Abstract
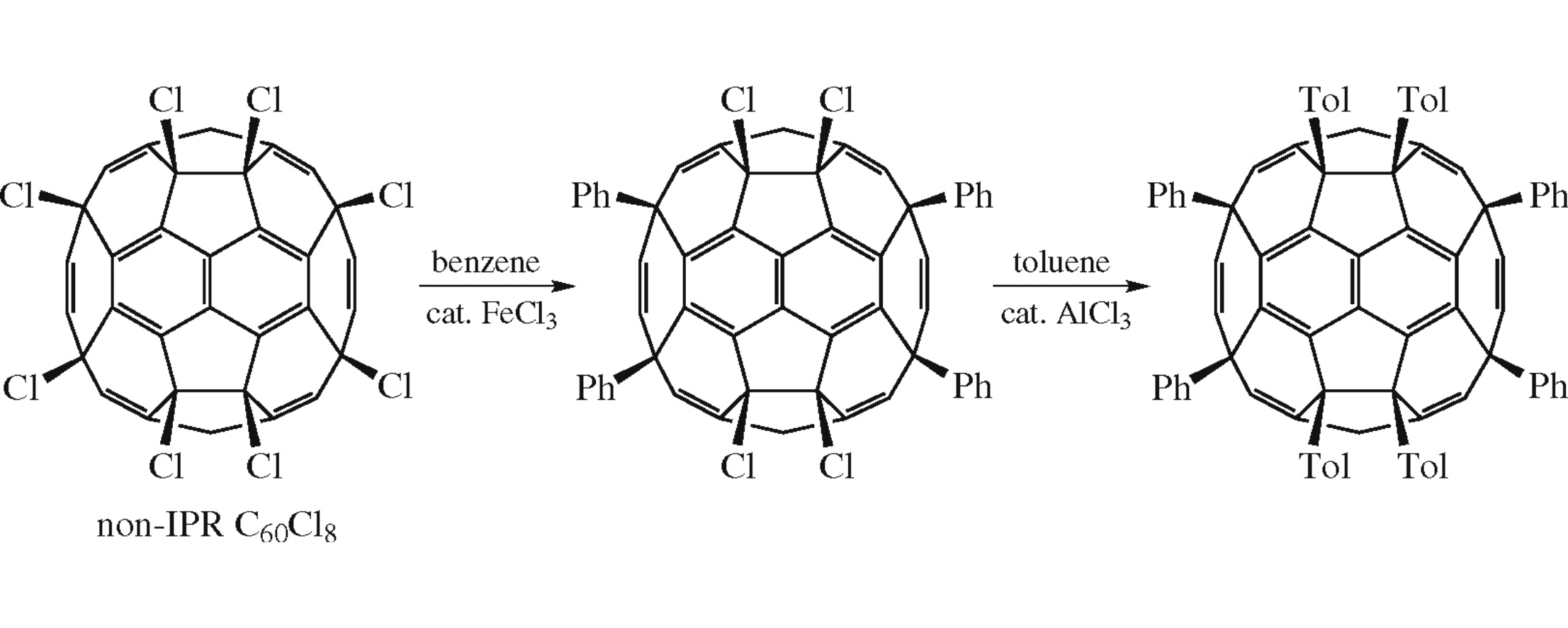
The eight chlorines of C60Cl8 with two pairs of fused pentagons are successfully substituted by sterically encumbering groups through the Friedel–Crafts reaction to get C60(C6H4Me)4Ph4, which provides a facile way for multiple modification of special fullerenes.
References
1.

Stevenson S., Fowler P.W., Heine T., Duchamp J.C., Rice G., Glass T., Harich K., Hajdu E., Bible R., Dorn H.C.
Nature,
2000
2.

Shi Z., Wu X., Wang C., Lu X., Shinohara H.
Angewandte Chemie - International Edition,
2006
3.

Ioffe I., Goryunkov A., Tamm N., Sidorov L., Kemnitz E., Troyanov S.
Angewandte Chemie - International Edition,
2009
4.

Amsharov K.Y., Ziegler K., Mueller A., Jansen M.
Chemistry - A European Journal,
2012
5.

Tan Y., Liao Z., Qian Z., Chen R., Wu X., Liang H., Han X., Zhu F., Zhou S., Zheng Z., Lu X., Xie S., Huang R., Zheng L.
Nature Materials,
2008
6.

7.

I. Troyanov S., Kemnitz E.
Current Organic Chemistry,
2012
8.

Ziegler K., Mueller A., Amsharov K.Y., Jansen M.
Chemistry - An Asian Journal,
2011
9.

Suzuki M., Mizorogi N., Yang T., Uhlik F., Slanina Z., Zhao X., Yamada M., Maeda Y., Hasegawa T., Nagase S., Lu X., Akasaka T.
Chemistry - A European Journal,
2013
10.

Yang S., Wei T., Wang S., Ignat'eva D.V., Kemnitz E., Troyanov S.I.
Chemical Communications,
2013
11.

Alegret N., Salvadó P., Rodríguez-Fortea A., Poblet J.M.
Journal of Organic Chemistry,
2013
12.
![Synthesis and Characterization of a Non-IPR Fullerene Derivative: Sc3N@C68[C(COOC2H5)2]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cai T., Xu L., Shu C., Reid J.E., Gibson H.W., Dorn H.C.
Journal of Physical Chemistry C,
2008
13.
10.1016/j.mencom.2016.01.012_bib0065
Fowler
An Atlas of Fullerenes,
1995
14.
![Arylation of [60]fullerene via electrophilic aromaticsubstitution involving the electrophile C60Cl6:frontside nucleophilic substitution of fullerenes](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Birkett P.R., Avent A.G., Darwish A.D., Hahn I., Kroto H.W., Langley G.J., O’Loughlin J., Taylor R., Walton D.R.
Journal of the Chemical Society Perkin Transactions 2,
1997
15.
Kornev A.B., Peregudov A.S., Martynenko V.M., Guseva G.V., Sashenkova T.E., Rybkin A.Y., Faingold I.I., Mishchenko D.V., Kotelnikova R.A., Konovalova N.P., Balzarini J., Troshin P.A.
Mendeleev Communications,
2013
16.
Kobzar O.L., Trush V.V., Tanchuk V.Y., Voronov I.I., Peregudov A.S., Troshin P.A., Vovk A.I.
Mendeleev Communications,
2015
17.
Biglova Y.N., Mikheev V.V., Torosyan S.A., Biglova R.Z., Miftakhov M.S.
Mendeleev Communications,
2015