Keywords
Bismuth
copper
halide complexes
semiconductors
silver
Abstract
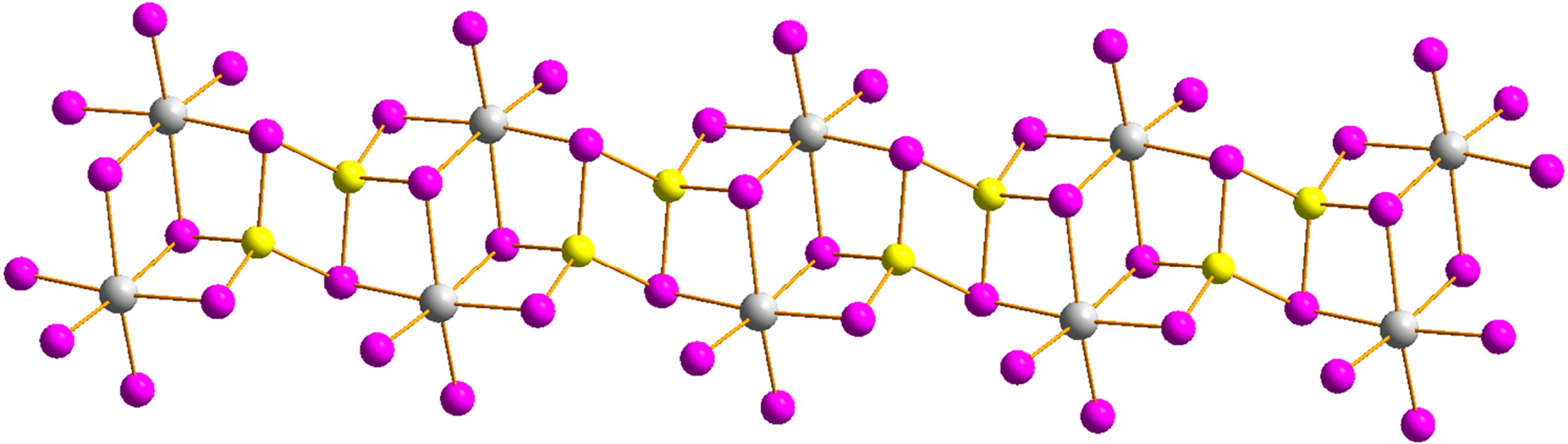
Reactions of BiI3, copper or silver iodide and 4-(dimethyl-amino)-1-methylpyridinium iodide [(Me-DMAP)I] result in formation of heterometallic complexes (MeDMAP)2{[Bi2Cu2I10]} and (MeDMAP)2{[Bi2Ag2I10]}. Their crystal structures, thermal stability and optical properties have been studied. Optical band gaps calculated from diffuse reflectance spectroscopy data are 1.81 and 2.06 eV for copper- and silver-containing complexes, respectively.
References
1.

Wu L., Wu X., Chen L.
Coordination Chemistry Reviews,
2009
2.

Buikin P.A., Rudenko A.Y., Ilyukhin A.B., Simonenko N.P., Yorov K.E., Kotov V.Y.
Russian Journal of Coordination Chemistry/Koordinatsionnaya Khimiya,
2020
3.
10.1016/j.mencom.2022.11.015_b0015
Buikin
Russ. J. Inorg. Chem.,
2021
4.

Buikin P.A., Rudenko A.Y., Ilyukhin A.B., Kotov V.Y.
Russian Journal of Inorganic Chemistry,
2021
5.
![Synthesis and structure of bismuth complexes [Ph3MeP] 2 + [BiI3.5Br1.5(C5H5N)]2− · C5H5N, [Ph4Bi] 4 + [Bi4I16]4− · 2Me2C=O, and [Ph3(iso-Am)P] 4 + [Bi8I28]4− · 2Me2C=O](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Sharutin V.V., Egorova I.V., Klepikov N.N., Boyarkina E.A., Sharutina O.K.
Russian Journal of Inorganic Chemistry,
2009
6.

Wheaton A.M., Streep M.E., Ohlhaver C.M., Nicholas A.D., Barnes F.H., Patterson H.H., Pike R.D.
ACS Omega,
2018
7.
![Synthesis and characterization of (RPh3P)3[Bi3I12] (R = Me, Ph) iodobismuthate complexes for photocatalytic degradation of organic pollutants](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Moyet M.A., Kanan S.M., Varney H.M., Abu-Farha N., Gold D.B., Lain W.J., Pike R.D., Patterson H.H.
Research on Chemical Intermediates,
2019
8.

Ahern J.C., Nicholas A.D., Kelly A.W., Chan B., Pike R.D., Patterson H.H.
Inorganica Chimica Acta,
2018
9.

Dehnhardt N., Paneth H., Hecht N., Heine J.
Inorganic Chemistry,
2020
10.
![A step closer to the binary: the 1∞[Bi6I20]2−anion](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Heine J.
Dalton Transactions,
2015
11.

Shestimerova T.A., Golubev N.A., Yelavik N.A., Bykov M.A., Grigorieva A.V., Wei Z., Dikarev E.V., Shevelkov A.V.
Crystal Growth and Design,
2018
12.

Yelovik N.A., Mironov A.V., Bykov M.A., Kuznetsov A.N., Grigorieva A.V., Wei Z., Dikarev E.V., Shevelkov A.V.
Inorganic Chemistry,
2016
13.

Shestimerova T.A., Golubev N.A., Mironov A.V., Bykov M.A., Shevelkov A.V.
Russian Chemical Bulletin,
2018
14.
![Diimidazolium Halobismuthates [Dim]2[Bi2X10] (X = Cl–, Br–, or I–): A New Class of Thermochromic and Photoluminescent Materials](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
García-Fernández A., Marcos-Cives I., Platas-Iglesias C., Castro-García S., Vázquez-García D., Fernández A., Sánchez-Andújar M.
Inorganic Chemistry,
2018
15.

Lin R., Xu G., Wang M., Lu G., Li P., Guo G.
Inorganic Chemistry,
2013
16.

Toma O., Mercier N., Botta C.
European Journal of Inorganic Chemistry,
2013
17.
![Specific heat anomaly in ferroelectric: Bis(imidazolium) pentachloroantimonate(III) (C3 N2 H5)2[SbCl5]](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Przesławski J., Piecha-Bisiorek A., Jakubas R.
Journal of Molecular Structure,
2016
18.

Mencel K., Kinzhybalo V., Jakubas R., Zaręba J.K., Szklarz P., Durlak P., Drozd M., Piecha-Bisiorek A.
Chemistry of Materials,
2021
19.
![Ferroelectricity in Ethylammonium Bismuth-Based Organic–Inorganic Hybrid: (C2H5NH3)2[BiBr5]](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Jakubas R., Ga̧gor A., Winiarski M.J., Ptak M., Piecha-Bisiorek A., Ciżman A.
Inorganic Chemistry,
2019
20.
10.1016/j.mencom.2022.11.015_b0100
Szklarz
Dalton Trans.,
2022
21.
![Raman-Infrared spectroscopy, thermal behaviour, dielectric, and UV-fluorescence studies of [C6H5NH3]3BiCl6·3H2O](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
El-Adel L., Ouasri A., Rhandour A., Hajji L.
Solid State Communications,
2021
22.

Marwitz A.C., Nicholas A.D., Breuer L.M., Bertke J.A., Knope K.E.
Inorganic Chemistry,
2021
23.

Jin J., Lin Y., Lin L., Xiao C., Song Y., Shen N., Gong L., Zhang Z., Du K., Huang X.
CrystEngComm,
2021
24.

Jin J., Shen N., Wang Z., Peng Y., Huang X.
Coordination Chemistry Reviews,
2021
25.

Benin B.M., McCall K.M., Wörle M., Morad V., Aebli M., Yakunin S., Shynkarenko Y., Kovalenko M.V.
Angewandte Chemie - International Edition,
2020
26.

Shlenskaya N., Belich N.A., Grätzel M., Goodilin E.A., Tarasov A.B.
Journal of Materials Chemistry A,
2018
27.
Grishko A.Y., Zharenova E.A., Goodilin E.A., Tarasov A.B.
Mendeleev Communications,
2021
28.
Marchenko E.I., Fateev S.A., Petrov A.A., Goodilin E.A., Tarasov A.B.
Mendeleev Communications,
2020
29.

Tutantsev A.S., Udalova N.N., Fateev S.A., Petrov A.A., Chengyuan W., Maksimov E.G., Goodilin E.A., Tarasov A.B.
Journal of Physical Chemistry C,
2020
30.

Petrov A.A., Fateev S.A., Khrustalev V.N., Li Y., Dorovatovskii P.V., Zubavichus Y.V., Goodilin E.A., Tarasov A.B.
Chemistry of Materials,
2020
31.

Beyond methylammonium lead iodide: prospects for the emergent field of ns2containing solar absorbers
Ganose A.M., Savory C.N., Scanlon D.O.
Chemical Communications,
2017
32.

Liu G., Zhang X., Wang H., Xu H., Wang Z., Meng X., Dong Y., Zhao R., Li C.
Dalton Transactions,
2017
33.

Huang L., Zhou J.
Inorganic Chemistry,
2020
34.

Hao P., Qiao Y., Yu T., Shen J., Dai D., Fu Y.
RSC Advances,
2016
35.

Artem′ev A.V., Rakhmanova M.I., Brylev K.A., Bagryanskaya I.Y.
Journal of Structural Chemistry,
2020
36.

Deng M., Wu Y., Rao P., Zhang W., Wang R., Doert T.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2018
37.
10.1016/j.mencom.2022.11.015_b0185
Jiang
Opt. Mater. (Amsterdam, Neth.),
2020
38.

Yang L., Zhou J., Fu L.
Dyes and Pigments,
2020
39.

Artem'ev A.V., Berezin A.S., Taidakov I.V., Bagryanskaya I.Y.
Inorganic Chemistry Frontiers,
2020
40.

Xu Z., Wu H., Li D., Wu W., Li L., Luo J.
Journal of Materials Chemistry C,
2021
41.

Yao Y., Kou B., Peng Y., Wu Z., Li L., Wang S., Zhang X., Liu X., Luo J.
Chemical Communications,
2020
42.
![CuBi7I19(C4H8O3H)3(C4H8O3H2), a Novel Complex Bismuth Iodide Containing One-Dimensional [CuBi5I19]3- Chains](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Feldmann C.
Inorganic Chemistry,
2001
43.

Bi L., Hu Y., Li M., Hu T., Zhang H., Yin X., Que W., Lassoued M.S., Zheng Y.
Journal of Materials Chemistry A,
2019
44.

Fu D., Wu S., Liu Y., Yao Y., He Y., Zhang X.
Inorganic Chemistry Frontiers,
2021
45.

Bi L., Hu T., Li M., Ling B., Lassoued M.S., Hu Y., Wu Z., Zhou G., Zheng Y.
Journal of Materials Chemistry A,
2020
46.
![Silver Iodobismuthates: Syntheses, Structures, Properties, and Theoretical Studies of [Bi2Ag2I102-]n and [Bi4Ag2I162-]n](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chai W., Wu L., Li J., Chen L.
Inorganic Chemistry,
2007
47.

Chai W., Wu L., Li J., Chen L.
Inorganic Chemistry,
2007
48.

Kelly A.W., Wheaton A.M., Nicholas A.D., Barnes F.H., Patterson H.H., Pike R.D.
European Journal of Inorganic Chemistry,
2017
49.

Dehnhardt N., Borkowski H., Schepp J., Tonner R., Heine J.
Inorganic Chemistry,
2017
50.

Möbs J., Heine J.
Inorganic Chemistry,
2019
51.

Dehnhardt N., Klement P., Chatterjee S., Heine J.
Inorganic Chemistry,
2019
52.

Möbs J., Gerhard M., Heine J.
Dalton Transactions,
2020
53.

The influence of copper on the optical band gap of heterometallic iodido antimonates and bismuthates
Möbs J., Luy J., Shlyaykher A., Tonner R., Heine J.
Dalton Transactions,
2021
54.

Dehnhardt N., Luy J., Klement P., Schipplick L., Chatterjee S., Tonner R., Heine J.
Angewandte Chemie - International Edition,
2021
55.

Zhao H., Schuck P.
Acta Crystallographica Section D Biological Crystallography,
2015
56.

Hübschle C.B., Sheldrick G.M., Dittrich B.
Journal of Applied Crystallography,
2011