Abstract
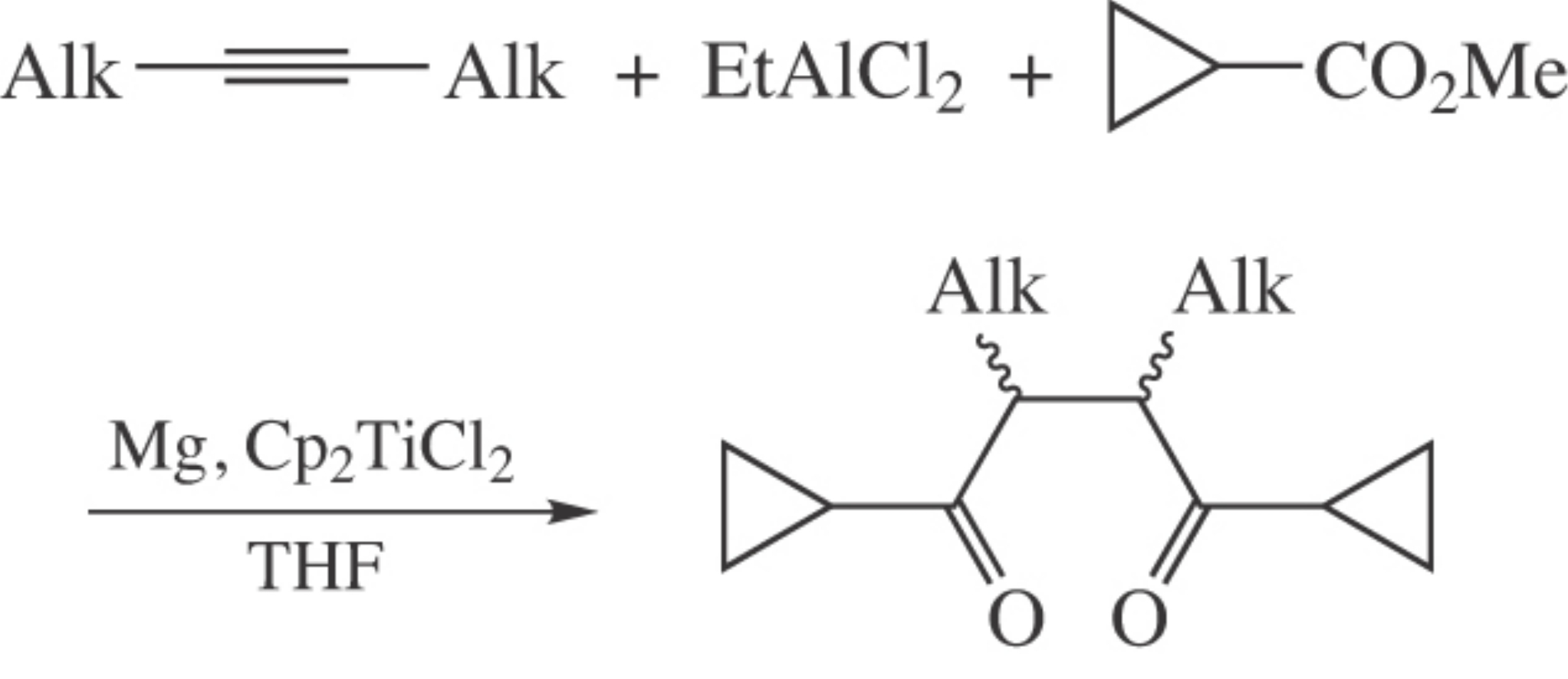
2,3-Dialkyl-l,4-dicyclopropylbutane-1,4-diones were prepared in one pot from symmetrical acetylenes, ethylaluminum dichloride and methyl cyclopropanecarboxylate in the presence of magnesium metal and Cp2TiCl2 (10 mol%) as a catalyst in 62–66% yield. Comparison of 1,4-dicyclopropylbutane-1,4-diones with other analogues to form furans in the course of the Paal–Knorr reaction was made by quantum chemical DFT calculations.
References
1.

2.

3.

Oshima K., Yamamoto H., Nozaki H.
Journal of the American Chemical Society,
1973
4.
10.1016/j.mencom.2016.04.015_sbref0005d
Stetter
Org. React.,
1991
5.

Lu X., Ji J., Ma D., Shen W.
Journal of Organic Chemistry,
1991
6.

Motoyoshiya J., Hongo Y., Tanaka H., Hayashi S.
Synthetic Communications,
1991
7.

Freeman F., Kim D.S., Rodriguez E.
Journal of Organic Chemistry,
1992
8.
10.1016/j.mencom.2016.04.015_sbref0015a
Hassner
1991
9.

Stetter H., Bender H.
Chemische Berichte,
1981
10.
11.
10.1016/j.mencom.2016.04.015_sbref0020b
O’Neill
1991
12.

Crisp G.T., Bubner T.P.
Synthetic Communications,
1990
13.

Verlhac J.B., Pereyre M., Shin H.A.
Organometallics,
1991
14.

Cui L., Zhang G., Zhang L.
Bioorganic and Medicinal Chemistry Letters,
2009
15.

Stork G., Borch R.
Journal of the American Chemical Society,
1964
16.

Büchi G., Wüest H.
Journal of Organic Chemistry,
1966
17.

Crombie L., Hemesley P., Pattenden G.
Journal of the Chemical Society C Organic,
1969
18.

Mc Murry J.E., Glass T.E.
Tetrahedron Letters,
1971
19.

Ellison R.A., Woessner W.D.
Journal of the Chemical Society Chemical Communications,
1972
20.

Shaibakova M.G., Khafizova L.O., Chobanov N.M., Gubaidullin R.R., Popod’ko N.R., Dzhemilev U.M.
Tetrahedron Letters,
2014
21.

Khafizova L.O., Shaibakova M.G., Chobanov N.M., Gubaidullin R.R., Tyumkina T.V., Dzhemilev U.M.
Russian Journal of Organic Chemistry,
2015
22.
L. O. Khafizova, M.G. Shaibakova, T.V. Tyumkina, N.M. Chobanov and U. M. Dzhemilev, Patent RF 2565789 C1, 08.04.2014.
23.

Szakal-Quin G., Graham D.G., Millington D.S., Maltby D.A., McPhail A.T.
Journal of Organic Chemistry,
1986
24.

Paal C.
Berichte der deutschen chemischen Gesellschaft,
1884
25.

Knorr L.
Berichte der deutschen chemischen Gesellschaft,
1884
26.

Mothana B., Boyd R.J.
Journal of Molecular Structure THEOCHEM,
2007
27.

Amarnath V., Amarnath K., Amarnath K., Davies S., Roberts L.J.
Chemical Research in Toxicology,
2004
28.

Amarnath V., Anthony D.C., Amarnath K., Valentine W.M., Wetterau L.A., Graham D.G.
Journal of Organic Chemistry,
1991
29.

Amarnath V., Amarnath K.
Journal of Organic Chemistry,
1995
30.

Kulinkovich O.G.
Pure and Applied Chemistry,
2000
31.

Wu Y., Yu Z.
Journal of the American Chemical Society,
2001
32.

Laikov D.N.
Chemical Physics Letters,
1997
33.

Laikov D.N., Ustynyuk Y.A.
Russian Chemical Bulletin,
2005
34.

Perdew J.P., Burke K., Ernzerhof M.
Physical Review Letters,
1996
35.
G. A. Zhurko and D. A. Zhurko, ChemCraft 1.6, 2009.
36.

Dewar M.J., Harris J.M.
Journal of the American Chemical Society,
1968
37.

Nikoletić M., Borçić S., Sunko D.E.
Tetrahedron,
1967
38.

Lambert J.B., Napoli J.J., Johnson K.K., Taba K.N.
Journal of Organic Chemistry,
1985
39.

Frost J.R., Cheong C.B., Akhtar W.M., Caputo D.F., Stevenson N.G., Donohoe T.J.
Journal of the American Chemical Society,
2015
40.

Kagermeier N., Werner K., Keller M., Baumeister P., Bernhardt G., Seifert R., Buschauer A.
Bioorganic and Medicinal Chemistry,
2015
41.

Yang T., Li Q., Lin J., Xiao J.
Chemical Communications,
2014
42.
![Chiral synthons from carvone. Part 50.† Enantiospecific approaches to both enantiomers of bicyclo[4.3.0]nonane-3,8-dione derivatives](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Srikrishna A., Reddy T.J.
Journal of the Chemical Society Perkin Transactions 1,
2001
43.
![2,6-Bis[(2S)-tetrahydrofuran-2-yl]phenyl diselenide: An effective reagent for asymmetric electrophilic addition reactions to olefins](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Déziel R., Malenfant E., Thibault C., Fréchette S., Gravel M.
Tetrahedron Letters,
1997
44.
![Synthesis of the Valeriana waalichi hydrocarbon sesquifenchene. Route to specifically functionalized 7,7-disubstituted bicyclo[2.2.1]heptane derivatives](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Grieco P.A., Masaki Y.
Journal of Organic Chemistry,
1975
45.

Yin G., Wang Z., Chen A., Gao M., Wu A., Pan Y.
Journal of Organic Chemistry,
2008