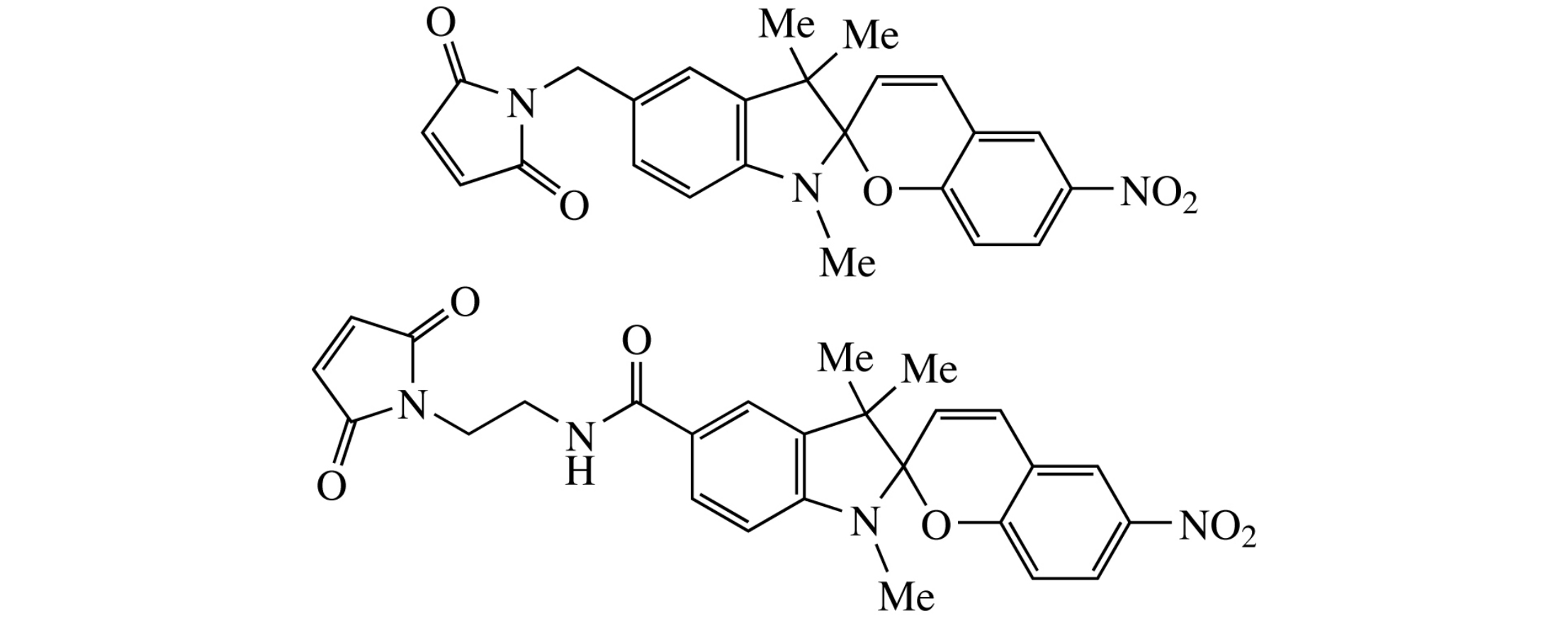
Abstract
Two new photochromic labels of spiro[chromene-2,2’-indole] series supplied with 5-positioned maleimide moieties suitable for labelling macromolecules with sulfhydryl groups were synthesized.
References
1.

Gregory J.D.
Journal of the American Chemical Society,
1955
2.

Smyth D., Blumenfeld O., Konigsberg W.
Biochemical Journal,
1964
3.

Misu R., Oishi S., Setsuda S., Noguchi T., Kaneda M., Ohno H., Evans B., Navenot J., Peiper S.C., Fujii N.
Bioorganic and Medicinal Chemistry Letters,
2013
4.

Ito T., Inoue M., Akamatsu K., Kusaka E., Tanabe K., Nishimoto S.
Bioorganic and Medicinal Chemistry Letters,
2011
5.

Wängler C., Schäfer M., Schirrmacher R., Bartenstein P., Wängler B.
Bioorganic and Medicinal Chemistry,
2011
6.

Garimella P.D., Datta A., Romanini D.W., Raymond K.N., Francis M.B.
Journal of the American Chemical Society,
2011
7.
10.1016/j.mencom.2014.06.020_sbref0010e
Liu
Bioconjugate Chem.,
2012
8.

Tolmachev V., Altai M., Sandström M., Perols A., Karlström A.E., Boschetti F., Orlova A.
Bioconjugate Chemistry,
2011
9.

Cleland W.W.
Biochemistry,
1964
10.

Klüter S., Grütter C., Naqvi T., Rabiller M., Simard J.R., Pawar V., Getlik M., Rauh D.
Journal of Medicinal Chemistry,
2009
11.

Harada M., Sisido M., Hirose J., Nakanishi M.
Bulletin of the Chemical Society of Japan,
1994
12.
10.1016/j.mencom.2014.06.020_sbref0025a
Sakata
J. Org. Chem., 2005,
2009
13.
10.1016/j.mencom.2014.06.020_sbref0025b
Marriott
US Patent 0195309,
2007
14.
Laptev A.V., Lukin A.Y., Belikov N.E., Barachevskii V.A., Demin O.V., Khodonov A.A., Varfolomeev S.D., Shvets V.I.
Mendeleev Communications,
2013
15.

A High Yielding Synthesis of N-Alkyl Maleimides Using a Novel Modification of the Mitsunobu Reaction
Walker M.A.
Journal of Organic Chemistry,
1995
16.
10.1016/j.mencom.2014.06.020_sbref0040a
Photochromism: Molecules and Systems,
2003
17.

Zakhs �.R., Martynova V.M., �fros L.S.
Chemistry of Heterocyclic Compounds,
1979
18.

Demina O.V., Levin P.P., Belikov N.E., Laptev A.V., Lukin A.Y., Barachevsky V.A., Shvets V.I., Varfolomeev S.D., Khodonov A.A.
Journal of Photochemistry and Photobiology A: Chemistry,
2013
19.
![Synthesis and study of the photochromic behavior of 3-[6′-nitro-1,3,3-trimethylspiro(indolino-2,2′-[2H]-chromen-5-yl)]propenoic acid and its ethyl ester](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Laptev A.V., Lukin A.Y., Belikov N.E., Zemtsov R.V., Shvets V.I., Demina O.V., Varfolomeev S.D., Barachevskii V.A., Khodonov A.A.
High Energy Chemistry,
2010