Keywords
2-aminoglutarimide
Cereblon
E3 ubiquitin ligase
immunomodulatory drugs
isocyanides
oxo acids
Ugi reaction
Abstract
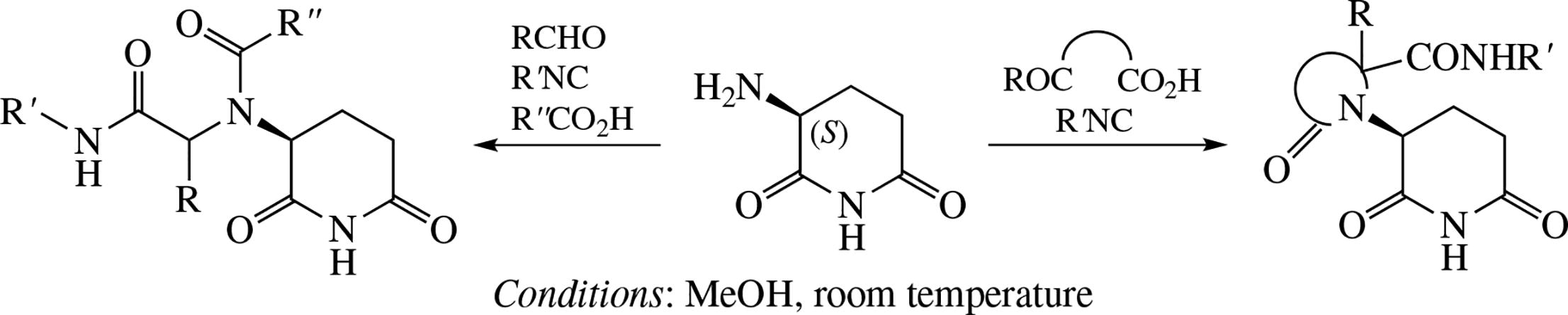
The glutarimide moiety, common in many immuno-modulatory drugs, was decorated with lactam and diamide side chains via two variants of the Ugi reaction, namely, with isocyanide, aldehyde and acid or with isocyanide and oxo acid. The resulting diastereomerically pure compounds were evaluated for their affinity towards the E3 ubiquitin ligase substrate receptor Cereblon.
References
1.

Quach H., Ritchie D., Stewart A.K., Neeson P., Harrison S., Smyth M.J., Prince H.M.
Leukemia,
2009
2.

Fischer E.S., Böhm K., Lydeard J.R., Yang H., Stadler M.B., Cavadini S., Nagel J., Serluca F., Acker V., Lingaraju G.M., Tichkule R.B., Schebesta M., Forrester W.C., Schirle M., Hassiepen U., et. al.
Nature,
2014
3.

Krönke J., Udeshi N.D., Narla A., Grauman P., Hurst S.N., McConkey M., Svinkina T., Heckl D., Comer E., Li X., Ciarlo C., Hartman E., Munshi N., Schenone M., Schreiber S.L., et. al.
Science,
2014
4.

Coll-Martínez B., Delgado A., Crosas B.
Molecules,
2020
5.

Scheepstra M., Hekking K.F., van Hijfte L., Folmer R.H.
Computational and Structural Biotechnology Journal,
2019
6.

Békés M., Langley D.R., Crews C.M.
Nature Reviews Drug Discovery,
2022
7.

Krasavin M., Bubyrev A., Kazantsev A., Heim C., Maiwald S., Zhukovsky D., Dar’in D., Hartmann M.D., Bunev A.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2022
8.

Kazantsev A., Krasavin M.
Expert Opinion on Therapeutic Patents,
2021
9.

Bricelj A., Steinebach C., Kuchta R., Gütschow M., Sosič I.
Frontiers in Chemistry,
2021
10.

Alcock L.J., Chang Y., Jarusiewicz J.A., Actis M., Nithianantham S., Mayasundari A., Min J., Maxwell D., Hunt J., Smart B., Yang J.J., Nishiguchi G., Fischer M., Mullighan C.G., Rankovic Z., et. al.
ACS Medicinal Chemistry Letters,
2022
11.

Boichenko I., Bär K., Deiss S., Heim C., Albrecht R., Lupas A.N., Hernandez Alvarez B., Hartmann M.D.
ACS Omega,
2018
12.
10.1016/j.mencom.2022.11.013_b0060
Ugi
Angew. Chem.,
1959
13.
10.1016/j.mencom.2022.11.013_b0065
Tsaloev
Tetrahedron Lett.,
1800
14.

Hu S., Yuan L., Yan H., Li Z.
Bioorganic and Medicinal Chemistry Letters,
2017
15.
Krasavin M., Nikulnikov M.M.
Mendeleev Communications,
2012
16.

Maiwald S., Heim C., Hernandez Alvarez B., Hartmann M.D.
ACS Medicinal Chemistry Letters,
2020
17.

Zorba A., Nguyen C., Xu Y., Starr J., Borzilleri K., Smith J., Zhu H., Farley K.A., Ding W., Schiemer J., Feng X., Chang J.S., Uccello D.P., Young J.A., Garcia-Irrizary C.N., et. al.
Proceedings of the National Academy of Sciences of the United States of America,
2018