Keywords
N-(alkoxymethyl)nitramines
azidation
azides
formation enthalpy
halo alcohols
nitramines
α,ω-dichloro-β-nitrazaalkanes
Abstract
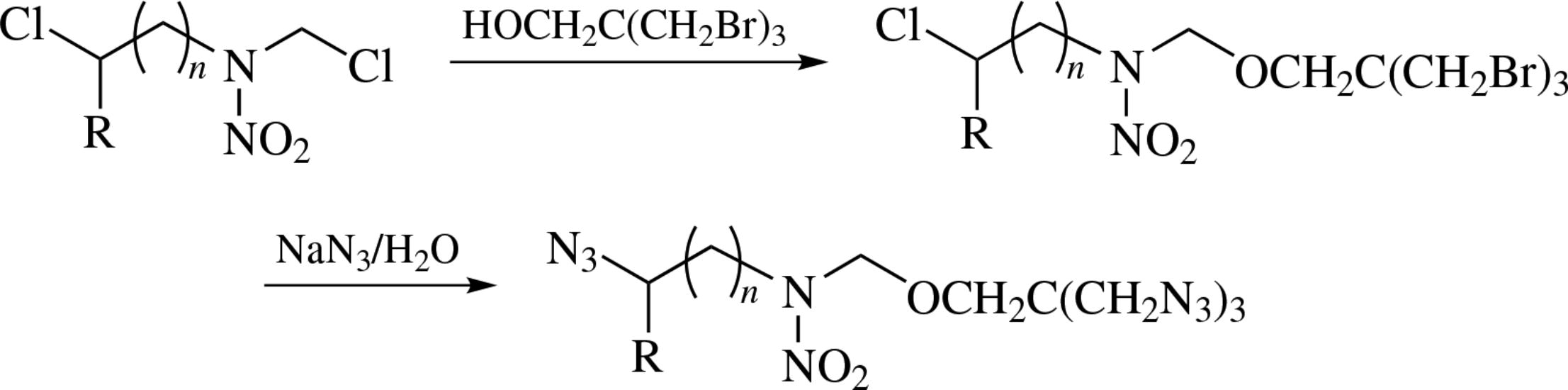
α,ω-Dichloro-β-nitrazaalkanes on treatment with (poly)halo alcohols undergo chemoselective substitution in the activated N-chloromethyl moiety. The thus prepared polyhalo derivatives are converted into the corresponding polyazido compounds which can be the promising energetic plasticizers and monomers.
References
1.
10.1016/j.mencom.2022.11.004_b0005
Chute
J. Can. Res., Sect. B,
1948
2.
10.1016/j.mencom.2022.11.004_b0010
Blomquist
US Patent,
1949
3.
V. M. Zinov’ev, G. V. Kutsenko and A. S. Ermilov, Sovremennye i perspektivnye vysokoenergeticheskie komponenty smesevykh i ballistitnykh tverdykh raketnykh topliv (Modern and Prospective Energetic Components of Composite and Ballistic Solid Propellants), Izd. PGTU, Perm, 2010 (in Russian).
4.
R. L. Simmons, 25th International Annual Conference ICT ‘Thermochemistry of NENA Plasticizers’, Karlsruhe, 1994, 10/1.
5.

Rao K. ., Sikder A., Kulkarni M., Bhalerao M., Gandhe B.
Propellants, Explosives, Pyrotechnics,
2004
6.

Witucki E.F., Wilson E.R., Flanagan J.E., Frankel M.B.
Journal of Chemical & Engineering Data,
1983
7.
10.1016/j.mencom.2022.11.004_b0035
Sinev
J. Org. Chem.,
1982
8.
J. E. Flanagan, E. R. Wilson and M. B. Frankel, US Patent 5013856, 1991.
9.

Unterhalt B., Leiblein F.
Archiv der Pharmazie,
1978
10.
10.1016/j.mencom.2022.11.004_b0050
Spiridonov
SU Patent,
1976
11.
Vinogradov D.B., Bulatov P.V., Petrov E.Y., Tartakovsky V.A.
Mendeleev Communications,
2021