Keywords
annulated bicyclic isothiourea
colchicine
Cyclization
non-aromatic heterocycles
Podophyllotoxin
thioureas
tubulin
zwitterions
Abstract
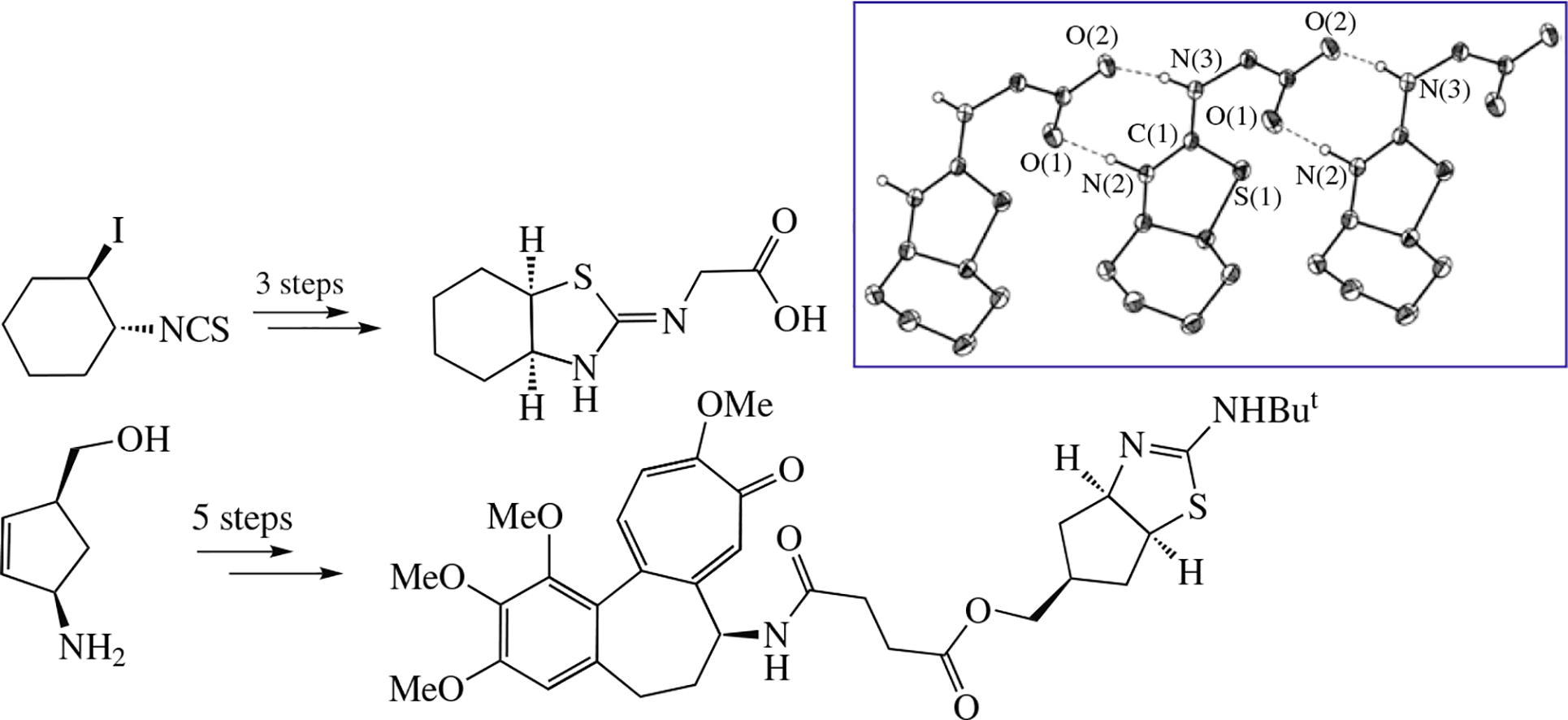
Two bicyclic annulated isothiourea derivatives were synthesized using as a key stage either the reaction of isothiocyanate halide with sodium sulfide or cyclization of unsaturated thiourea in the presence of bromine. X-ray molecular structure of N-[(3aSR,7aRS,Z)-hexahydro-1,3-benzothiazol-2(3H)-ylidene]glycine was determined. The conjugate of colchicine with [(3aR,5S,6aS)-2-(tert-butylamino)-3a,5,6,6a-tetrahydro-4H-cyclopenta[d]thiazol-5-yl]methanol obtained demonstrated pronounced cytotoxic effect on cancer cells.
References
1.

Nurieva E.V., Alexeev A.A., Zefirova O.N.
Chemistry of Heterocyclic Compounds,
2021
2.

Wu G., Xu B., Yang Y., Zhang X., Fang K., Ma T., Wang H., Xue N., Chen M., Guo W., Jia X., Wang P., Lei H.
European Journal of Medicinal Chemistry,
2018
3.

Han H., Lin H., He D., Ren Y., Sun W., Liang L., Du M., Li D., Chu Y., Yang M., Wang X., Yang Y.
Chemistry and Biodiversity,
2018
4.

Hu C., Zhu X., Wang G., Wu X., Han H., Lu G., Qi J., Pang Y., Yang R., Wang X., Yang Y.
Medicinal Chemistry Research,
2017
5.

Larocque K., Ovadje P., Djurdjevic S., Mehdi M., Green J., Pandey S.
PLoS ONE,
2014
6.

Krzywik J., Nasulewicz-Goldeman A., Mozga W., Wietrzyk J., Huczyński A.
ACS Omega,
2021
7.

Zefirova O.N., Nurieva E.V., Wobith B., Schulz S., Zefirov N.A., Kuznetsov S.A.
Pure and Applied Chemistry,
2020
8.

Nicolaus N., Zapke J., Riesterer P., Neudörfl J., Prokop A., Oschkinat H., Schmalz H.
ChemMedChem,
2010
9.
10.1016/j.mencom.2022.11.019_b0045
Thomopoulou
Chem. Lett.,
2015
10.

Nurieva E.V., Zefirov N.A., Temnyakova N.S., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2020
11.

Kim S., Cho S., Kim H., Seok H., Kim S., Kyu Kwon T., Chang J.
Experimental and Molecular Medicine,
2013
12.
Zefirov N.A., Gadert L., Fatkulin A.R., Shibilev V.M., Butov G.M., Mokhov V.M., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
13.

Zefirov N.A., Evteeva Y.A., Wobith B., Kuznetsov S.A., Zefirova O.N.
Structural Chemistry,
2018
14.

Williams R.F., Mumford C.L., Williams G.A., Floyd L.J., Aivaliotis M.J., Martinez R.A., Robinson A.K., Barnes L.D.
Journal of Biological Chemistry,
1985
15.

Passarella D., Peretto B., Blasco y Yepes R., Cappelletti G., Cartelli D., Ronchi C., Snaith J., Fontana G., Danieli B., Borlak J.
European Journal of Medicinal Chemistry,
2010
17.

Finkelstein Y., Aks S.E., Hutson J.R., Juurlink D.N., Nguyen P., Dubnov-Raz G., Pollak U., Koren G., Bentur Y.
Clinical Toxicology,
2010
18.

McLoughlin E.C., O’Boyle N.M.
Pharmaceuticals,
2020
19.

Gracheva I.A., Shchegravina E.S., Schmalz H., Beletskaya I.P., Fedorov A.Y.
Journal of Medicinal Chemistry,
2020
20.

Ghawanmeh A.A., Chong K.F., Sarkar S.M., Bakar M.A., Othaman R., Khalid R.M.
European Journal of Medicinal Chemistry,
2018
21.

Crielaard B.J., van der Wal, Lammers, Le, Hennink, Schiffelers R., Storm, Fens
International Journal of Nanomedicine,
2011
22.

Pallante L., Rocca A., Klejborowska G., Huczynski A., Grasso G., Tuszynski J.A., Deriu M.A.
Frontiers in Chemistry,
2020
23.
Evdokimova A.V., Alexeev A.A., Nurieva E.V., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2021
24.
Nurieva E.V., Trofimova T.P., Alexeev A.A., Proshin A.N., Chesnakova E.A., Grishin Y.K., Lyssenko K.A., Filimonova M.V., Bachurin S.O., Zefirova O.N.
Mendeleev Communications,
2018
25.

Lebeau L., Ducray P., Mioskowski C.
Synthetic Communications,
1997
26.

Bagnato J.D., Eilers A.L., Horton R.A., Grissom C.B.
Journal of Organic Chemistry,
2004
27.
Zefirov N.A., Evteeva Y.A., Krasnoperova A.I., Mamaeva A.V., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Mendeleev Communications,
2020
28.

Zefirov N.A., Mamaeva A.V., Krasnoperova A.I., Evteeva Y.A., Milaeva E.R., Kuznetsov S.A., Zefirova O.N.
Russian Chemical Bulletin,
2021