Keywords
3-benzylamino-2
4-enyne
benzylamine
tetrahydroindole
tetrahydroindolyldiyne
tetrahydroindolylpyridine
Abstract
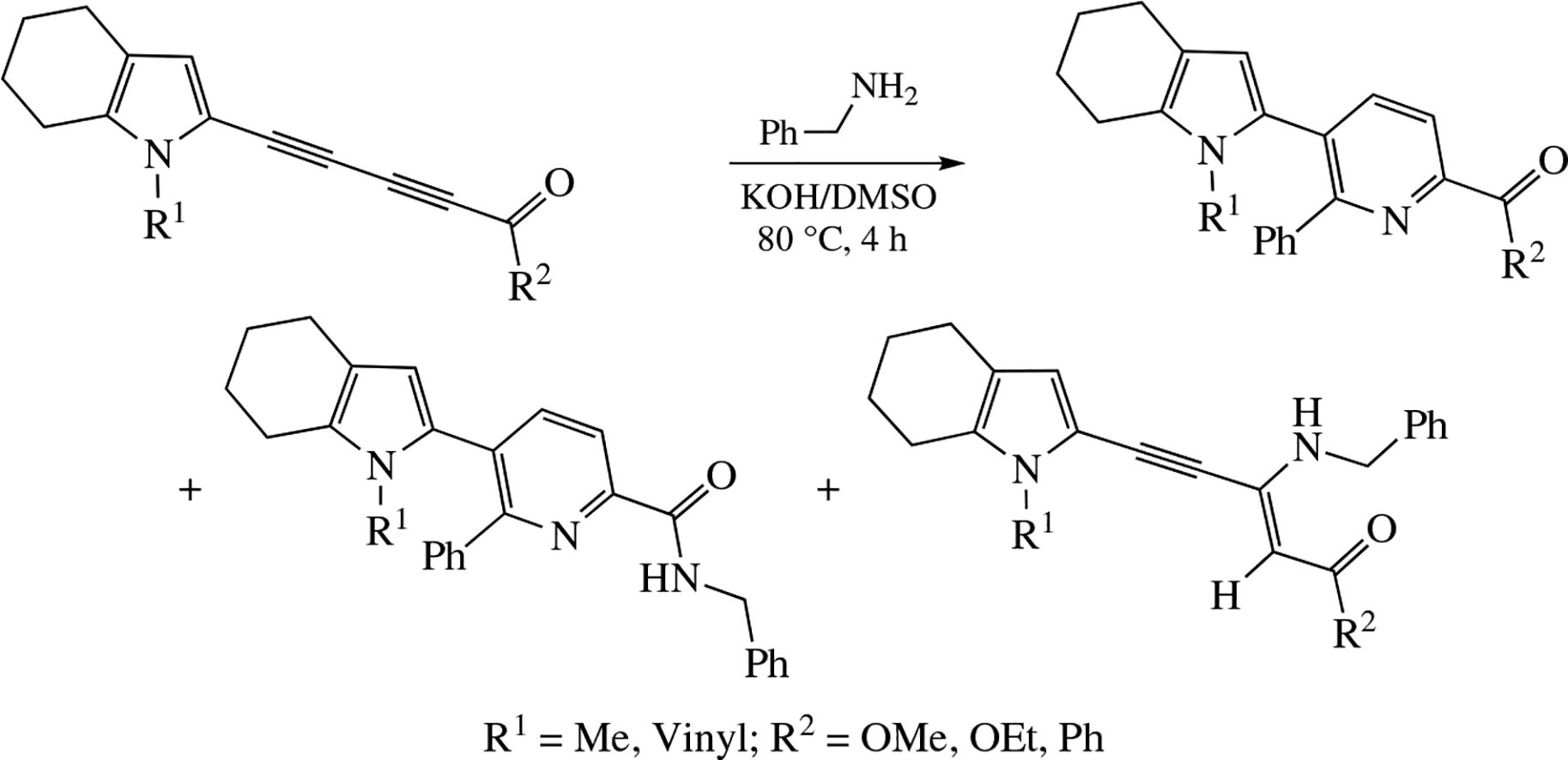
1-Acyl-4-tetrahydroindolyldiynes (available from cross-coupling of 4,5,6,7-tetrahydroindole with 1-acyl-4-bromo-1,3-diynes) undergo oxidative [4+2]-cycloaddition with benzylamine in KOH/DMSO or K3PO4/DMSO systems (80°C, 4 h) to afford 2-acyl-6-phenyl-5-tetrahydroindolyl-pyridines in ∼40% yields. Non-catalyzed reaction of the same reactants in boiling MeOH (4 h) or in DABCO/MeCN system (reflux, 1 h) gives 3-benzylaminoalk-2-en-4-ynones in ∼40% yields.
References
1.

Tomilin D.N., Pigulski B., Gulia N., Arendt A., Sobenina L.N., Mikhaleva A.I., Szafert S., Trofimov B.A.
RSC Advances,
2015
2.

3.
(b) E. Abele and E. Lukevics, The Chemistry of Hydroxylamines, Oximes and Hydroxamic Acids, eds. Z. Rappoport and J. F. Liebman, Wiley, Chichester, 2008
5.

Trofimov B.A., Mikhaleva A.I., Schmidt E.Y., Sobenina L.N.
Advances in Heterocyclic Chemistry,
2010
6.
(e) J. Bergman and T. Janosik, Modern Heterocyclic Chemistry, eds. J. Alvarez-Builla J. J. Vaquero and J. Barluenga, Wiley, Weinheim, 2011, vol. 4, p. 288
7.
(f) Name Reactions in Heterocyclic Chemistry II, ed. J. J. Li, Wiley, Hoboken, 2011, pp. 72-82.
8.

Younis F.M., Krieck S., Görls H., Westerhausen M.
Dalton Transactions,
2016
9.

Younis F.M., Krieck S., Al-Shboul T.M., Görls H., Westerhausen M.
Inorganic Chemistry,
2016
10.

Zheng S., Shen Z.
Tetrahedron Letters,
2010
11.

Zheng Q., Hua R., Jiang J., Zhang L.
Tetrahedron,
2014
12.

You X., Xie X., Sun R., Chen H., Li S., Liu Y.
Organic Chemistry Frontiers,
2014
13.

Wang L., Yu X., Feng X., Bao M.
Journal of Organic Chemistry,
2013
14.
(b) X. Yu, N. Huang, X. Feng, Y. Yamamoto and M. Bao, Synthesis, 2014, 46, 2422
15.

Bassaco M.M., Fortes M.P., Kaufman T.S., Silveira C.C.
RSC Advances,
2015
16.
(d) S. Hariprasad and H. T. Srinivasa, Liquid Crystals, 2015, 42, 1612
17.

Ötvös S.B., Georgiádes Á., Ozsvár D., Fülöp F.
RSC Advances,
2019
18.

Meng Y., Zhang T., Gong X., Zhang M., Zhu C.
Tetrahedron Letters,
2019
19.

Wang L., Yu X., Feng X., Bao M.
Organic Letters,
2012
20.

Zhou X., Guo D., Jiang Y., Gong D., Zhao X., Zhou L.
Tetrahedron Letters,
2017
21.

Guo M., Chen B., Zhu Q., Jin H., Peng Q., Kang Y.
Molecules,
2017
22.

Zhang L., Zhao M., Zhao X.
Chemical Communications,
2015
23.
(b) X. Zhao and L. Zhang, Patent CN 104725322A, 2015.
24.

Sun H., Wu X., Hua R.
Tetrahedron Letters,
2011
25.

Choi J., Park K., Lim J., Jung H.M., Lee S.
Asian Journal of Organic Chemistry,
2015
26.

Li Y., Qiu S., Fan L., Yin G.
ChemCatChem,
2020
27.
10.1016/j.mencom.2022.11.007_b0050
Zheng
Curr. Org. Synth.,
2013
28.

Andrade C.B., Carvalho D.B., Trefzger O.S., Kassab N.M., Guerrero P.G., Barbosa S.L., Shiguemoto C.Y., Baroni A.C.
European Journal of Organic Chemistry,
2018
29.

Huerta G., Fomina L., Rumsh L., Zolotukhin M.G.
Polymer Bulletin,
2006