kuchurov I.V., Verbitskaya T.A., Rastorguev E.A., Kalinin V.N., Zlotin S.G., Davankov V.A.
Journal of Supercritical Fluids,
2010
24.

Fujita S., Yoshida H., Asai K., Meng X., Arai M.
Journal of Supercritical Fluids,
2011
25.

Galia A., Scialdone O., Tortorici E.
Journal of Supercritical Fluids,
2011
26.

Rodríguez L., Rossell O., Seco M., Orejón A., Masdeu-Bultó A.M.
Journal of Supercritical Fluids,
2011
27.

Guerrero-Gutiérrez E.M., Suleiman D.
Journal of Applied Polymer Science,
2012
28.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
29.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007