Keywords
1,2,4-triazines
2-aminopyridines
6-amino-2,2'-bipyridines
arylhydrazines
autoclave processes
aza-Diels-Alder reaction
ipso-substitution
Abstract
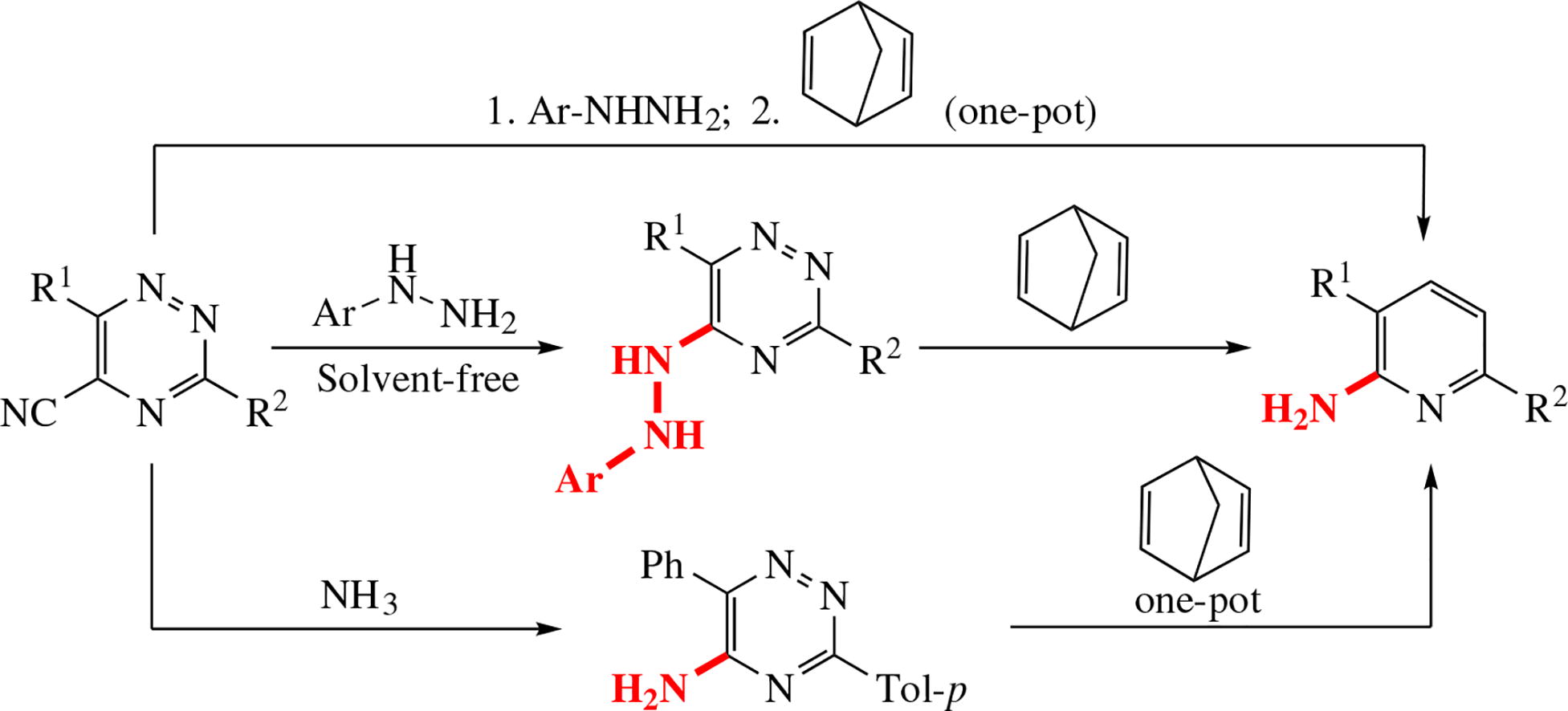
Reaction between 5-cyano-3,6-di(het)aryl-1,2,4-triazines and arylhydrazines with the following aza-Diels-Alder autoclave reaction affords hardly available 2-amino-3,6-di(het)aryl-pyridines in up to 67% yield after two steps and in 75% yield for the one-pot way. The compounds obtained can be promising for medicinal chemistry.
References
1.

Pfu¨ller O.C., Sauer J.
Tetrahedron Letters,
1998
2.

Kozhevnikov V.N., Kozhevnikov D.N., Nikitina T.V., Rusinov V.L., Chupakhin O.N., Zabel M., König B.
Journal of Organic Chemistry,
2003
3.
Savchuk M.I., Krinochkin A.P., Rammohan A., Khasanov A.F., Kopchuk D.S., Egorov I.N., Santra S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2020
4.

Wang S., Guo W., Wen L., Li M.
RSC Advances,
2014
5.
10.1016/j.mencom.2022.11.006_b0025
Catozzi
Synlett,
2007
6.

Savchuk M.I., Shtaitz Y.K., Kopchuk D.S., Zyryanov G.V., Eltsov O.S., Pospelova T.А., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2019
7.

Bachollet S.P., Volz D., Fiser B., Münch S., Rönicke F., Carrillo J., Adams H., Schepers U., Gómez-Bengoa E., Bräse S., Harrity J.P.
Chemistry - A European Journal,
2016
8.

Prokhorov A.M., Kozhevnikov D.N.
Chemistry of Heterocyclic Compounds,
2012
9.

Birckner E., Grummt U.-., Göller A.H., Pautzsch T., Egbe D.A., Al-Higari M., Klemm E.
Journal of Physical Chemistry A,
2001
10.

Harriman A., Hissler M., Ziessel R.
Physical Chemistry Chemical Physics,
1999
11.

Vicente J., Gil-Rubio J., Barquero N., Jones P.G., Bautista D.
Organometallics,
2008
12.

Mizuyama N., Tominaga Y., Kohra S., Ueda K., Hirayama S., Shigemitsu Y.
Bulletin of the Chemical Society of Japan,
2006
13.

Sellstedt M., Nyberg A., Rosenbaum E., Engström P., Wickström M., Gullbo J., Bergström S., Johansson L.B., Almqvist F.
European Journal of Organic Chemistry,
2010
14.

Camerel F., Ulrich G., Ziessel R.
Organic Letters,
2004
15.
10.1016/j.mencom.2022.11.006_b0075
Lange
Patent WO,
2003
16.
TESHIMA Y., SHIN-YA K., SHIMAZU A., FURIHATA K., CHUL H.S., FURIHATA K., HAYAKAWA Y., NAGAI K., SETO H.
Journal of Antibiotics,
1991
17.

Boger D.L., Panek J.S.
Journal of Organic Chemistry,
1981
18.
10.1016/j.mencom.2022.11.006_b0090
Niewerth
Antimicrob. Agents Chemother.,
1805
19.

Koster G., Bekema H.J., Wetterslev J., Gluud C., Keus F., van der Horst I.C.
Intensive Care Medicine,
2016
20.

TePaske M.R., Gloer J.B., Wicklow D.T., Dowd P.F.
Tetrahedron Letters,
1991
21.
10.1016/j.mencom.2022.11.006_b0105
Morimoto
Patent WO,
1999
22.

Hagimori M., Mizuyama N., Tominaga Y., Mukai T., Saji H.
Dyes and Pigments,
2015
23.
Zyryanov G.V., Kopchuk D.S., Kovalev I.S., Santra S., Rahman M., Khasanov A.F., Krinochkin A.P., Taniya O.S., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2020
24.
Kopchuk D.S., Khasanov A.F., Kovalev I.S., Zyryanov G.V., Rusinov V.L., Chupakhina O.N.
Mendeleev Communications,
2013
25.

Lorion M., Guillaumet G., Brière J., Suzenet F.
Organic Letters,
2015
26.
Chepchugov N.V., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2016
27.
10.1016/j.mencom.2022.11.006_b0135
Kopchuk
Proc.,
2020
28.
![Combination of the SNH/aza-Diels–Alder Reactions as Effective Synthetic Approach to 8-Hydroxy(methoxy)-Substituted 2-[6-(1-Methylindol-3-yl)pyridin-2-yl]quinoline Ligands/Fluorophores](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Savchuk М.I., Kopchuk D.S., Egorov I.N., Khasanov А.F., Rybakova S.S., Zyryanov G.V., Rusinov V.L., Chupakhin О.N.
Russian Journal of General Chemistry,
2021
29.

Kopchuk D.S., Khasanov A.F., Chepchugov N.V., Kovalev I.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Russian Journal of Organic Chemistry,
2017
30.

Fernández Sainz Y., Raw S.A., Taylor R.J.
Journal of Organic Chemistry,
2005
31.

Fershtat L.L., Larin A.A., Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Ananyev I.V., Makhova N.N.
RSC Advances,
2016
32.
![Benzyne-mediated rearrangement of 3-(2-pyridyl)-1,2,4-triazines into 10-(1H-1,2,3-triazol-1-yl)pyrido[1,2-a]indoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Nikonov I.L., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Khasanov A.F., Slepukhin P.A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2013
33.

Kopchuk D.S., Nikonov I.L., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Chemistry of Heterocyclic Compounds,
2014
34.

Kopchuk D.S., Chepchugov N.V., Taniya O.S., Khasanov A.F., Giri K., Kovalev I.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2016
35.

Kopchuk D.S., Nikonov I.L., Khasanov A.F., Giri K., Santra S., Kovalev I.S., Nosova E.V., Gundala S., Venkatapuram P., Zyryanov G.V., Majee A., Chupakhin O.N.
Organic and Biomolecular Chemistry,
2018
36.

Krinochkin A.P., Guda M.R., Rammohan A., Kopchuk D.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Chimica Techno Acta,
2020
37.
10.1016/j.mencom.2022.11.006_b0185
Shtaitz
Proc.,
2019
38.

Savchuk M.I., Starnovskaya E.S., Shtaitz Y.K., Kopchuk D.S., Nosova E.V., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Russian Journal of General Chemistry,
2018
39.

Krayushkin M.M., Sedishev I.P., Yarovenko V.N., Zavarzin I.V., Kotovskaya S.K., Kozhevnikov D.N., Charushin V.N.
Russian Journal of Organic Chemistry,
2008
40.

Shafikov M.Z., Kozhevnikov D.N., Bodensteiner M., Brandl F., Czerwieniec R.
Inorganic Chemistry,
2016
41.

Alonso F., Candela P., Gómez C., Yus M.
Advanced Synthesis and Catalysis,
2003
42.

Zhang Y., Tang Q., Luo M.
Organic and Biomolecular Chemistry,
2011
43.
Ren F., Zhang Y., Hu L., Luo M.
Arkivoc,
2013
44.

Zhou B., Song J., Zhou H., Wu T., Han B.
Chemical Science,
2016
45.

Kozhevnikov D.N., Kozhevnikov V.N., Kovalev I.S., Rusinov V.L., Chupakhin O.N., Aleksandrov G.G.
Russian Journal of Organic Chemistry,
2002
46.

Rao R.N., Chanda K.
Chemical Communications,
2022
47.
John Orie K., Ukachukwu Duru R., I-oro Ngochindo R.
2021
48.
10.1016/j.mencom.2022.11.006_b0240
Marinescu
Int. J. Pharm. Biol. Sci.,
2017