Keywords
alkyl bromides
alkyl esters
alkyl-H-phosphinic acids
alkylation
esterification
red phosphorus
Abstract
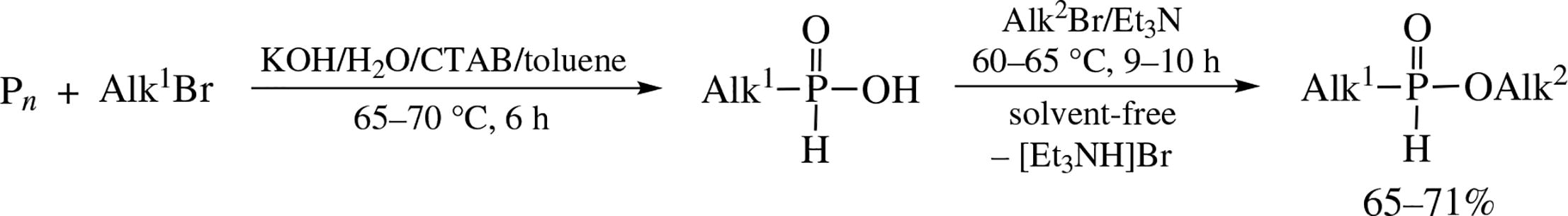
Alkyl-H-phosphinic acid alkyl esters are synthesized in 65–71% yield via chemoselective reaction of alkyl bromides with available alkyl-H-phosphinic acids (60–65 °C, Et3N). The latter are prepared, in turn, by direct phosphorylation of alkyl bromides with red phosphorus under phase-transfer conditions.
References
1.

Walton J.W., Bourdolle A., Butler S.J., Soulie M., Delbianco M., McMahon B.K., Pal R., Puschmann H., Zwier J.M., Lamarque L., Maury O., Andraud C., Parker D.
Chemical Communications,
2013
2.

Liu K., Wang Y., Li P., Cai W., Xiang J.
Catalysis Communications,
2012
3.

Cross J.M., Gallagher N., Gill J.H., Jain M., McNeillis A.W., Rockley K.L., Tscherny F.H., Wirszycz N.J., Yufit D.S., Walton J.W.
Dalton Transactions,
2016
4.
(a) W. Froestl, S. J. Mickel, G. von Sprecher, P. J. Diel, R. G. Hall, L. Maier, D. Strub, V. Melillo, P. A. Baumann, R. Bernasconi, C. Gentsch, K. Hauser, J. Jaekel, G. Karlsson, K. Klebs, L.Maitre, C. Marescaux, M. F. Pozza, M. Schmutz, M. W. Steinmann, H. van Riezen, A.Vassout, C. Mondadori, H.-R. Olpe, P. C. Waldmeier and H. Bittiger, J. Med. Chem., 1995, 38, 3313
5.

Abdou M.M., El-Saeed R.A.
Bioorganic Chemistry,
2019
6.

Jia X., Weber S., Schols D., Meier C.
Journal of Medicinal Chemistry,
2020
7.
![2-Arylbenzo[d]oxazole Phosphinate Esters as Second-Generation Modulators of Utrophin for the Treatment of Duchenne Muscular Dystrophy](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Babbs A., Berg A., Chatzopoulou M., Davies K.E., Davies S.G., Edwards B., Elsey D.J., Emer E., Guiraud S., Harriman S., Lecci C., Moir L., Peters D., Robinson N., Rowley J.A., et. al.
Journal of Medicinal Chemistry,
2020
8.

Finkbeiner P., Hehn J.P., Gnamm C.
Journal of Medicinal Chemistry,
2020
9.

Wendels S., Chavez T., Bonnet M., Salmeia K., Gaan S.
Materials,
2017
10.
(a) J. Wang, M. Xie, X. Liu and S. Xu, J. Visualized Exp., 2017, 128, e56156
11.

Carson I., Tasker P.A., Love J.B., Moser M., Fischmann A.J., Jakovljevic B., Soderstrom M.D., Morrison C.A.
European Journal of Inorganic Chemistry,
2018
12.

Li K., Chen J., Zou D., Deng Y., Li D.
Hydrometallurgy,
2019
13.

Keglevich G., Jablonkai E., Balázs L.B.
RSC Advances,
2014
14.

Montchamp J.
Accounts of Chemical Research,
2013
15.

Bálint E., Tóth R.E., Keglevich G.
Heteroatom Chemistry,
2016
16.

Chen C., Bao Y., Zhao J., Zhu B.
Chemical Communications,
2019
17.
(a) I. Abrunhosa-Thomas, P. Ribière, A. C. Adcock and J.-L. Montchamp, Synthesis, 2006, 0325; (b) M. I. Antczak and J.-L. Montchamp, Synthesis, 2006, 3080; (c) L.Coudray, K. Bravo-Altamirano and J.-L. Montchamp, Org. Lett., 2008, 10, 1123; (d) K. P. Yadavalli, J. E. Cummines, J. Carlisle and S. D. Lepore, Chem. Commun., 2022, 58, 6441; (e) K. Juhász, B. Varga, P. Bagi and Z. Hell, Catal. Lett., 2022, 152, 1100; (f) N. Popovics-Toth, M. Turpanova, K. Nemeth, L. Hackler, L. G. Puskas and E. Balint, Tetrahedron, 2022, 111, 132720; (g) L. Zheng, L. Cai, W. Mei, G. Liu, L. Deng, X. Zou, X. Zhuo, Y. Zhong and W. Guo, J. Org. Chem., 2022, 87, 6224; (h) K. R. Winters, C. Ricke and J.-L. Montchamp, Eur. J. Org. Chem., 2022, e202101130.
18.

Gallagher M.J., Ranasinghe M.G., Jenkins I.D.
Phosphorus, Sulfur and Silicon and the Related Elements,
1996
19.

Gavara L., Petit C., Montchamp J.
Tetrahedron Letters,
2012
20.

Dumond Y.R., Baker R.L., Montchamp J.
Organic Letters,
2000
21.
Trofimov B.A., Gusarova N.K.
Mendeleev Communications,
2009
22.
Gusarova N.K., Sutyrina A.O., Kuimov V.A., Malysheva S.F., Belogorlova N.A., Volkov P.A., Trofimov B.A.
Mendeleev Communications,
2019
23.

Gusarova N.K., Trofimov B.A.
Russian Chemical Reviews,
2020
24.

Kuimov V.A., Malysheva S.F., Belogorlova N.A., Gusarova N.K., Trofimov B.A.
Organic and Biomolecular Chemistry,
2021
25.

Trofimov B.A., Volkov P.A., Khrapova K.O., Telezhkin A.A., Ivanova N.I., Albanov A.I., Gusarova N.K., Chupakhin O.N.
Chemical Communications,
2018
26.
Volkov P.A., Ivanova N.I., Khrapova K.O., Telezhkin A.A., Borodina T.N., Gusarova N.K., Trofimov B.A.
Mendeleev Communications,
2018
27.

Trofimov B.A., Volkov P.A., Telezhkin A.A., Khrapova K.O., Ivanova N.I., Albanov A.I., Gusarova N.K.
Journal of Organic Chemistry,
2020
28.

Trofimov B.A., Volkov P.A., Telezhkin A.A.
Molecules,
2021