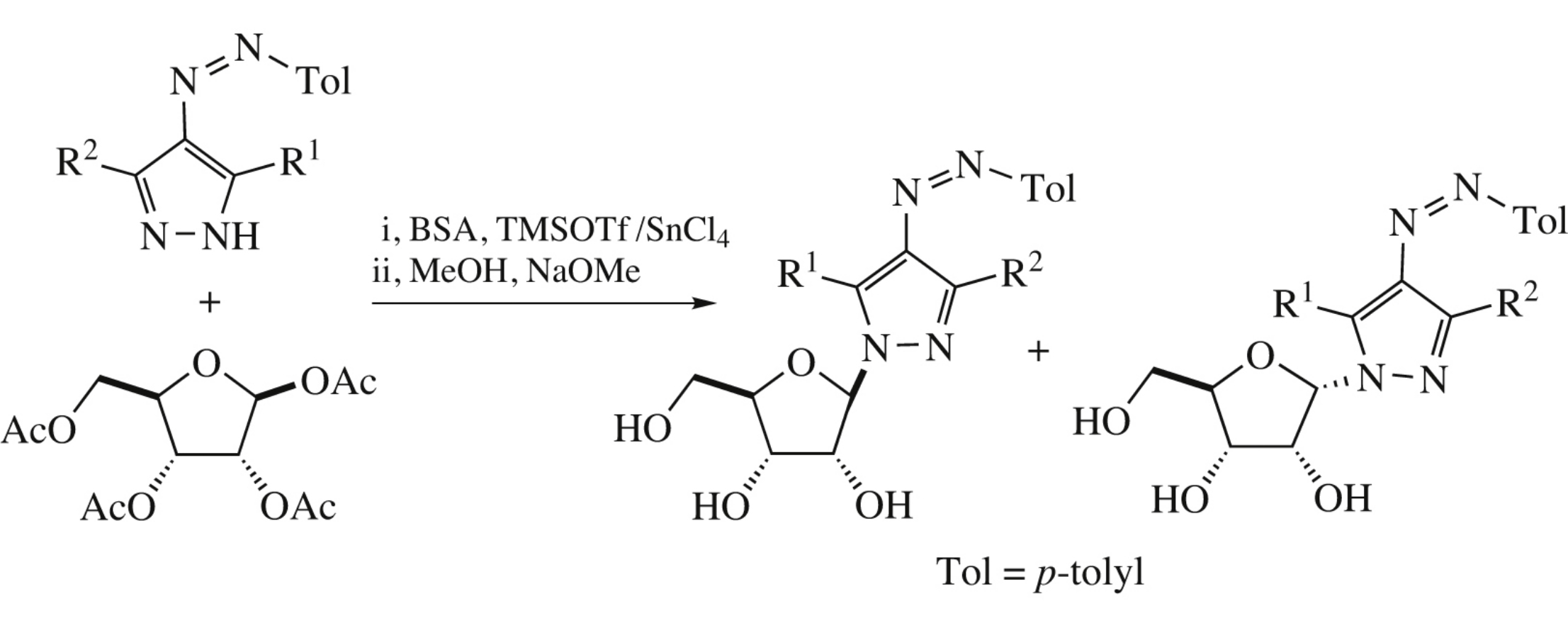
Abstract
Ribosylation of 4-aryldiazenyl-3-R-5-R’-pyrazoles with 1,2,3,5-tetra-O-acetyl-β-D-ribofuranose in the presence of tin(IV) chloride gives α- and β-anomeric N-(2,3,5-tri-O-acetylribofuranosyl)-substituted derivatives. Their HPLC separation and deacetylation (MeOH, MeONa) affords 1-(β-D-ribofuranosyl)-3-R-5-R’-4-(aryldiazenyl)-1H-pyrazoles.
References
1.

Elgemeie G.H., Zaghary W.A., Amin K.M., Nasr T.M.
Nucleosides, Nucleotides and Nucleic Acids,
2005
2.

Manfredini S., Bazzanini R., Baraldi P.G., Simoni D., Vertuani S., Pani A., Pinna E., Scintu F., Lichino D., Colla P.L.
Bioorganic and Medicinal Chemistry Letters,
1996
3.

Garcia-Lopez M.T., Herranz R., Alonso G.
Journal of Medicinal Chemistry,
1979
4.

Storer R., Ashton C.J., Baxter A.D., Hann M.M., Marr C.L., Mason A.M., Mo C., Myers P.L., Noble S.A., Penn C.R., Weir N.G., Woods J.M., Coe P.L.
Nucleosides and Nucleotides,
1999
5.

Abdou I., Saleh A., Zohdi H.
Molecules,
2004
6.

Ivanova A.E., Burgart Y.V., Saloutin V.I.
Chemistry of Heterocyclic Compounds,
2013
7.
![Synthesis and the reactions of trifluoromethylated 1,2,3-triketones 2-(het)arylhydrazones and 4,7-dihydroazolo[5,1-c]triazines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Khudina O.G., Shchegol’kov E.V., Burgart Y.V., Kodess M.I., Kazheva O.N., Chekhlov A.N., Shilov G.V., Dyachenko O.A., Saloutin V.I., Chupakhin O.N.
Journal of Fluorine Chemistry,
2005
8.

Kett W.C., Batley M., Redmond J.W.
Carbohydrate Research,
1997
9.

Khudina O.G., Shchegol’kov E.V., Burgart Y.V., Kodess M.I., Saloutin V.I., Kazheva O.N., Shilov G.V., D’yachenko O.A., Grishina M.A., Potemkin V.A., Chupakhin O.N.
Russian Journal of Organic Chemistry,
2007
10.
10.1016/j.mencom.2016.03.006_bib0050
Sankpal
J. Chem. Pharm. Res.,
2010
11.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007