Keywords
magnesium citrate
Mg3(C6H5O7)2·10H2O
nanocrystalline magnesium oxide
thermolysis mechanism
X-ray amorphous products
Abstract
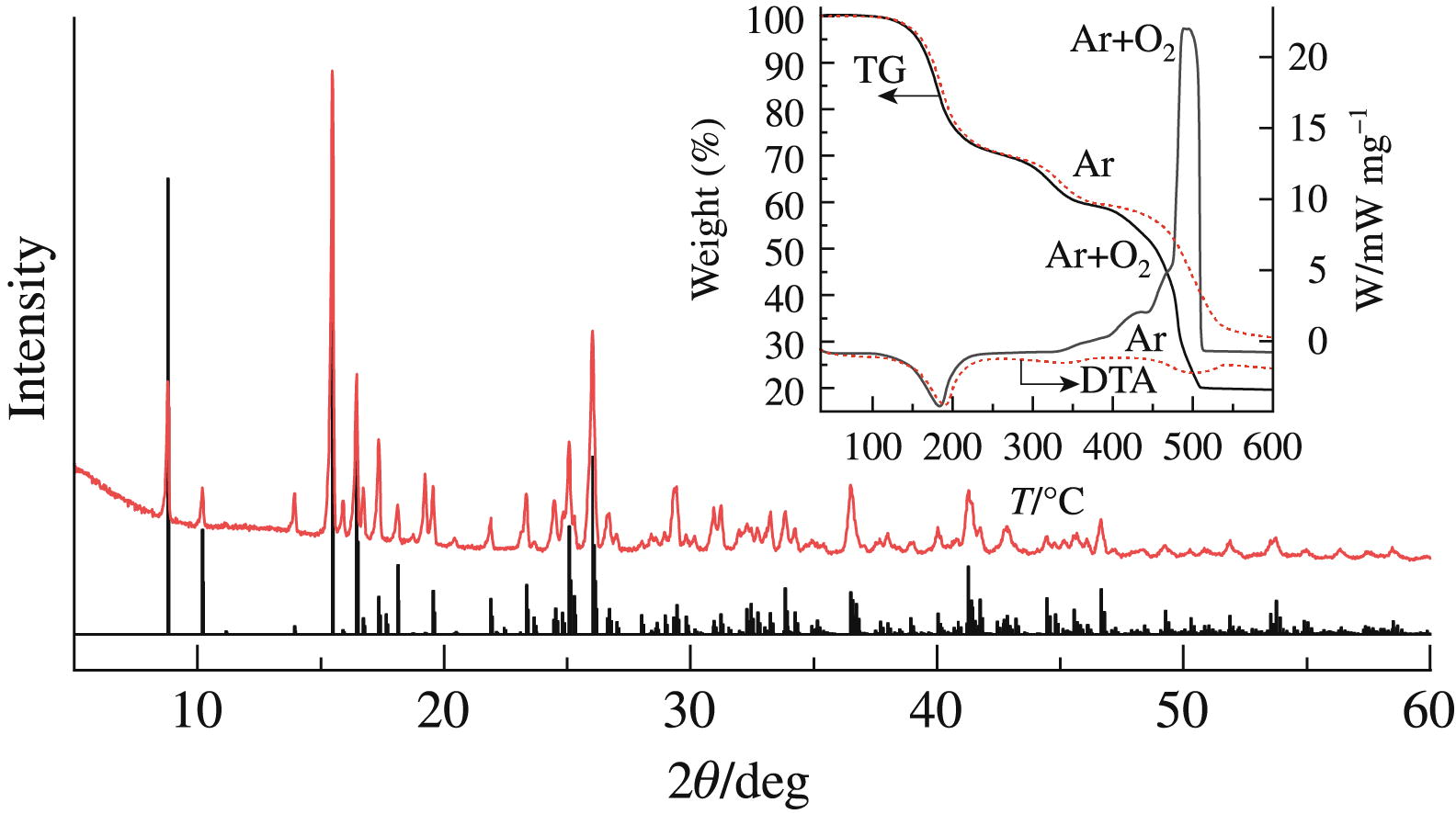
Magnesium citrate, obtained by dissolving magnesium oxide in an aqueous solution of citric acid, turned out to be a decahydrate of the composition Mg3(C6H5O7)2·10H2O, which was established by the results of synchronous thermal analysis, X-ray diffraction and IR spectroscopy. It is shown that the thermal decomposition of this salt proceeds in three stages in the temperature ranges of 120–250, 250–370 and 370–550 °C to form nanocrystalline magnesium oxide with grain sizes from 7 to 23 nm.
References
1.

Inagaki M., Toyoda M., Soneda Y., Tsujimura S., Morishita T.
Carbon,
2016
2.

Teng W., Wu Z., Fan J., Chen H., Feng D., Lv Y., Wang J., Asiri A.M., Zhao D.
Energy and Environmental Science,
2013
3.

Abbas Q., Mirzaeian M., Ogwu A.A.
International Journal of Hydrogen Energy,
2017
4.

Zhu S., Zhao N., Li J., Deng X., Sha J., He C.
Nano Today,
2019
5.

Hard template synthesis of crystalline mesoporous anatase TiO2 for photocatalytic hydrogen evolution
Zhang Z., Zuo F., Feng P.
Journal of Materials Chemistry A,
2010
6.

Morishita T., Tsumura T., Toyoda M., Przepiórski J., Morawski A.W., Konno H., Inagaki M.
Carbon,
2010
7.

Kado Y., Imoto K., Soneda Y., Yoshizawa N.
Journal of Power Sources,
2014
8.

Konno H., Onishi H., Yoshizawa N., Azumi K.
Journal of Power Sources,
2010
9.

Liu W., Wang C., Wang J., Jia F., Zheng J., Chen M.
Chinese Science Bulletin,
2013
10.

Mansour S.A.
Thermochimica Acta,
1994
11.
![X-ray crystal analysis of the substrates of aconitase. V. Magnesium citrate decahydrate [Mg(H2O)6][MgC6H5O7(H2O)]2.2H2O](/storage/images/resized/xG3oC19920mmFVqkoVEGKGaMCeoalzyvWxPkVox5_small_thumb.webp)
Johnson C.K.
Acta Crystallographica,
1965
12.

Kaduk J.A.
Acta Crystallographica Section E: Crystallographic Communications,
2020
13.
10.1016/j.mencom.2022.09.044_b0065
Goldberg
Encyclopedia of Microbiology,
2009