Keywords
o-benzothienylarenes
o-thieno[2,3-b]pyridinyl- arenes
3,4-dihydroisoquinolines
heterocyclization
recyclization
β-arylethylamines
Abstract
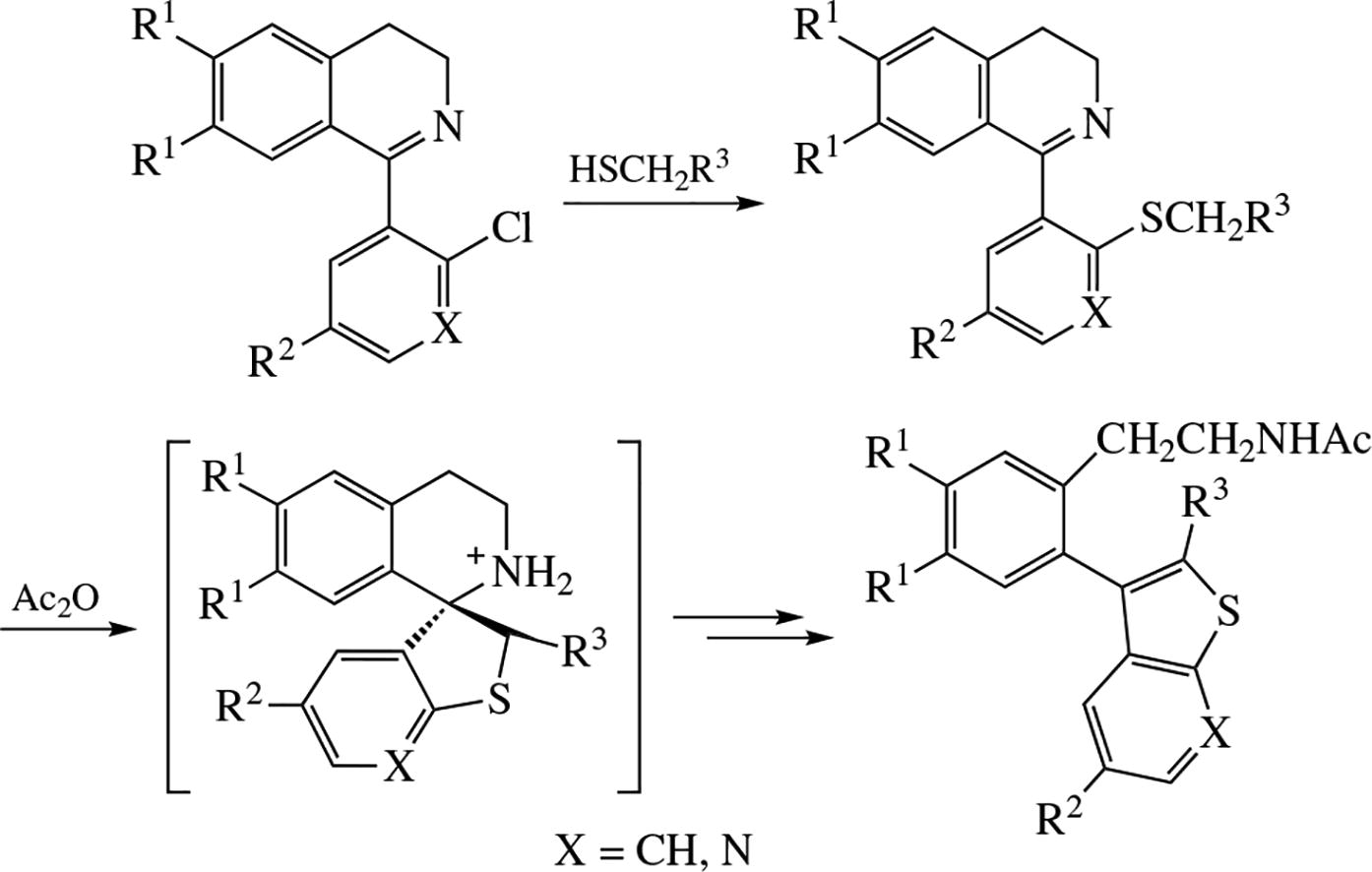
A new recyclization of 3,4-dihydroisoquinolines is a convenient route to derivatives of new bbb-(o-benzothienylaryl)-and bbb-(o-thieno[2,3-b]pyridinylaryl)-containing ethylamines. These compounds look promising monoaminergic agents.
References
1.
(a) A. Rosas-Cruz, N. Salinas-Jazmín and M. A. Velasco-Velázquez, Technol. Cancer Res. Treat., 2021, 15330338211027913
2.

Roney M.S., Park S.
Archives of Pharmacal Research,
2018
3.

Weissenrieder J.S., Neighbors J.D., Mailman R.B., Hohl R.J.
Journal of Pharmacology and Experimental Therapeutics,
2019
4.

LEE J., NAM D., LEE J.
Oncology Letters,
2016
5.

Kitson S.
Current Pharmaceutical Design,
2007
6.

Genet N., Billaud M., Rossignol R., Dubois M., Gillibert-Duplantier J., Isakson B.E., Marthan R., Savineau J., Guibert C.
Frontiers in Physiology,
2017
7.

The Rise and Fall of a Scaffold: A Trend Analysis of Scaffolds in the Medicinal Chemistry Literature
Zdrazil B., Guha R.
Journal of Medicinal Chemistry,
2017
8.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Demidov O.P., Kartsev V.G., Sochnev V.S., Klimenko A.I., Dobaeva N.M., Borodkin G.S., Bodryakov A.N., Bodryakova M.A., Fetisov L.N.
Mendeleev Communications,
2021
9.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Demidov O.P., Sochnev V.S., Borodkina I.G., Drobin Y.D., Spasov A.A.
Mendeleev Communications,
2020
10.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Sochnev V.S., Demidov O.P., Klimenko A.I., Fetisov L.N., Bodryakov A.N., Bodryakova M.A., Borodkin G.S.
Mendeleev Communications,
2022
11.
(d) A. A. Zubenko, A. S. Morkovnik, L. N. Divaeva, V. S. Sochnev, O. P. Demidov, A. N. Bodryakov, L. N. Fetisov, K. N. Kononenko, M. A. Bodryakova and A. I. Klimenko, Russ. J. Gen. Chem., 2021, 91, 792 (Zh. Obshch. Khim., 2021, 91, 703)
12.

Suna E., Trapencieris P.
Chemistry of Heterocyclic Compounds,
2000
13.

Kuo C., Wu M., Kuo Y.
European Journal of Medicinal Chemistry,
2006
14.
CrysAlisPro Software System, version 1.171.38.41, Rigaku Oxford Diffraction, Oxford, 2015.
15.
10.1016/j.mencom.2022.11.029_b0045
Sheldrick
Acta Crystallogr.,
2015
16.
10.1016/j.mencom.2022.11.029_b0050
Sheldrick
Acta Crystallogr.,
2015
17.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
18.

Kułaga D., Drabczyk A.K., Satała G., Latacz G., Rózga K., Plażuk D., Jaśkowska J.
European Journal of Medicinal Chemistry,
2022