Keywords
alkoxide anion
bis(alkyl) complexes
carbinol
donor ligand
helical chirality
rare-earth metals
scandium complexes
Abstract
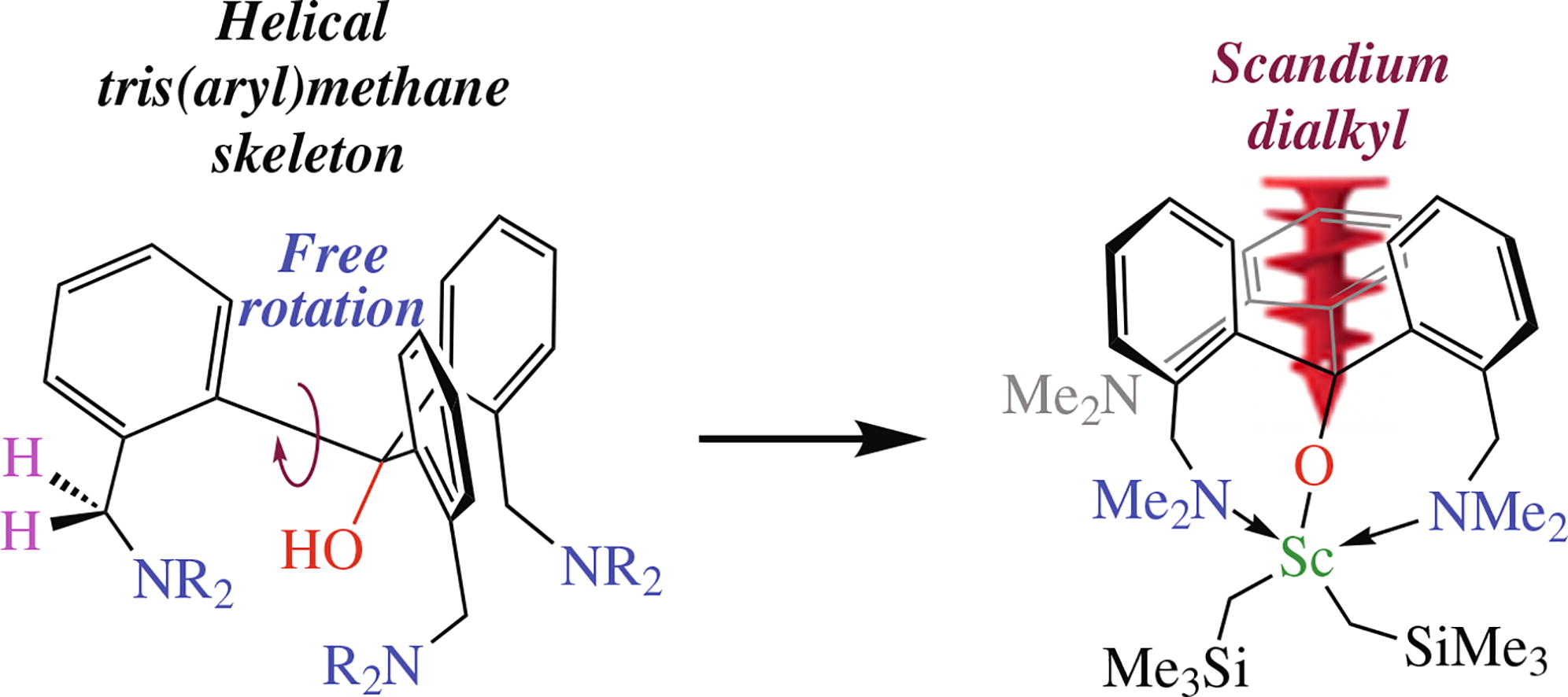
The reactions of aryllithium reagents o-LiC6H4CH2NR2 with (MeO)2CO afford two new tris(aryl)carbinols bearing pendant-NR2 donor groups in the side chain [o-R NCH C H ] COH [R = Me, R + R = (CH) ]. These alcohols feature helical chirality due to differently inclined aromatic fragments and are presented in a crystalline cell as two M and P enantiomers. Carbinol (R = Me) readily reacts with (Me3SiCH2)3Sc(THF)2 to give a scandium bis(alkyl) complex [(o-C6H4CH2NMe2)3CO]Sc(CH2SiMe3)2 featuring rigid binding of the alkoxy anion through a κ1-O, κ2-N chelating coordination mode
References
1.
(a) S. A. Cotton, Coord. Chem. Rev., 1997, 160, 93; (b) F. T. Edelmann, D. M. M. Freckmann and H. Schumann, Chem. Rev., 2002, 102, 1851; (c) R. Litlabø, M. Zimmermann, K. Saliu, J. Takats, K. W. Törnroos and R. Anwander, Angew. Chem., Int. Ed., 2008, 47, 9560; (d) M. Nishiura and Z. Hou, Nat. Chem., 2010, 2, 257; (e) W. Fegler, A. Venugopal, M. Kramer and J. Okuda, Angew. Chem., Int. Ed., 2015, 54, 1724; (f) J. Okuda, Coord. Chem. Rev., 2017, 340, 2; (g) D. Schädle and R. Anwander, Chem. Soc. Rev., 2019, 48, 5752; (h) L. Giusti, V. R. Landaeta, M. Vanni, J. A. Kelly, R. Wolf and M. Caporali, Coord. Chem. Rev., 2021, 441, 213927; (i) Y. Guan, E. Lu and X. Xu, J. Rare Earths, 2021, 39, 1017; (j) F. Ortu, Chem. Rev., 2022, 122, 6040.
2.

Trifonov A.A.
Coordination Chemistry Reviews,
2010
3.

Zimmermann M., Anwander R.
Chemical Reviews,
2010
4.

Nishiura M., Guo F., Hou Z.
Accounts of Chemical Research,
2015
5.
(d)A. A. Trifonov and D. M. Lyubov, Coord. Chem. Rev., 2017, 254, 1327
6.

Khristolyubov D.O., Lyubov D.M., Trifonov A.A.
Russian Chemical Reviews,
2021
7.

Liu D., Liu B., Pan Z., Li J., Cui C.
Science China Chemistry,
2019
8.

Pan Y., Jiang X., So Y., To C.T., He G.
Catalysts,
2020
9.

Nie K., Han Y., Wang C., Cheng X.
Applied Organometallic Chemistry,
2021
10.

Pellissier H.
Coordination Chemistry Reviews,
2016
11.

Shannon R.D.
Acta Crystallographica Section A,
1976
12.

Morss L.R.
Chemical Reviews,
1976
13.

Sadow A.D., Tilley T.D.
Journal of the American Chemical Society,
2003
14.

Seth K.
Organic Chemistry Frontiers,
2022
15.

Shi X., Nishiura M., Hou Z.
Journal of the American Chemical Society,
2016
16.

Luo Y., Teng H., Nishiura M., Hou Z.
Angewandte Chemie - International Edition,
2017
17.

Luo Y., Ma Y., Hou Z.
Journal of the American Chemical Society,
2017
18.

Nagae H., Kundu A., Inoue M., Tsurugi H., Mashima K.
Asian Journal of Organic Chemistry,
2018
19.

Selikhov A.N., Boronin E.N., Cherkasov A.V., Fukin G.K., Shavyrin A.S., Trifonov A.A.
Advanced Synthesis and Catalysis,
2020
20.
![Diastereodivergent [3 + 2] Annulation of Aromatic Aldimines with Alkenes via C–H Activation by Half-Sandwich Rare-Earth Catalysts](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Cong X., Zhan G., Mo Z., Nishiura M., Hou Z.
Journal of the American Chemical Society,
2020
21.

Lou S., Mo Z., Nishiura M., Hou Z.
Journal of the American Chemical Society,
2020
22.

Lou S., Zhang L., Luo Y., Nishiura M., Luo G., Luo Y., Hou Z.
Journal of the American Chemical Society,
2020
23.

Lin H., Li Y., Wang J., Zhang M., Jiang T., Li J., Chen Y.
Applied Organometallic Chemistry,
2021
24.

Lou S., Zhuo Q., Nishiura M., Luo G., Hou Z.
Journal of the American Chemical Society,
2021
25.

Lou S., Luo G., Yamaguchi S., An K., Nishiura M., Hou Z.
Journal of the American Chemical Society,
2021
26.

Huang W., Diaconescu P.L.
Advances in Organometallic Chemistry,
2015
27.

Arnold P.L., McMullon M.W., Rieb J., Kühn F.E.
Angewandte Chemie - International Edition,
2014
28.

Gandeepan P., Müller T., Zell D., Cera G., Warratz S., Ackermann L.
Chemical Reviews,
2018
29.

Scott J., Basuli F., Fout A., Huffman J., Mindiola D.
Angewandte Chemie - International Edition,
2008
30.

Lu E., Li Y., Chen Y.
Chemical Communications,
2010
31.

Chu T., Piers W.E., Dutton J.L., Parvez M.
Organometallics,
2012
32.

Jian Z., Rong W., Mou Z., Pan Y., Xie H., Cui D.
Chemical Communications,
2012
33.

Rong W., Cheng J., Mou Z., Xie H., Cui D.
Organometallics,
2013
34.

Evans W.J.
Inorganic Chemistry,
2007
35.

Wedal J.C., Evans W.J.
Journal of the American Chemical Society,
2021
36.

Zhang Z., Cui D., Trifonov A.A.
European Journal of Inorganic Chemistry,
2010
37.

Arnold P.L., Turner Z.R., Bellabarba R., Tooze R.P.
Journal of the American Chemical Society,
2011
38.

Arnold P.L., Marr I.A., Zlatogorsky S., Bellabarba R., Tooze R.P.
Dalton Transactions,
2014
39.

Elvidge B.R., Arndt S., Spaniol T.P., Okuda J.
Dalton Transactions,
2006
40.

Zhang J., Ma X., Wang X., Cai J., Wu X., Niu Y., Zhou N., Hua R.
Journal of Molecular Structure,
2015
41.

Niu Y., Zhang J., Wang X., Ma X., Cai J., Zhou N., Zheng Z., Hua R.
Journal of Molecular Structure,
2015
42.

Driesschaert B., Robiette R., Le Duff C.S., Collard L., Robeyns K., Gallez B., Marchand-Brynaert J.
European Journal of Organic Chemistry,
2012
43.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
44.
10.1016/j.mencom.2022.11.023_b0165
Sheldrick
Acta Crystallogr.,
2015
45.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007