Keywords
2-selenohydantoins
2-thiohydantoins
dehydration
exocyclic C=C bond
isoselenocyanates
isothiocyanates
l-serine
Abstract
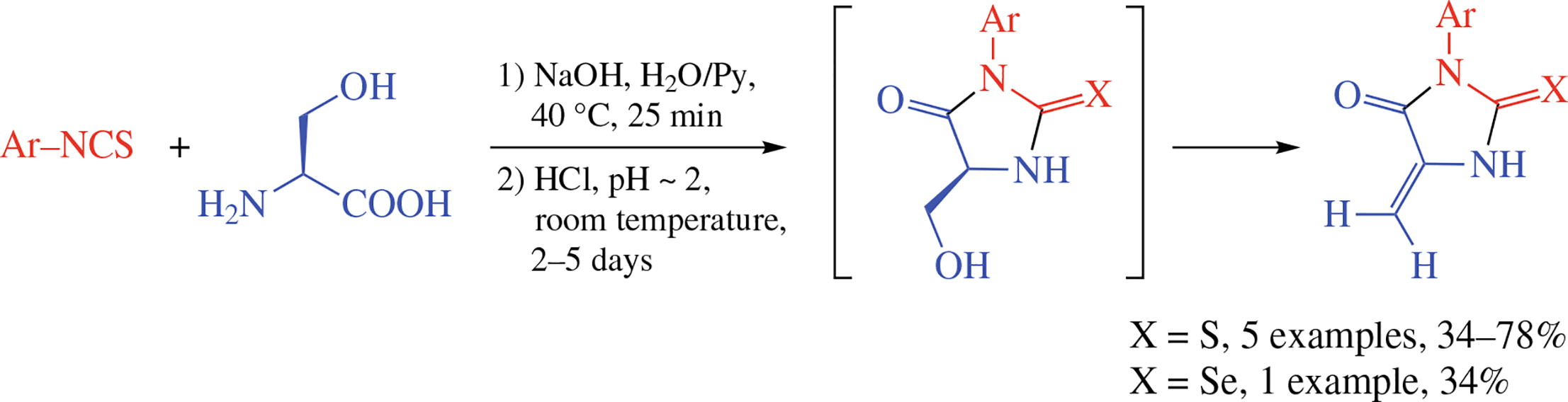
3-Aryl-5-methylidenethiohydantoins and 3-aryl-5-methylidene-selenohydantoins were obtained by one-pot reaction of aryl isothiocyanates or isoselenocyanates with l-serine in NaOH–H2O–Py mixture followed by room temperature HCl-promoted dehydration.
References
1.
10.1016/j.mencom.2022.11.020_b0005
da Silva Bortoleti
Chem.-Biol. Interact.,
2022
2.

Kuptsova A.O., Vinogradova E.E., Kravchenko A.N., Gazieva G.A.
Russian Chemical Bulletin,
2022
3.

Abdellatif K.R., Fadaly W.A., Mostafa Y.A., Zaher D.M., Omar H.A.
Bioorganic Chemistry,
2019
4.

Vanitha U., Elancheran R., Manikandan V., Kabilan S., Krishnasamy K.
Journal of Molecular Structure,
2021
5.

Finko A.V., Skvortsov D.A., Laikov D.N., Averochkin G.M., Dlin E.A., Kalinina M.A., Aladinskiy V.A., Vorobyeva N.S., Mironov A.V., Beloglazkina E.K., Zyk N.V., Ivanenkov Y.A., Majouga A.G.
Bioorganic Chemistry,
2020
6.
![Synthesis of 4,4′-substituted 2,2′-[ethane-1,2-diylbis(selanediyl)]bis(1H-imidazol-5(4H)-ones)](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Finko A.V., Sokolov A.I., Vasilyeva L.A., Skvortsov D.A., Al-Khazraji A.S., Ofitserov E.N., Zyk N.V., Majouga A.G., Beloglazkina E.K.
Russian Chemical Bulletin,
2021
7.

Vasiljeva J., Domracheva I., Arsenyan P.
Chemistry of Heterocyclic Compounds,
2021
8.

Morja M.I., Chauhan P.M., Chikhalia K.H.
Tetrahedron Letters,
2021
9.

Cho S., Kim S., Shin D.
European Journal of Medicinal Chemistry,
2019
10.

Sarma B., Mugesh G.
Chemistry - A European Journal,
2008
11.
![Selenium Containing Heterocycles: Synthesis and Pharmacological Activities of Some New Selenolo[2,3-b]quinoline Derivatives and Related Pentacyclic Systems](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Abdel-Hafez S.H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2010
12.

Suzuki M., Endo M., Shinohara F., Echigo S., Rikiishi H.
Cancer Chemotherapy and Pharmacology,
2009
13.

Santhosh Kumar B., Tiwari S.K., Saikant R., Manoj G., Kunwar A., Sivaram G., Abid Z., Ahmad A., Priyadarsini K.I., Khan A.A.
Journal of Trace Elements in Medicine and Biology,
2010
14.

Aras M., Altaş M., Meydan S., Nacar E., Karcıoğlu M., Ulutaş K.T., Serarslan Y.
International Journal of Neuroscience,
2014
15.

Cao S., Durrani F.A., Tóth K., Rustum Y.M.
British Journal of Cancer,
2014
16.
Kukushkin M.E., Karpov N.A., Shybanov D.E., Zyk N.V., Beloglazkina E.K.
Mendeleev Communications,
2022
17.
10.1016/j.mencom.2022.11.020_b0085
Hassaneen
Phosphorus, Sulfur Silicon Relat. Elem.,
1995
18.
Shybanov D.E., Kukushkin M.E., Tafeenko V.A., Zyk N.V., Grishin Y.K., Roznyatovsky V.A., Beloglazkina E.K.
Mendeleev Communications,
2021
19.

Żesławska E., Oleksyn B.J., Korohoda M.J., Stadnicka K.
Phosphorus, Sulfur and Silicon and the Related Elements,
2003
20.
10.1016/j.mencom.2022.11.020_b0100
Vyhivskyi
Sci.,
2019
21.

Han J., Dong H., Xu Z., Lei J., Wang M.
International Journal of Molecular Sciences,
2013
22.

Evdokimov N.M., Magedov I.V., McBrayer D., Kornienko A.
Bioorganic and Medicinal Chemistry Letters,
2016
23.
Novotortsev V.K., Kukushkin M.E., Tafeenko V.A., Zyk N.V., Beloglazkina E.K.
Mendeleev Communications,
2020
24.

Fraile J.M., Lafuente G., Mayoral J.A., Pallarés A.
Tetrahedron,
2011
25.

L. Gleason J., A. Cernak T.
Heterocycles,
2007
26.

van Dijk M., Postma T.M., Rijkers D.T., Liskamp R.M., van Nostrum C.F., Hennink W.E.
Polymer,
2010
27.

Gupta A., Chauhan V.S.
Biopolymers,
1990
28.
10.1016/j.mencom.2022.11.020_b0140
Ranganathan
J. Chem. Soc., Chem. Commun.,
1992
29.

Stocking E.M., Sanz-Cervera J.F., Williams R.M.
Journal of the American Chemical Society,
2000
30.

Yang Y., Ji M., Lu Z., Jiang M., Huang W., He X.
Synthetic Communications,
2016
31.

Ganiu M.O., Nepal B., Van Houten J.P., Kartika R.
Tetrahedron,
2020
32.
10.1016/j.mencom.2022.11.020_b0160
Andruszkiewicz
Synthesis,
1982
33.

Hjerrild P., Tørring T., Poulsen T.B.
Natural Product Reports,
2020
34.

Fernández-Bolaños J.G., López Ó., Ulgar V., Maya I., Fuentes J.
Tetrahedron Letters,
2004