Keywords
C–H functionalization
furazanopyrazines
hydrocarbons
iron trichloride
organic semiconductors
polycyclic aromatic
SH-reaction
Scholl reaction
Abstract
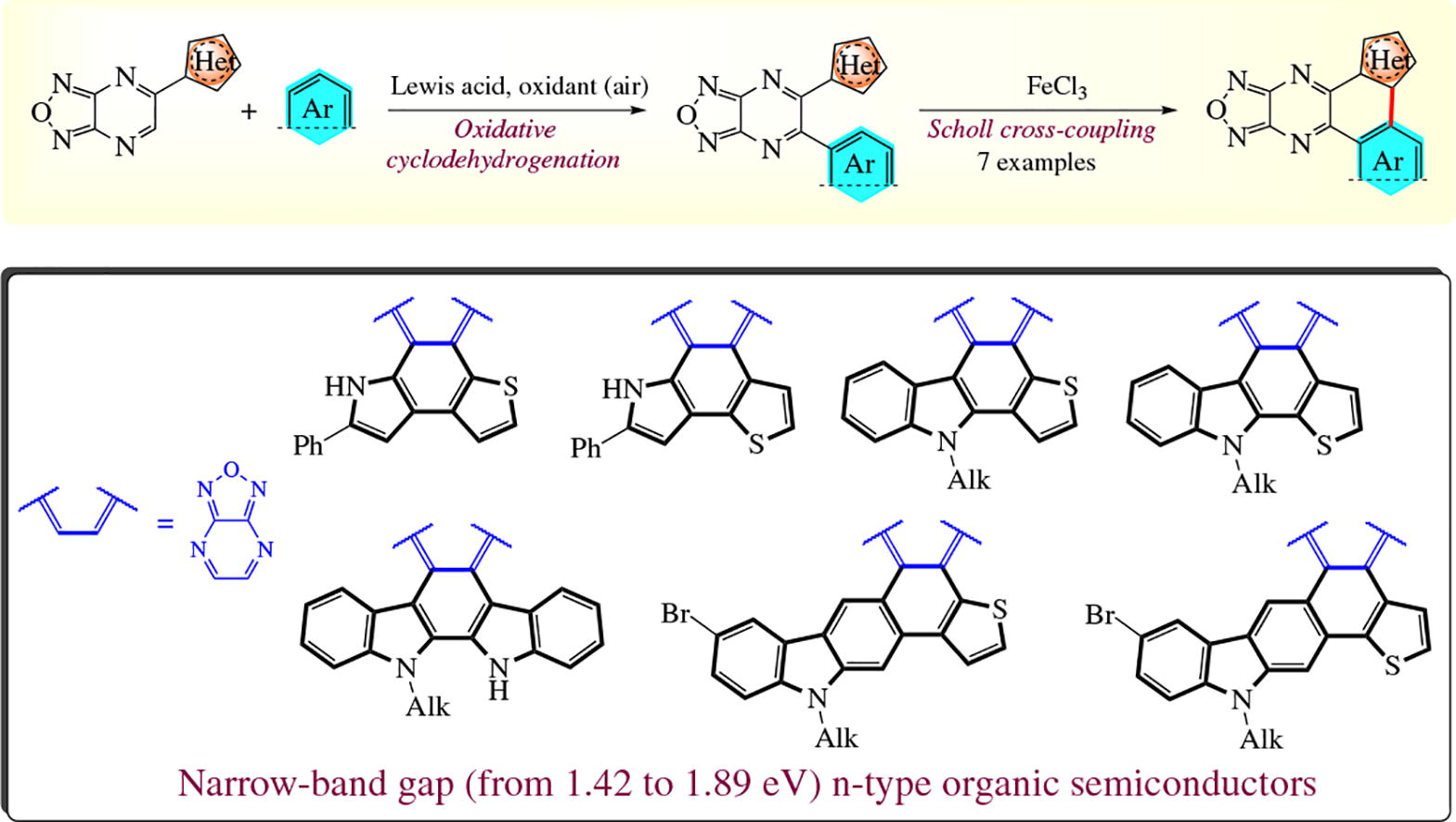
A facile synthetic protocol to hard-to-get polycyclic (hetero)-aromatic compounds annulated to [1,2,4]oxadiazolo[3,4-b]-pyrazine scaffold via the FeCl3-mediated intramolecular Scholl cross-coupling has been developed. Based on the electrochemical and photophysical measurements, the synthesized polycyclic systems may be regarded as narrow band-gap (from 1.42 to 1.89 eV) n-type organic semiconductors whose energy levels are comparable to those of the commercially available n-type semiconductors
References
1.

Feng X., Pisula W., Müllen K.
Pure and Applied Chemistry,
2009
2.

Grzybowski M., Sadowski B., Butenschön H., Gryko D.T.
Angewandte Chemie - International Edition,
2020
3.

Li J., Hao L., Li J.
Science China Technological Sciences,
2019
4.

Borissov A., Maurya Y.K., Moshniaha L., Wong W., Żyła-Karwowska M., Stępień M.
Chemical Reviews,
2021
5.

Biagiotti G., Perini I., Richichi B., Cicchi S.
Molecules,
2021
6.
(a) M. Grzybowski, K. Skonieczny, H. Butenschçn and D. T. Gryko, Angew. Chem., Int. Ed., 2013, 52, 9900; (b) R. S. Jassas, E. U. Mughal, A. Sadiq, R. I. Alsantali, M. M. Al-Rooqi, N. Naeem, Z. Moussa and S. A. Ahmed, RSC Adv., 2021, 11, 32158; (c) M. S. Little, S. G. Yeates, A. A. Alwattar, K. W. J. Heard, J. Raftery, A. C. Edwards, A. V. S. Parry and P. Quayle, Eur. J. Org. Chem., 2017, 1694; (d) C. Maeda, S. Nomoto, K. Akiyama, T. Tanaka and T. Ema, Chem. – Eur. J., 2021, 27, 15699.
7.

Mohr B., Enkelmann V., Wegner G.
Journal of Organic Chemistry,
1994
8.
Verbitskiy E.V., Rusinov G.L., Charushin V.N.
Arkivoc,
2017
9.

Kvashnin Y.A., Verbitskiy E.V., Zhilina E.F., Rusinov G.L., Chupakhin O.N., Charushin V.N.
ACS Omega,
2020
10.
![Dibenzo[f,h]furazano[3,4-b]quinoxalines: Synthesis by Intramolecular Cyclization through Direct Transition Metal-Free C–H Functionalization and Electrochemical, Photophysical, and Charge Mobility Characterization](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Kvashnin Y.A., Verbitskiy E.V., Eltsov O.S., Slepukhin P.A., Tameev A.R., Nekrasova N.V., Rusinov G.L., Nunzi J., Chupakhin O.N., Charushin V.N.
ACS Omega,
2020
11.
Verbitskiy E.V., Dinastiya E.M., Eltsov O.S., Zhilina E.F., Schepochkin A.V., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Mendeleev Communications,
2020
12.

Verbitskiy E.V., Eltsov O.S., Zhilina E.F., Pakhomov I.M., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Tetrahedron,
2019
13.

Verbitskiy E.V., Dinastiya E.M., Baranova A.A., Eltsov O.S., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Chemistry of Heterocyclic Compounds,
2017
14.
Kvashnin Y.A., Kazin N.A., Verbitskiy E.V., Svalova T.S., Ivanova A.V., Kozitsina A.N., Slepukhin P.A., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Arkivoc,
2016
15.

Purushotham U., Sastry G.N.
Physical Chemistry Chemical Physics,
2013
16.

Tang M.L., Oh J.H., Reichardt A.D., Bao Z.
Journal of the American Chemical Society,
2009
17.
Online catalog of chemical reagents Sigma-Aldrich (fullerene-C60), https://www.sigmaaldrich.com/RU/en/product/aldrich/572500.
18.
Online catalog of chemical reagents Sigma-Aldrich (perylene-3,4,9,10- tetracarboxylic dianhydride), https://www.sigmaaldrich.com/RU/en/ product/aldrich/p11255.