Keywords
1,4,6-trialkylsemithioglycolurils
4,5-dihydroxyimidazolidin-2-ones
4,5-dihydroxyimidazolidine-2- thiones
alkylthioureas
dialkylureas
one-pot synthesis
Abstract
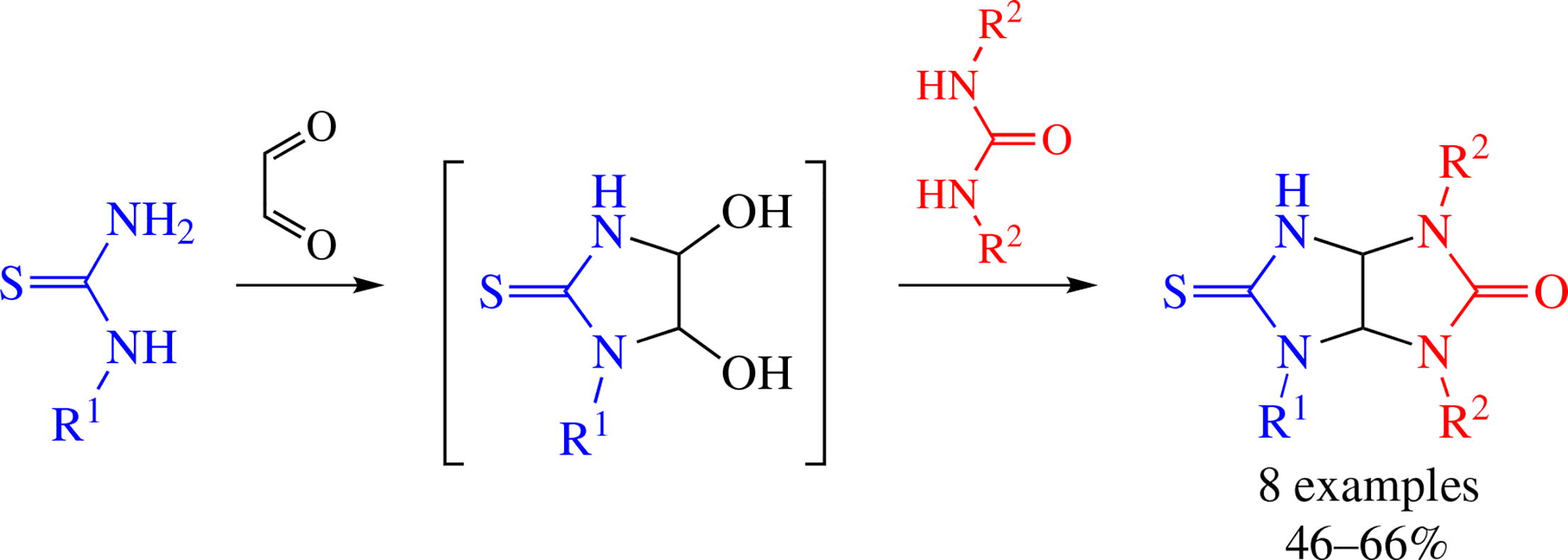
Condensation of 1-alkyl-4,5-dihydroxyimidazolidine-2-thiones (monoalkylthiourea-glyoxal cyclic adducts) with 1,3-dialkylureas affords novel 1,4,6-trialkylsemithio- glycolurils having non-substituted NH group linked to C=S function. Such compounds can be accessed in two-stage one-pot reaction sequence from alkylthioureas and glyoxal followed by treatment of the resulting adduct with 1,3-dialkylureas
References
1.

Kravchenko A.N., Baranov V.V., Gazieva G.A.
Russian Chemical Reviews,
2018
2.

Makhova N.N., Belen’kii L.I., Gazieva G.A., Dalinger I.L., Konstantinova L.S., Kuznetsov V.V., Kravchenko A.N., Krayushkin M.M., Rakitin O.A., Starosotnikov A.M., Fershtat L.L., Shevelev S.A., Shirinian V.Z., Yarovenko V.N.
Russian Chemical Reviews,
2020
3.

Karnoukhova V.A., Baranov V.V., Vologzhanina A.V., Kravchenko A.N., Fedyanin I.V.
CrystEngComm,
2021
4.
Baranov V.V., Vol'khina T.N., Kolotyrkina N.G., Kravchenko A.N.
Mendeleev Communications,
2022
5.

Kölbel M., Menger F.M.
Chemical Communications,
2001
6.

Kang J., Ju H., Jo J.
Supramolecular Chemistry,
2004
7.

Kang J., Jo J., In S.
Tetrahedron Letters,
2004
8.

In S., Kang J.
Tetrahedron Letters,
2005
9.

Moretti F., Poisson G., Marsura A.
Heteroatom Chemistry,
2016
10.

Prokopov A.A., Berlyand A.S., Kazantseva O.N.
Pharmaceutical Chemistry Journal,
2003
11.

Ajami D., Rebek J.
Journal of Organic Chemistry,
2009
12.

13.

Ajami D., Liu L., Rebek Jr. J.
Chemical Society Reviews,
2015
14.

Lizal T., Sindelar V.
Israel Journal of Chemistry,
2017
15.

Sijbesma R.P., Nolte R.J.
Topics in Current Chemistry,
1995
16.

Cotelle Y., Hardouin-Lerouge M., Legoupy S., Alévêque O., Levillain E., Hudhomme P.
Beilstein Journal of Organic Chemistry,
2015
17.

Hardouin–Lerouge M., Hudhomme P., Sallé M.
Chemical Society Reviews,
2011
18.

Assaf K.I., Nau W.M.
Chemical Society Reviews,
2015
19.

Pothinathan S., Muthukannan M., Selvapalam N., Gnanaraj S.C.
International Review of Applied Sciences and Engineering,
2021
20.

Jadhav A.N., Singh S.B., Mane M.V., Kumbhar A.S.
Inorganica Chimica Acta,
2022
21.
(c) M. Shin, M. H. Kim, T. H. Ha, J. Jeon, K.-H. Chung, J. S. Kim and Y. G. Kim, Tetrahedron, 2014, 70, 1617
22.
(d) B. Lee, M. Shin, Y. Seo, M. H. Kim, H. R. Lee, J. S. Kim, K. Chung, D. Yoo and Y. G. Kim, Tetrahedron, 2018, 74, 130
23.
(e) B. Lee, N. Kim, S. Jang, J. H. Park, M. Song, K. Kwon, S. Kim and Y. G. Kim, J. Ind. Eng. Chem., 2022, 113, 360
24.

Shimizu T.
Soil Science and Plant Nutrition,
1987
25.
(a) E. Berdimurodov, A. Kholikov, K. Akbarov, L. Guo, S. Kaya, D. K. Verma, M. Rbaa and O. Dagdag, J. Electroanal. Chem., 2022, 907, 116055; (b) P. Patel, S. Nandi, T. Menapara, A. V. Biradar, R. K. Nagarale, N. H. Khan and R. I. Kureshy, Appl. Catal., A, 2018, 565, 127; (c) V. S. Mane, A. S. Kumbhar and R. P. Thummel, Dalton Trans., 2017, 46, 12901; (d) M. Zarei, H. Sepehrmansourie, M. A. Zolfigol, R. Karamian and S. H. M. Farida, New J. Chem., 2018, 42, 14308; (e) J. P. Bruekers, J. A. A. W. Elemans and R. J. M. Nolte, in Supramolecular Catalysis: New Directions and Developments, eds. P. W. N. M. van Leeuwen and M. Raynal, Wiley-VCH, 2022, ch. 21, pp. 303–320; (f) L. Isaacs and D. Witt, Angew. Chem., Int. Ed., 2002, 41, 1905; (g) W. Sliwa, G. Matusiak and J. Peszke, Heterocycles, 2004, 63, 419; (h) Ya. A. Barsegyan, V. V. Baranov and A. N. Kravchenko, Chem.
26.

Kuptsova A.O., Vinogradova E.E., Kravchenko A.N., Gazieva G.A.
Russian Chemical Bulletin,
2022
27.
Baranov V.V., Barsegyan Y.A., Strelenko Y.A., Karnoukhova V.A., Kravchenko A.N.
Mendeleev Communications,
2020
28.
Baranov V.V., Barsegyan Y.A., Kolotyrkina N.G., Kravchenko A.N.
Mendeleev Communications,
2019
29.

Singh M., Solel E., Keinan E., Reany O.
Chemistry - A European Journal,
2014
30.
![Semithiobambus[6]uril is a transmembrane anion transporter.](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Lang C., Mohite A., Deng X., Yang F., Dong Z., Xu J., Liu J., Keinan E., Reany O.
Chemical Communications,
2017
31.

Reany O., Mohite A., Keinan E.
Israel Journal of Chemistry,
2018
32.

Mondal P., Solel E., Mitra S., Keinan E., Reany O.
Organic Letters,
2019
33.

Verner J., Potáček M.
Open Chemistry,
2004
34.
![A new route to 1,4-disubstituted 5-thioxoperhydroimidazo[4,5-d]imidazol-2-ones](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Verner J., Taraba J., Potáček M.
Tetrahedron Letters,
2002
35.

Cow C.N., Harrison P.H.
Journal of Organic Chemistry,
1997
36.

New access to thioglycolurils by condensation of 4,5-dihydroxyimidazolidin-2-ones(thiones) with HSCN
Baranov V.V., Nelyubina Y.V., Kravchenko A.N., Kolotyrkina N.G., Biriukova K.A.
Tetrahedron Letters,
2015
37.

Baranov V.V., Galochkin A.A., Nelyubina Y.V., Kravchenko A.N., Makhova N.N.
Synthesis,
2020
38.
![Dimroth-type N/S-interchange ofN-aminothioglycolurils in the synthesis of 2-hydrazonoimidazo[4,5-d]thiazolones](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Vinogradova E.E., Gazieva G.A., Izmest'ev A.N., Karnoukhova V.A., Kravchenko A.N.
RSC Advances,
2021
39.
![Efficient method for the synthesis of 1,3-unsubstituted 2-imino-5-oxooctahydroimidazo[4,5-d]imidazolium iodides based on thioglycolurils](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Baranov V.V., Yatsenko E.L., Melnikova E.K., Nelyubina Y.V., Kravchenko A.N.
Chemistry of Heterocyclic Compounds,
2019
40.

Kravchenko A.N., Baranov V.V., Nelyubina Y.V., Gazieva G.A., Svitan’ko I.V.
Russian Chemical Bulletin,
2012
41.

Gazieva G.A., Kravchenko A.N., Lebedev O.V., Strelenko Y.A., Chegaev K.Y.
Russian Chemical Bulletin,
1998
42.

Jia C., Wang Q., Begum R.A., Day V.W., Bowman-James K.
Organic and Biomolecular Chemistry,
2015
43.

Izmest’ev A.N., Anikina L.V., Zanin I.E., Kolotyrkina N.G., Izmalkova E.S., Kravchenko A.N., Gazieva G.A.
New Journal of Chemistry,
2022
44.

Prevost J.R., Kozlova A., Es Saadi B., Yildiz E., Modaffari S., Lambert D.M., Pochet L., Wouters J., Dolušić E., Frédérick R.
Tetrahedron Letters,
2018
45.
Baranov V.V., Vol'khina T.N., Nelyubina Y.V., Kravchenko A.N.
Mendeleev Communications,
2021
46.

Guihéneuf S., Paquin L., Carreaux F., Durieu E., Meijer L., Bazureau J.P.
Organic and Biomolecular Chemistry,
2012