Keywords
aromaticity degree
bis(diazolo)pyrazines
molecular magnet
open shell aromaticity
quantum chemical calculations
radical anions
Abstract
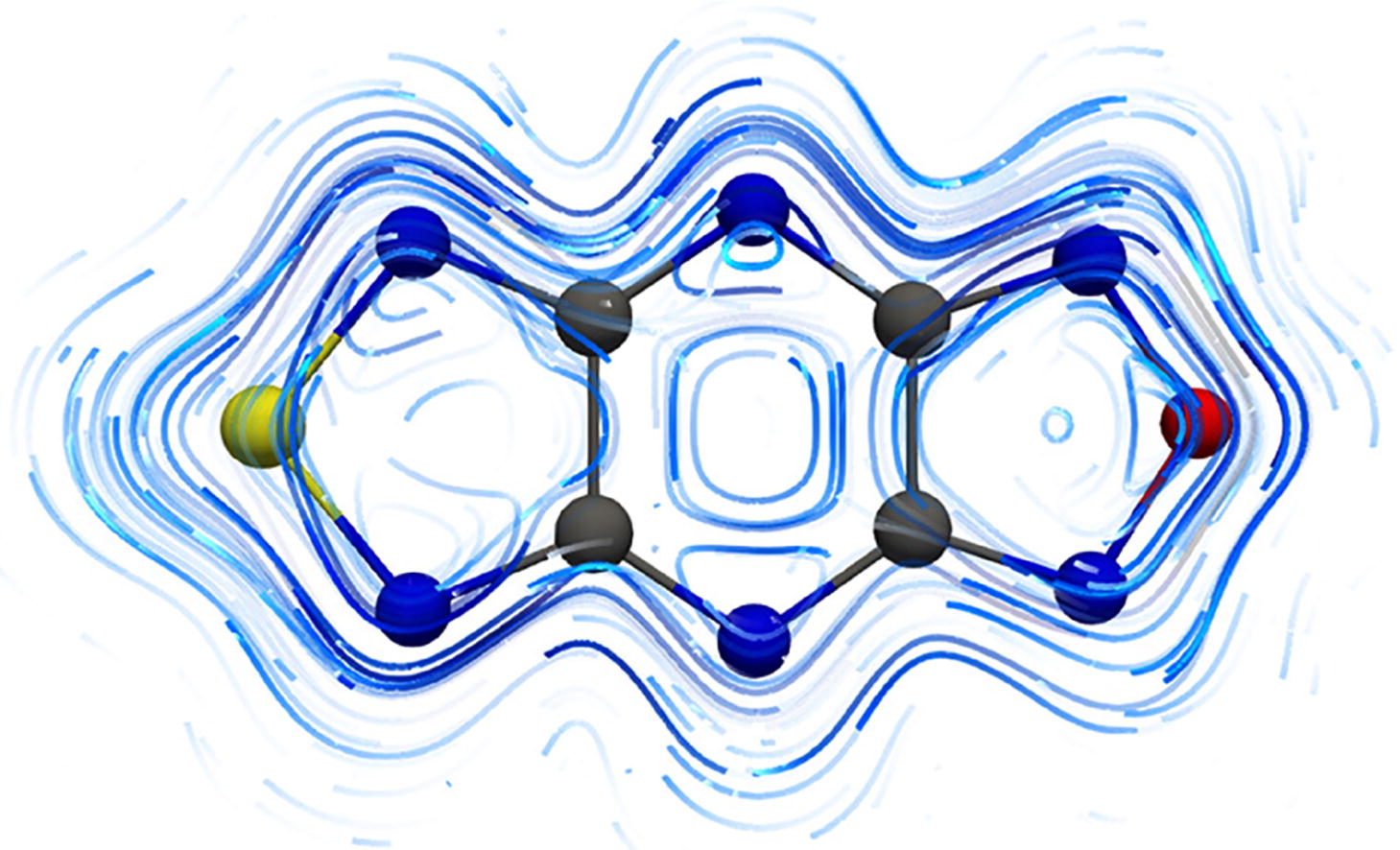
The aromaticity of a series of heterocyclic radical anions of bis(diazolo)pyrazine type, X(CN)2N2(CN)2Y (X, Y = O, S, Se, Te) was explored by the methods of electron density of delocalized bonds (EDDB) and gauge-included magnetically induced currents (GIMIC). The existence of T-aromaticity that encloses the entire molecule, which was due to delocalization of seven β-electrons, was shown. The degree of aromaticity depends on the nature of the X(Y) heteroatom and varies in the series S > O > Se > Te.
References
1.
10.1016/j.mencom.2022.11.008_b0005
Stable Radicals: Fundamentals and Applied Aspects of Odd-Electron Compounds,
2010
2.
10.1016/j.mencom.2022.11.008_b0010
World Scientific Reference on Spin in Organics,
2018
3.

4.
10.1016/j.mencom.2022.11.008_b0020
Inoue
in p-Electron Magnetism: From Molecules to Magnetic Materials, ed,
2001
5.
Organic Redox Systems: Synthesis, Properties, and Applications, ed. T. Nishinaga, Wiley, Hoboken, 2016
6.

7.

Yonekuta Y., Susuki K., Oyaizu K., Honda K., Hiroyuki Nishide*
Journal of the American Chemical Society,
2007
8.

Oyaizu K., Nishide H.
Advanced Materials,
2009
9.

Lee J., Lee E., Kim S., Bang G.S., Shultz D.A., Schmidt R.D., Forbes M.D., Lee H.
Angewandte Chemie - International Edition,
2011
10.

11.

Coronado E.
Nature Reviews Materials,
2019
12.

Han W., Pi K., McCreary K.M., Li Y., Wong J.J., Swartz A.G., Kawakami R.K.
Physical Review Letters,
2010
13.

Gaudenzi R., Burzurí E., Reta D., Moreira I.D., Bromley S.T., Rovira C., Veciana J., van der Zant H.S.
Nano Letters,
2016
14.

Herrmann C., Solomon G.C., Ratner M.A.
Journal of the American Chemical Society,
2010
15.
10.1016/j.mencom.2022.11.008_b0075
Hu
Beilstein J. Nanotechnol.,
1919
16.
Ovcharenko V.I., Sheremetev A.B., Strizhenko K.V., Fokin S.V., Romanenko G.V., Bogomyakov A.S., Morozov V.A., Syroeshkin M.A., Kozmenkova A.Y., Lalov A.V., Egorov M.P.
Mendeleev Communications,
2021
17.
![Comparative Theoretical Studies on a Series of Novel Energetic Salts Composed of 4,8-Dihydrodifurazano[3,4-b,e]pyrazine-based Anions and Ammonium-based Cations](/storage/images/resized/MjH1ITP7lMYGxeqUZfkt2BnVLgjkk413jwBV97XX_small_thumb.webp)
Duan B., Liu N., Wang B., Lu X., Mo H.
Molecules,
2019
18.
![4H,8H-bis(1,2,5-oxadiazolo)[3,4-b:3',4'-e]Pyrazine](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Starchenkov I.B., Andrianov V.G.
Chemistry of Heterocyclic Compounds,
1996
19.
10.1016/j.mencom.2022.11.008_b0095
Li
Cryst. Growth Des.,
1896
20.

Konstantinova L., Knyazeva E., Rakitin O.
Molecules,
2015