Keywords
2-(2-hydroxyphenyl)benzoxazole
Antibacterial activity
nickel(ii) complexes
X-ray diffraction analysis
Abstract
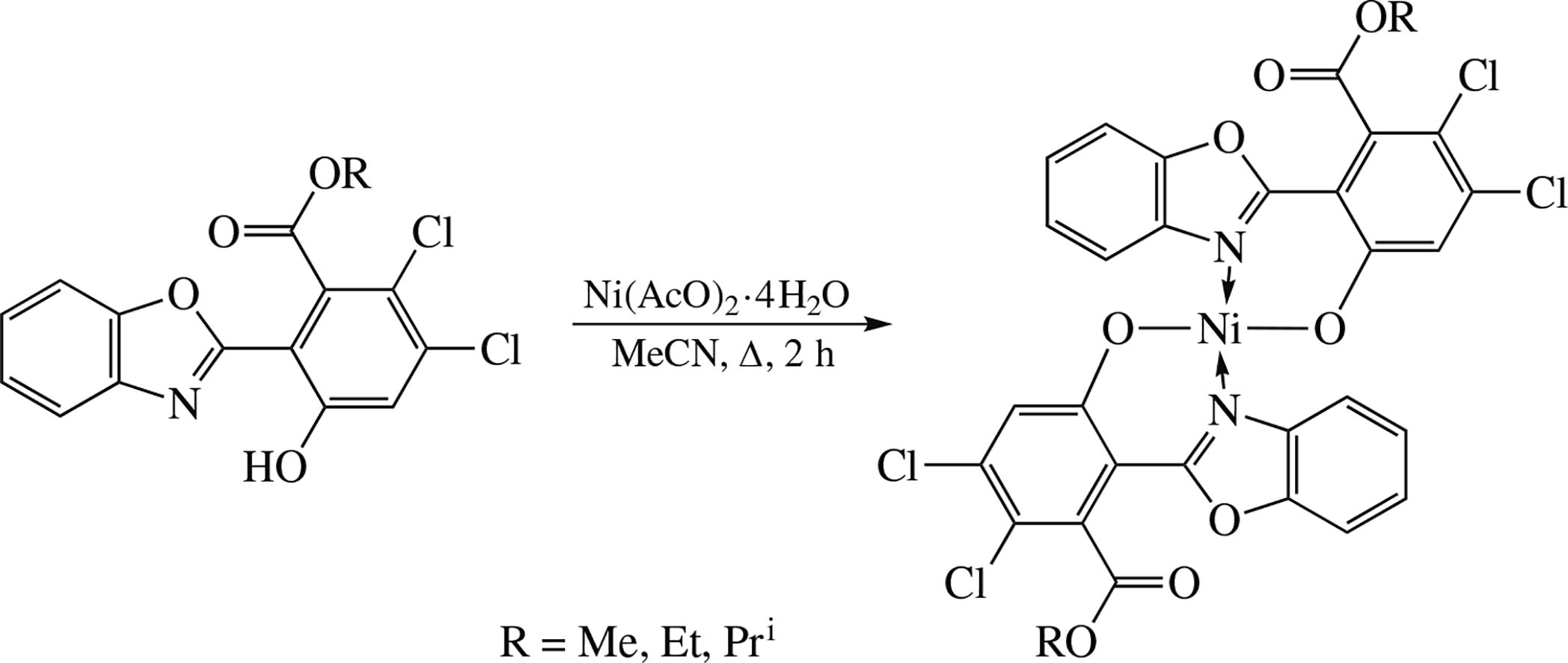
New Niii complexes based on the substituted 2-(2-hydroxy-phenyl)benzoxazole have been synthesized from the corresponding ligands and nickel acetate. The crystal structure of bis[2-(1,3-benzoxazol-2-yl-κN)-4,5-dichloro-3-(methoxycarbonyl)phenolate-κO]nickel(ii) has been determined by X-ray diffraction. The new Niii complexes have been screened for their antibacterial, protistocidal and fungistatic activities
References
1.

Mishra V.R., Ghanavatkar C.W., Mali S.N., Qureshi S.I., Chaudhari H.K., Sekar N.
Computational Biology and Chemistry,
2019
2.

Padalkar V.S., Borse B.N., Gupta V.D., Phatangare K.R., Patil V.S., Umape P.G., Sekar N.
Arabian Journal of Chemistry,
2016
3.

Temiz-Arpaci O., Zeyrek C.T., Arisoy M., Erol M., Celik I., Kaynak-Onurdag F.
Journal of Molecular Structure,
2021
4.
Galieva N.A., Saveliev D.A., Eltsov O.S., Bakulev V.A., Lubec G., Xing J., Fan Z., Beryozkina T.V.
Mendeleev Communications,
2021
5.
![Synthesis, antimicrobial activity, pharmacophore analysis of some new 2-(substitutedphenyl/benzyl)-5-[(2-benzofuryl)carboxamido]benzoxazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Alper-Hayta S., Arisoy M., Temiz-Arpaci Ö., Yildiz I., Aki E., Özkan S., Kaynak F.
European Journal of Medicinal Chemistry,
2008
6.

Ertan T., Yildiz I., Tekiner-Gulbas B., Bolelli K., Temiz-Arpaci O., Ozkan S., Kaynak F., Yalcin I., Aki E.
European Journal of Medicinal Chemistry,
2009
7.
10.1016/j.mencom.2022.11.018_b0035
Tas¸ci
Indian J. Chem., Sect. B: Org. Chem. Incl. Med. Chem.,
2018
8.

RIDA S., ASHOUR F., ELHAWASH S., ELSEMARY M., BADR M., SHALABY M.
European Journal of Medicinal Chemistry,
2005
9.

Jauhari P.K., Bhavani A., Varalwar S., Singhal K., Raj P.
Medicinal Chemistry Research,
2008
10.

Kumar D., Jacob M.R., Reynolds M.B., Kerwin S.M.
Bioorganic and Medicinal Chemistry,
2002
11.
![Synthesis and preliminary evaluation of 2-substituted-1,3-benzoxazole and 3-[(3-substituted)propyl]-1,3-benzoxazol-2(3H)-one derivatives as potent anticancer agents](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Murty M.S., Ram K.R., Rao R.V., Yadav J.S., Rao J.V., Cheriyan V.T., Anto R.J.
Medicinal Chemistry Research,
2010
12.

Kim D., Won H.Y., Hwang E.S., Kim Y., Choo H.P.
Bioorganic and Medicinal Chemistry,
2017
13.

Dong J.Q., Liu J., Smith P.C.
Biochemical Pharmacology,
2005
14.

Abdelgawad M.A., Bakr R.B., Omar H.A.
Bioorganic Chemistry,
2017
15.
![Synthesis and Antimicrobial Activity of Some 5-[2-(Morpholin-4-yl)acetamido] and/or 5-[2-(4-Substituted piperazin-1-yl)acetamido]-2-(p-substituted phenyl)benzoxazoles](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Temiz-Arpacı Ö., Özdemir A., Yalçın İ., Yıldız İ., Akı-Şener E., Altanlar N.
Archiv der Pharmazie,
2005
16.

Deng B., Cullen M.D., Zhou Z., Hartman T.L., Buckheit R.W., Pannecouque C., Clercq E.D., Fanwick P.E., Cushman M.
Bioorganic and Medicinal Chemistry,
2006
17.

Kuroda K., Tsuyumine S., Kodama T.
Organic Process Research and Development,
2016
18.

19.
10.1016/j.mencom.2022.11.018_b0095
Tanaka
J. Mater. Chem.,
1999
20.
![Electron and Hole Mobility in Vacuum Deposited Organic Thin Films of Bis[2-(2-hydroxyphenyl)benzoxazolate]zinc and Its Derivatives](/storage/images/resized/yNSijlgQghQF53VZuyFLA30CKDe4j3HK74Vtpnxa_small_thumb.webp)
Yasuda T., Yamaguchi Y., Fujita K., Tsutsui T.
Chemistry Letters,
2003
21.

Kumar A., Palai A.K., Srivastava R., Kadyan P.S., Kamalasanan M.N., Singh I.
Journal of Organometallic Chemistry,
2014
22.
![Synthesis and blue electroluminescent properties of zinc (II) [2-(2-hydroxyphenyl)benzoxazole]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kim W.S., You J.M., Lee B., Jang Y., Kim D., Kwon Y.
Thin Solid Films,
2007
23.

Zhang G., Zhao Y., Peng B., Li Z., Xu C., Liu Y., Zhang C., Voelcker N.H., Li L., Huang W.
Journal of Materials Chemistry B,
2019
24.

Jiang J., Tang X., Dou W., Zhang H., Liu W., Wang C., Zheng J.
Journal of Inorganic Biochemistry,
2010
25.

Bouchoucha A., Terbouche A., Bourouina A., Djebbar S.
Inorganica Chimica Acta,
2014
26.

Samota M.K., Seth G.
Heteroatom Chemistry,
2010
27.

Watanabe G., Sekiya H., Tamai E., Saijo R., Uno H., Mori S., Tanaka T., Maki J., Kawase M.
Chemical and Pharmaceutical Bulletin,
2018
28.
SPENGLER G., KINCSES A., RÁCZ B., VARGA B., WATANABE G., SAIJO R., SEKIYA H., TAMAI E., MAKI J., MOLNÁR J., KAWASE M.
Anticancer Research,
2018
29.

Kincses A., Szabó S., Rácz B., Szemerédi N., Watanabe G., Saijo R., Sekiya H., Tamai E., Molnár J., Kawase M., Spengler G.
Antibiotics,
2020
30.

Vetrova E.V., Tupaeva I.O., Sayapin Y.A., Gusakov E.A., Nikolaevskii S.A., Demidov O.P., Minkin V.I., Metelitsa A.V.
Dyes and Pigments,
2020
31.

Sayapin Y.A., Tupaeva I.O., Kolodina A.A., Gusakov E.A., Komissarov V.N., Dorogan I.V., Makarova N.I., Metelitsa A.V., Tkachev V.V., Aldoshin S.M., Minkin V.I.
Beilstein Journal of Organic Chemistry,
2015
32.

Tupaeva I.O., Sayapin Y.A., Bang Z.N., Komissarov V.N., Tkachev V.V., Shilov G.V., Aldoshin S.M., Minkin V.I.
Russian Chemical Bulletin,
2013
33.

Layek S., Agrahari B., Kumar A., Dege N., Pathak D.D.
Inorganica Chimica Acta,
2020
34.
CrysAlisPro, version 1.171.38.41, Rigaku Oxford Diffraction, 2015, https://www.rigaku.com/en/products/smc/crysalis.
35.

Zhao H., Schuck P.
Acta Crystallographica Section D Biological Crystallography,
2015
36.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009