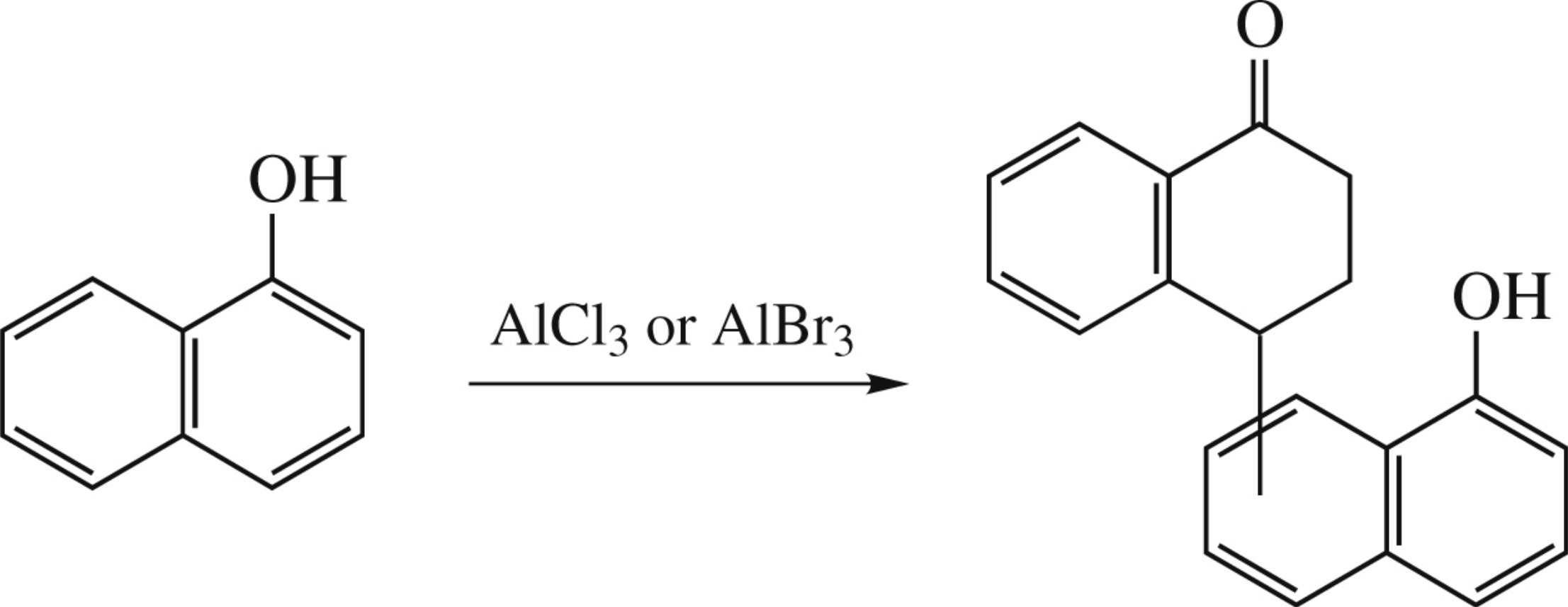
Abstract
Naphthols undergo dimerization in the presence of an excess of aluminum halides to give mixtures of isomeric hydroxynaphthyltetralones.
References
1.

Klussmann M., Sureshkumar D.
Synthesis,
2010
2.

Sarhan A.A., Bolm C.
Chemical Society Reviews,
2009
3.

Schenker S., Zamfir A., Freund M., Tsogoeva S.B.
European Journal of Organic Chemistry,
2011
4.

5.

Chen Y., Yekta S., Yudin A.K.
Chemical Reviews,
2003
6.

Kočovský P., Vyskočil Š., Smrčina M.
Chemical Reviews,
2003
7.
Karslyan E.E., Konovalov A.I., Borissova A.O., Petrovskii P.V., Kudinov A.R.
Mendeleev Communications,
2012
8.
Methoden der Organischen Chemie, ed. H. G. Thomas, Thieme, Stuttgart, 1976, vol. 7/2b, p. 1710.
9.
10.1016/j.mencom.2014.03.020_sbref0015b
Repinskaya
Sib. Khim. Zh.,
1993
10.
10.1016/j.mencom.2014.03.020_sbref0015c
Repinskaya
Russ. J. Org. Chem.,
1993
11.
10.1016/j.mencom.2014.03.020_sbref0020a
Koptyug
J. Org. Chem. USSR,
1971
12.
10.1016/j.mencom.2014.03.020_sbref0020b
Koltunov
Russ. J. Org. Chem.,
1994
13.

Koltunov K.Y., Chernov A.N., Surya Prakash G.K., Olah G.A.
Chemical and Pharmaceutical Bulletin,
2012
14.
10.1016/j.mencom.2014.03.020_sbref0025a
Koltunov
Russ. J. Org. Chem.,
1997
15.
10.1016/j.mencom.2014.03.020_sbref0025b
Koltunov
Russ. J. Org. Chem.,
1998
16.
10.1016/j.mencom.2014.03.020_bib0030
Olah
Superelectrophiles and Their Chemistry,
2008
17.
10.1016/j.mencom.2014.03.020_sbref0035a
Repinskaya
J. Org. Chem. USSR,
1988
18.
10.1016/j.mencom.2014.03.020_sbref0035b
Repinskaya
J. Org. Chem. USSR,
1992
19.

20.

Ksenofontov V.A., Vasina T.V., Zubarev Y.E., Kustov L.M.
Reaction Kinetics and Catalysis Letters,
2003
21.

BUU-HOÏ N.P., BIHAN H.L., BINON F.
Journal of Organic Chemistry,
1951
22.

BUU-HOÏ N.P., BIHAN H.L., BINON F., RAYET P.
Journal of Organic Chemistry,
1950
23.
10.1016/j.mencom.2014.03.020_bib0055
Koptyug
J. Org. Chem. USSR,
1970
24.

Koltunov K.Y., Prakash G.K., Rasul G., Olah G.A.
Journal of Organic Chemistry,
2007
25.
10.1016/j.mencom.2014.03.020_sbref0065a
Olah
Superacid Chemistry,
2009
26.

Smith G.P., Dworkin A.S., Pagni R.M., Zingg S.P.
Journal of the American Chemical Society,
1989
27.

Smith G.P., Dworkin A.S., Pagni R.M., Zingg S.P.
Journal of the American Chemical Society,
1989
28.

Koltunov K.Y., Prakash G.K., Rasul G., Olah G.A.
Journal of Organic Chemistry,
2002
29.

Fatiadi A.J.
Journal of Research of the National Bureau of Standards Section A Physics and Chemistry,
1968
30.
![Orthoamides, LVI [1]. A New Method of Wide Scope for the Preparation of Aryl Formates](/storage/images/resized/3SpVxcYL33bOvPq4sHxJLH2NeKNeDloahSUpNiO4_small_thumb.webp)
Ziegler G., Kantlehner W.
Zeitschrift fur Naturforschung - Section B Journal of Chemical Sciences,
2001
31.

Kantlehner W.
European Journal of Organic Chemistry,
2003