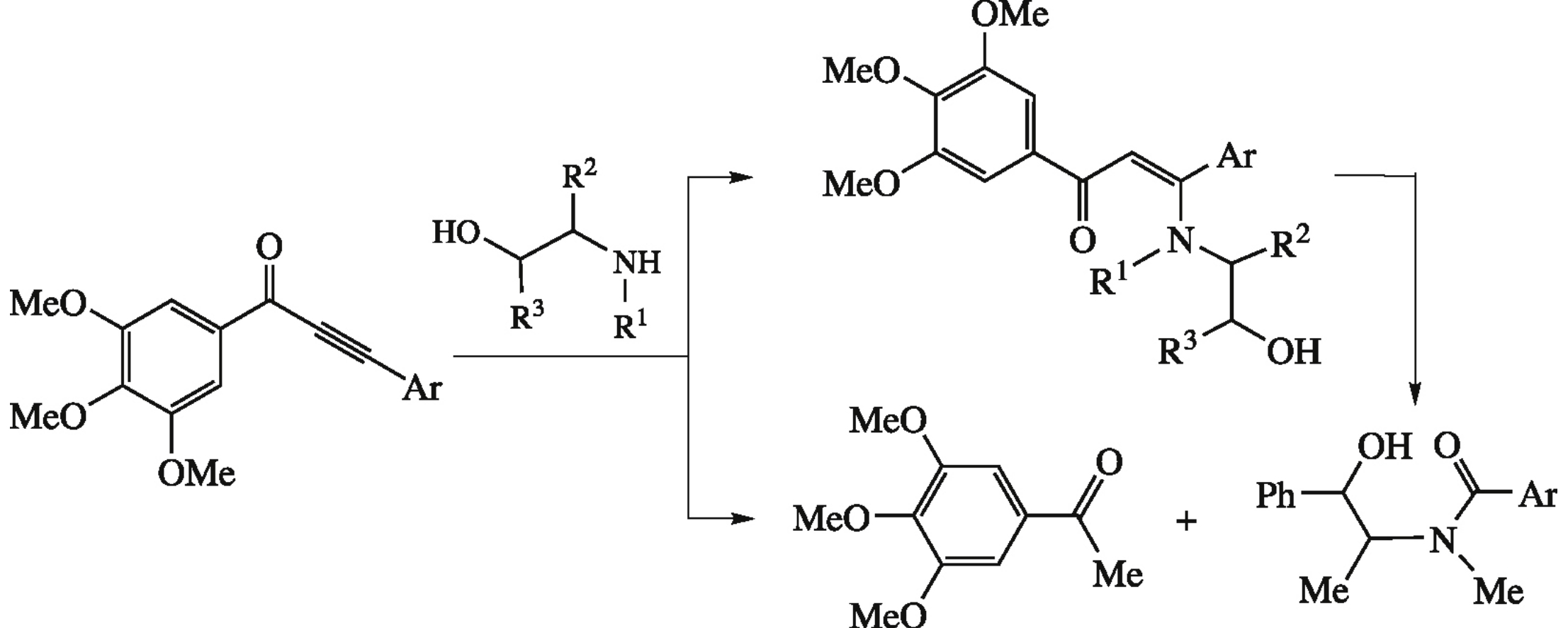
Abstract
Reaction of 3-aryl-1-(3,4,5-trimethoxyphenyl)prop-2-yn-1-ones with (+)-pseudoephedrine leads to products of alkyne moiety cleavage, namely, 1-(3,4,5-trimethoxyphenyl)ethanone and N-(1-hydroxy-1-phenylprop-2-yl)-N-methylbenzamides. In the course of the process one of alkyne carbons undergoes a formal reduction to a Me group, whereas the other one is oxidized to a C(O)NRR’ moiety.
References
1.
(a) Acetylene Chemistry: Chemistry, Biology and Material Science, eds. F. Diederich, P.J. Stang and R. R. Tykwinski, Wiley-VCH, Weinheim, 2005; (b) I. V. Alabugin and B. Gold, J. Org. Chem., 2013, 78, 7777; (c) J. C. Jewett and C. R. Bertozzi, Chem. Soc. Rev., 2010, 39, 1272; (d) E. T. Chernick and R. R. Tykwinski, J. Phys. Org. Chem., 2013, 26, 742; (e) B. Breiner, K. Kaya, S. Roy, W.-Y. Yang and I. V. Alabugin, Org. Biomol. Chem., 2012, 10, 3974; (f) V. V. Rostovtsev, L.G. Green, V.V. Fokin and K. B. Sharpless, Angew. Chem. Int. Ed., 2002, 41, 2596; (g) K. Gilmore and I. V. Alabugin, Chem. Rev., 2011, 111, 6513; (h) B. A. Trofimov, E. Yu. Schmidt, I.A. Bidusenko and N. A. Cherimichkina, Mendeleev Commun., 2015, 25, 17; (i) E. Yu. Schmidt, N.V. Zorina, E.V. Ivanova, I.V. Tatarinova, I.A. Ushakov, A.I. Mikhaleva and B. A. Trofimov, Mendeleev Commun., 2013, 23, 340; (j) D. A. Shabalin, T.E. Glotova, I.A. Ushakov, M. Yu. Dvorko, A.V. Vashchenko, V.I. Smirnov, E. Yu. Schmidt, A.I. Mikhaleva and B. A. Trofimov, Mendeleev Commun., 2014, 24, 368.
2.

Shi F., Luo S., Tao Z., He L., Yu J., Tu S., Gong L.
Organic Letters,
2011
3.

Wang Z., Chen Z., Bai S., Li W., Liu X., Lin L., Feng X.
Angewandte Chemie - International Edition,
2012
4.

Vasilevsky S.F., Baranov D.S., Mamatyuk V.I., Gatilov Y.V., Alabugin I.V.
Journal of Organic Chemistry,
2009
5.

Vasilevsky S.F., Mikhailovskaya T.F., Mamatyuk V.I., Salnikov G.E., Bogdanchikov G.A., Manoharan M., Alabugin I.V.
Journal of Organic Chemistry,
2009
6.

Davydova M.P., Vasilevskii S.F., Tolstikov G.A.
Russian Chemical Bulletin,
2011
7.

Roy S., Davydova M.P., Pal R., Gilmore K., Tolstikov G.A., Vasilevsky S.F., Alabugin I.V.
Journal of Organic Chemistry,
2011
8.
Vasilevsky S.F., Davydova M.P., Tomilin D.N., Sobenina L.N., Mamatuyk V.I., Pleshkova N.V.
Arkivoc,
2014
9.

Nenajdenko V.G., Muzalevskiy V.M., Shastin A.V., Balenkova E.S., Kondrashov E.V., Ushakov I.A., Rulev A.Y.
Journal of Organic Chemistry,
2010
10.

Yang X., Cheng G., Shen J., Kuai C., Cui X.
Organic Chemistry Frontiers,
2015
11.

Alabugin I.V., Gilmore K., Manoharan M.
Journal of the American Chemical Society,
2011
12.
10.1016/j.mencom.2015.09.021_bib0030
Basyouni
Acta Chim. Acad. Sci. Hung.,
1980
13.

Zanina A.S., Shergina S.I., Sokolov I.E., Myasnikova R.N.
Russian Chemical Bulletin,
1995