Keywords
azoxy compounds
combustion calorimetry
differential scanning calorimetry
enthalpy of formation
furazans
tetrazoles
X-ray diffraction
Abstract
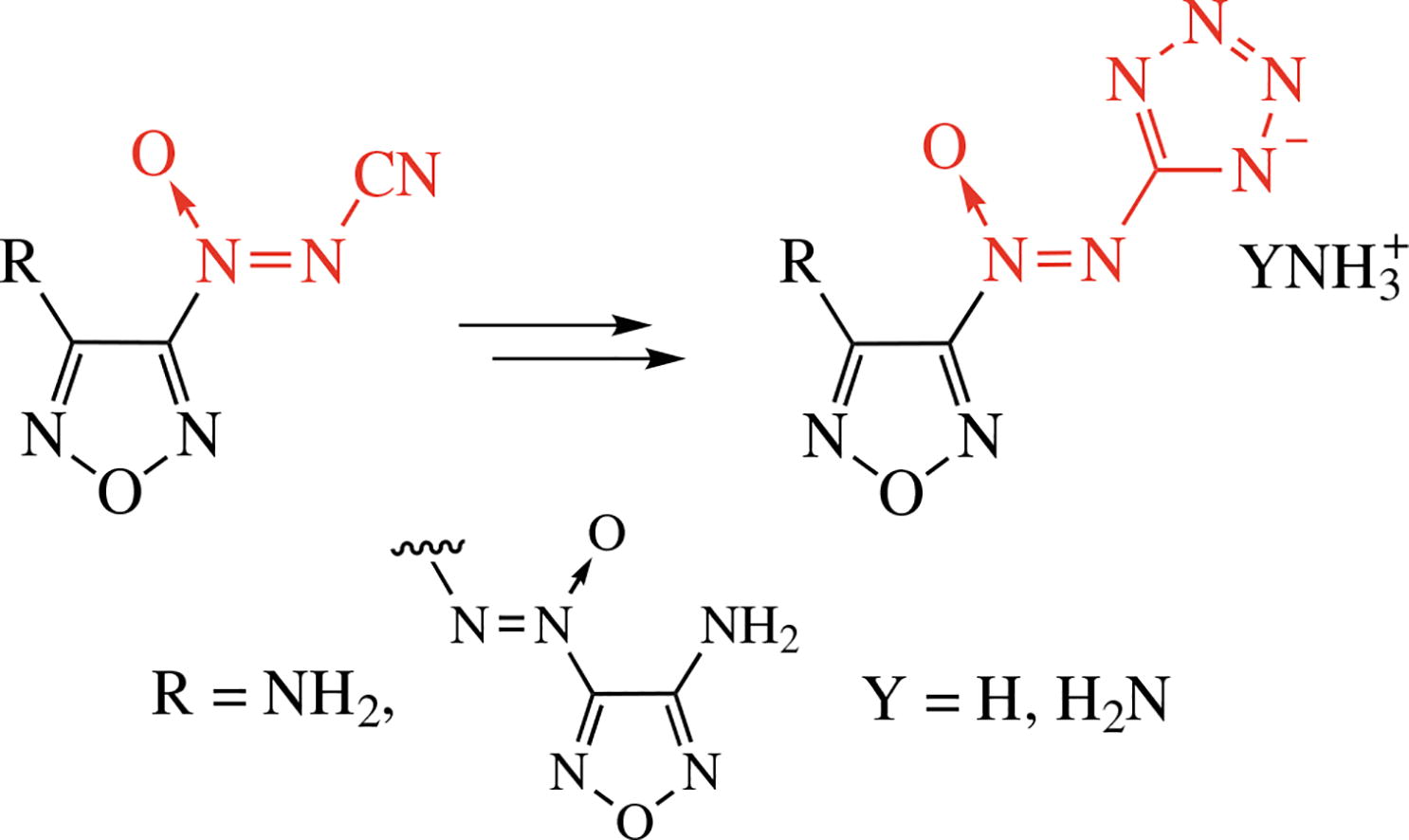
Six novel energetic furazans containing tetrazol-5-yl-NNO-azoxy moiety were synthesized using (cyano-NNO-azoxy)-furazans as starting compounds. The obtained compounds exhibit high enthalpies of formation (531–792 kcal kg–1), acceptable densities (1.70–1.76 g cm–3), good thermal stability (Tonset = 146–199 °C), and, as a result, excellent detonation performance (detonation velocities of 8.61–8.95 km s−1 and detonation pressures of 31.6–36.0 GPa).
References
1.
10.1016/j.mencom.2022.11.002_b0005
Agrawal
High Energy Materials: Propellants,
2010
2.
10.1016/j.mencom.2022.11.002_b0010
DeLuca
Chemical Rocket Propulsion: A Comprehensive Survey of Energetic Materials,
2017
3.
Zlotin S.G., Churakov A.M., Egorov M.P., Fershtat L.L., Klenov M.S., Kuchurov I.V., Makhova N.N., Smirnov G.A., Tomilov Y.V., Tartakovsky V.A.
Mendeleev Communications,
2021
4.
10.1016/j.mencom.2022.11.002_b0020
Dey
Cent. Eur. J. Energ. Mater.,
2015
5.
10.1016/j.mencom.2022.11.002_b0025
Sutton
Rocket Propulsion Elements,
2001
6.
10.1016/j.mencom.2022.11.002_b0030
Kubota
Propellants and Explosives: Thermochemical Aspects of Combustion,
2002
7.

Yanovskii L.S., Lempert D.B., Raznoschikov V.V., Aver’kov I.S.
Russian Journal of Applied Chemistry,
2019
8.
10.1016/j.mencom.2022.11.002_b0040
Lempert
Russ. Chem. Bull.,
1856
9.
![Kinetic Fundamental Aspects of Heat Release in Thermal Decomposition of 7-Amino-7H-difurazano[3,4-b:3′4′-f] furoxano[3″4″-d]azepine and Binary Fuel on Its Basis](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Kazakov A.I., Lempert D.B., Nabatova A.V., Dashko D.V., Raznoschikov V.V., Yanovskii L.S., Aldoshin S.M.
Russian Journal of Applied Chemistry,
2019
10.

Yin P., Zhang Q., Shreeve J.M.
Accounts of Chemical Research,
2015
11.

Yin P., Shreeve J.M.
Advances in Heterocyclic Chemistry,
2017
12.

Zhang J., Zhou J., Bi F., Wang B.
Chinese Chemical Letters,
2020
13.

Fershtat L.L., Makhova N.N.
ChemPlusChem,
2019
14.

Dalinger I.L., Shkineva T.K., Vatsadze I.A., Kormanov A.V., Kozeev A.M., Suponitsky K.Y., Pivkina A.N., Sheremetev A.B.
FirePhysChem,
2021
15.
10.1016/j.mencom.2022.11.002_b0075
Klapötke
Allg. Chem.,
1867
16.

17.

Klapötke T.M., Stein M., Stierstorfer J.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2008
18.

Wu B., Wang Z., Yang H., Lin Q., Ju X., Lu C., Cheng G.
New Journal of Chemistry,
2015
19.
10.1016/j.mencom.2022.11.002_b0095
Voronin
Energy Mater.,
2020
20.

Andrianov V.G., Eremeev A.V.
Chemistry of Heterocyclic Compounds,
1994
21.

Godovikova T.I., Vorontsova S.K., Konyushkin L.D., Firgang S.I., Rakitin O.A.
Russian Chemical Bulletin,
2009
22.

Wang R., Guo Y., Zeng Z., Twamley B., Shreeve J.
Chemistry - A European Journal,
2009
23.

Wang B., Zhang G., Huo H., Fan Y., Fan X.
Chinese Journal of Chemistry,
2011
24.

Leonard P.W., Chavez D.E., Pagoria P.F., by X-ray studies, Parrish D.L.
Propellants, Explosives, Pyrotechnics,
2011
25.

Liang L., Huang H., Wang K., Bian C., Song J., Ling L., Zhao F., Zhou Z.
Journal of Materials Chemistry A,
2012
26.

Huang H., Shi Y., Li Y., Liu Y., Yang J.
RSC Advances,
2016
27.
![[(3-Nitro-1H-1,2,4-triazol-1-yl)-NNO-azoxy]furazans: energetic materials containing an N(O)N–N fragment](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Gulyaev D.A., Klenov M.S., Churakov A.M., Strelenko Y.A., Fedyanin I.V., Lempert D.B., Kosareva E.K., Kon'kova T.S., Matyushin Y.N., Tartakovsky V.A.
RSC Advances,
2021
28.

Leonov N.E., Klenov M.S., Anikin O.V., Churakov A.M., Strelenko Y.A., Voronin A.A., Lempert D.B., Muravyev N.V., Fedyanin I.V., Semenov S.E., Tartakovsky V.A.
ChemistrySelect,
2020
29.

Zlotin S.G., Podgurskii A.I., Airapetova N.V., Luk'yanov O.A.
Russian Chemical Bulletin,
1991
30.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
31.
Leonov N.E., Sidorov F.M., Klenov M.S., Churakov A.M., Strelenko Y.A., Pivkina A.N., Fedyanin I.V., Lempert D.B., Kon'kova T.S., Matyushin Y.N., Tartakovsky V.A.
Mendeleev Communications,
2021
32.

Liu Y., He C., Tang Y., Imler G.H., Parrish D.A., Shreeve J.M.
Dalton Transactions,
2018
33.

Kumar D., Imler G.H., Parrish D.A., Shreeve J.M.
Journal of Materials Chemistry A,
2017
34.
10.1016/j.mencom.2022.11.002_b0170
Sheldrick
Acta Crystallogr.,
2015
35.

Coelho A.A.
Journal of Applied Crystallography,
2003
36.

Coelho A.A.
Journal of Applied Crystallography,
2018
37.

Favre-Nicolin V., Černý R.
Journal of Applied Crystallography,
2002
38.

Luk′yanov O.A., Parakhin V.V., Shlykova N.I., Dmitrienko A.O., Melnikova E.K., Kon'kova T.S., Monogarov K.A., Meerov D.B.
New Journal of Chemistry,
2020
39.
10.1016/j.mencom.2022.11.002_b0195
Sumin
Transactions of the 11th International Detonation Symposium,
1998
40.

Muravyev N.V., Meerov D.B., Monogarov K.A., Melnikov I.N., Kosareva E.K., Fershtat L.L., Sheremetev A.B., Dalinger I.L., Fomenkov I.V., Pivkina A.N.
Chemical Engineering Journal,
2021
41.
10.1016/j.mencom.2022.11.002_b0205
Meyer
Explosives,
2016